Abstract
Atezolizumab is a programmed cell death-ligand 1 antibody that modulates the immune system response and has shown great promise in treating malignancies. Cutaneous toxicities from immune checkpoint inhibitors are the most commonly reported immune-related adverse events, although toxicities related to immunotherapy are still being characterized. Herein, we present a novel case of inflamed actinic keratoses in a patient after receiving atezolizumab therapy that resolved without requirement of dose adjustment or discontinuation of treatment.
Introduction
Programmed cell death-1 (PD-1) is a cell surface receptor on activated T cells that binds programmed cell death-ligand 1 (PD-L1) on a host of cells, activating downstream signaling pathways and inhibiting T cell function. PD-1/PD-L1 inhibitors prevent the interaction of PD-1/PD-L1 receptors, blunting immune tolerance and facilitating the generation of stronger immune responses. 1 Cancer immunotherapy utilizing anti-PD-1/PD-L1 therapy has shown great promise in treating malignancies, but toxicities related to the immune checkpoint antibodies are still being characterized. Cutaneous toxicities are the most frequently reported immune-related adverse events (irAEs) induced by anti-PD-1/PD-L1 agents, including maculopapular eruption, pruritus, dyspigmentation, vitiligo, psoriasis, lichenoid dermatitis, bullous pemphigoid, Stevens–Johnson syndrome/toxic epidermal necrolysis, and inflamed disseminated actinic porokeratosis.2–5
Herein, we report a novel case of inflamed actinic keratoses (AKs), confirmed by histopathology, in a patient after receiving atezolizumab (PD-L1 antibody). Although inflammation of pre-existing AKs has been reported with other systemic chemotherapeutic agents, including 5-fluorouracil (FU), pembrolizumab, and numerous cytotoxic agents, we present the first reported case to our knowledge of such a reaction secondary to anti-PD-L1 therapy.3,5,6
Case report
A 51-year-old female with diffuse large B-cell lymphoma and no skin cancer history was evaluated by the inpatient dermatology consult service for a new onset rash. She is enrolled in clinical trial NCI10126 and was receiving atezolizumab in combination with gemcitabine–oxaliplatin and rituximab. For her first infusion, she received gemcitabine–oxaliplatin and rituximab, without skin toxicity. Her second infusion administered 2 weeks later, included atezolizumab in addition to the aforementioned chemotherapies. Within 24 h after this second infusion, she developed a new rash.
The eruption consisted of scaly erythematous macules and papules involving the forehead, cheeks, nose, and chin, with notable sparing of the skin above the upper lip (Figure 1(a) and (b)). Similar lesions were present on the upper chest, upper back, and bilateral arms in a photodistributed pattern. A shave biopsy of a representative lesion on the left temple showed parakeratosis, hypogranulosis, basilar keratinocytic atypia, and subjacent solar elastosis, consistent with AK. In addition, there were multifocal aggregates of lymphohistiocytic inflammation noted within the superficial reticular dermis (Figure 2). Given the clinical examination findings, medication administration timeline, and histopathologic findings, we diagnosed our patient with inflamed AKs induced by atezolizumab. The patient was prescribed hydrocortisone 2.5% cream for twice-daily application and noticed resolution of the macules and papules within less than a week (Figure 3). The patient remained on treatment protocol without requirement for dose adjustment or discontinuation of atezolizumab.

Clinical photos of characteristic lesions. Several erythematous macules and papules on the the face (a) with a close up (b) in a photodistributed pattern.

Histological slide from shave biopsy of the left temple demonstrating parakeratosis, hypogranulosis, basilar keratinocytic atypia, subjacent solar elastosis, and lymphohistiocytic inflammation in the superficial reticular dermis, consistent with inflamed actinic keratosis.
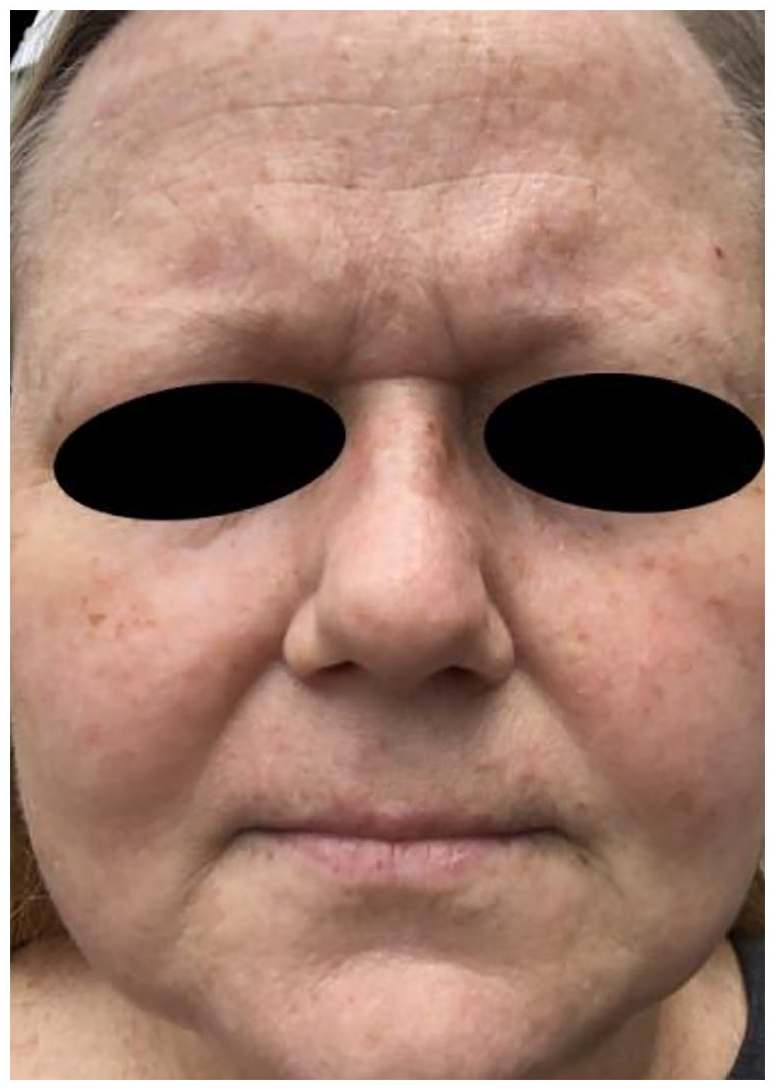
Follow-up photo showing resolution of erythematous macules and papules after twice-daily application of hydrocortisone 2.5% cream for 1 week.
Discussion
To our knowledge, this is the first reported case of atezolizumab-induced inflammation of AKs. This reaction has been reported following administration of systemic chemotherapeutics, including cisplatin, capecitabine, doxorubicin, pentostatin, pembrolizumab, and thioguanine.5,7 Clinical findings typically become apparent within 1 week of regimen initiation.7,8 Our patient developed a reaction within 24 h after her first atezolizumab infusion. Although we cannot definitively rule out gemcitabine or oxaliplatin as causative agents, the timeline of her presentation is most suggestive of reaction to atezolizumab. Gemcitabine and oxaliplatin have not been previously associated with inflammation of AKs.
This phenomenon was first observed by Falkson and Schulz in response to systemic 5-FU, where patients developed erythema and inflammation of AKs with subsequent resolution of erythema and AKs after therapy. 8 Pearse and Marks later demonstrated that perilesional skin adjacent to AKs contains an abnormal number of cells in DNA synthesis at one time, possibly explaining the mechanism of the selective sensitivity of sun-damaged skin to chemotherapeutic agents.9,10 Subsequently, topical formulations of 5-FU were approved for use in field treatment of AKs, allowing for the selective targeting of such lesions with sparing of normal skin. 11 The selective nature of systemic atezolizumab for AKs as seen in our patient demonstrates a similar clinical response to that observed with 5-FU and could prompt future investigation into topical formulations for treatment of AKs.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
The patient provided informed consent for publication of the case report and images.
