Abstract
Cryoglobulinemia is the presence of circulating cryoglobulin which can cause systemic vasculitis and glomerulonephritis. Monoclonal gammopathy of renal significance is strongly associated with type I cryoglobulinemia, but the role of detectable serum monoclonal gammopathy in mixed (type II) cryoglobulinemia is not clearly established. We report a case of a 71-year-old woman who presented with skin rash, leg edema, and azotemia. Investigations showed a positive result for rheumatoid factor, low complement C4 level, positive result for serum cryoglobulin, and positive M-spike on serum protein electrophoresis and IgM kappa monoclonal gammopathy on serum immunofixation. Kidney biopsy revealed membranoproliferative glomerulonephritis, polytypic IgM-dominant deposits in an immunofluorescence study, and microtubular substructures in an electron microscopic study. After an extensive workup, no evidence of myeloma or lymphoma was found. A diagnosis of monoclonal gammopathy of renal significance–associated mixed cryoglobulinemic glomerulonephritis was made. Due to the detectable IgM kappa monoclonal gammopathy in the patient’s serum, clonal-directed therapy was administered. The patient had been in clinical remission after treatment with clone-directed therapy with cyclophosphamide and steroids. The literature review for cases of type II cryoglobulinemic glomerulonephritis that have detectable serum monoclonal gammopathy are summarized in this study.
Introduction
Cryoglobulins are immunoglobulins that reversibly precipitate when the temperature is <37°C and redissolve upon rewarming. 1 Patients with cryoglobulinemia can develop systemic small-vessel vasculitis which involves with multiple organ systems. 2 Cryoglobulinemia is classified based on its immunoglobulin composition according to Brouet’s classification; 3 type I cryoglobulinemia comprises single monoclonal immunoglobulin and is associated with lymphoproliferative disorders; type II cryoglobulinemia is composed of a combination of monoclonal and polyclonal immunoglobulins; type III cryoglobulinemia consists of polyclonal IgM and IgG. Type II and type III cryoglobulins correspond with mixed cryoglobulinemia and are associated with chronic infection, particularly hepatitis C virus (HCV), as well as autoimmune diseases, such as Sjögren syndrome and systemic lupus erythematous. 2
Type I (monoclonal) cryoglobulinemia is produced from an abnormal clone in lymphoproliferative diseases and can deposit in kidney tissue and induce inflammation, which is one of the important causes of monoclonal gammopathy of renal significance (MGRS). For type II cryoglobulinemia, monoclonal proteins are usually discovered from the redissolved cryoprecipitate.4,5 However, an association between a detectable monoclonal gammopathy in patient’s serum and type II cryoglobulinemia (which contains a mixture of both monoclonal and polyclonal immunoglobulins) is not clearly established. The recognition of mixed cryoglobulinemic glomerulonephritis as a presenting feature of MGRS is important, since it is associated with an abnormal B-cell clone. Clinicians should aim for the treatment of an underlying lymphoproliferative disease rather than solely focusing on the treatment of cryoglobulinemia.
This article discusses a case of patients with type II cryoglobulinemic membranoproliferative glomerulonephritis (MPGN) with a detectable monoclonal IgM kappa in serum. In addition, an extensive literature review was conducted to gather all the information about previously reported cases of the same nature to demonstrate the possible association between serum monoclonal gammopathy and type II cryoglobulinemic glomerulonephritis.
Case presentation
A 71-year-old woman who had previously been diagnosed with rheumatoid arthritis presented with generalized edema, worsening hypertension, and non-blanchable erythematous papules with reticulation on both legs, which had been presented for 4 months, as shown in Figure 1. Rheumatoid arthritis had been diagnosed 5 years prior when she presented with polyarthralgia and positivity for rheumatoid factor. Prednisolone, methotrexate, and subcutaneous etanercept were prescribed, and complete clinical remission had been achieved after 2 years of treatment. The medications were then discontinued, and the patient sustained her clinical remission afterward.

Non-blanchable erythematous macules and palpable purpura on patient’s leg(s).
The patient had developed both ankle edema 4 months prior to this admission, which slowly progressed to the pretibial areas. Her urine appeared foamier for 2 months, which was accompanied by rising of blood pressure according to a home monitoring device. Urine color and volume were unchanged from the patient’s perspective. Physical examination indicated high blood pressure (190/94 mm Hg), bilateral pitting edema, and livedo reticularis of both legs. No other skin lesion was found. There was no active arthritis, fever, or lymphadenopathy. The results of the initial investigation are displayed in Table 1, which shows a serum creatinine of 1.86 mg/dL, dysmorphic red blood cells in urine, and proteinuria.
Initial laboratory investigation at presentation.
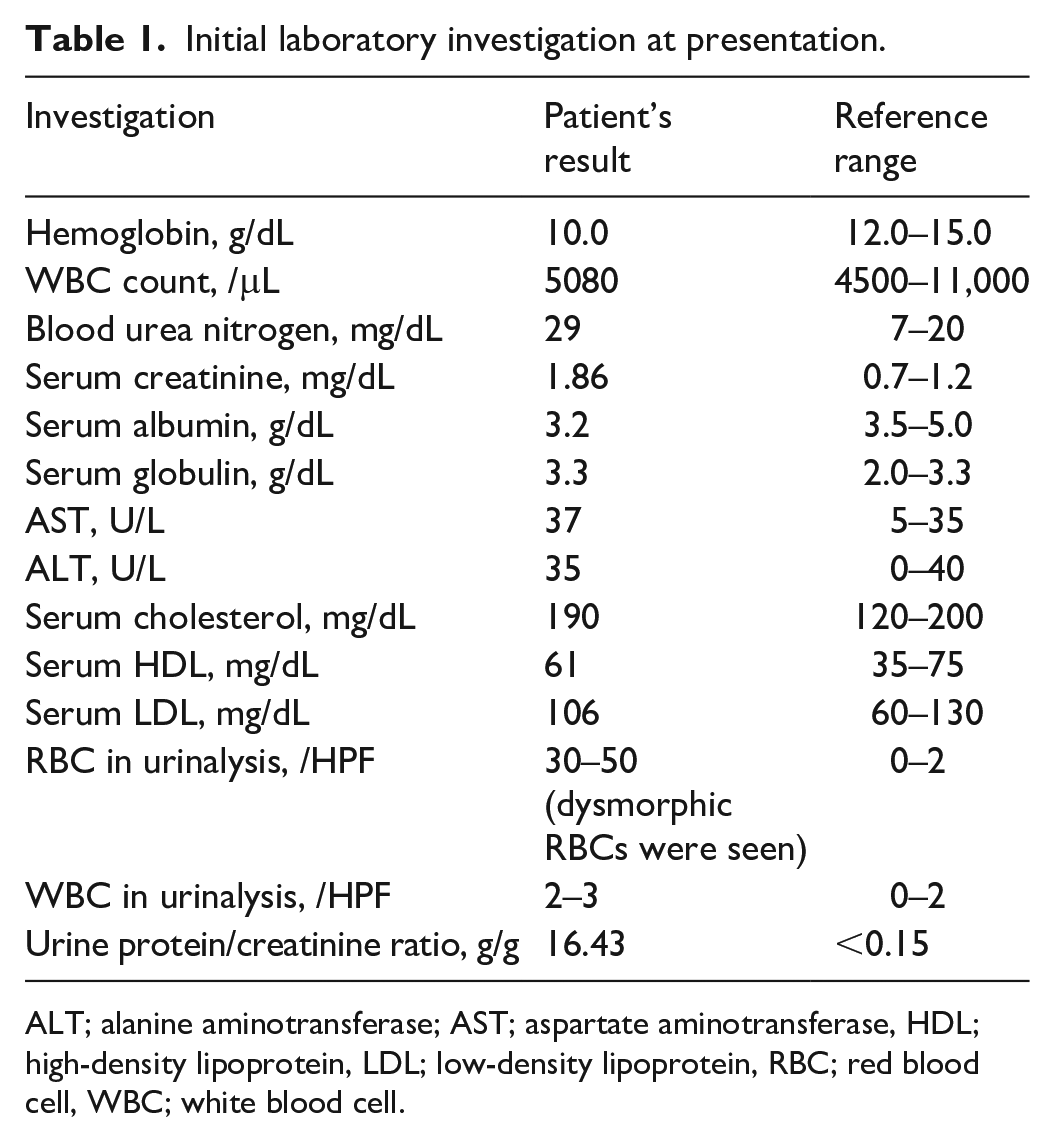
ALT; alanine aminotransferase; AST; aspartate aminotransferase, HDL; high-density lipoprotein, LDL; low-density lipoprotein, RBC; red blood cell, WBC; white blood cell.
Further investigations showed a positive result for antinuclear antibody (ANA) with a homogeneous, fine-speckled, and nucleolar pattern (titer > 1:1280). The level of rheumatoid factor was 54.40 IU/mL (normal < 20 IU/mL). Serum cryoglobulin was positive. Serological tests for HCV and human immunodeficiency virus (HIV) were negative. The serology results for hepatitis B virus (HBV) were negative for HBsAg, positive for anti-HBc, and positive for anti-HBs antibodies. Complement C3 was normal, and C4 was 3.09 mg/dL (10–40 mg/dL).
A percutaneous kidney biopsy was performed and kidney pathology is shown in Figures 2 and 3. The kidney pathology revealed diffuse endocapillary hypercellularity and mesangial expansion with lobular accentuation of glomeruli in hematoxylin and eosin (H&E) staining. Periodic acid-Schiff (PAS) staining demonstrated global and diffuse double-contour thickening of the glomerular basement membrane, which was compatible with a pattern of MPGN. No crescentic lesion was observed.

Kidney biopsy pathology. Light microscopic examination stained with hematoxylin and eosin (a; original magnification, ×400) and periodic acid-Schiff (b; original magnification, ×400). Immunofluorescence study of IgM (c; original magnification, ×400), kappa (d; original magnification, ×400), and lambda (e; original magnification, ×400).

Electron microscopic examination of kidney pathology showing electron-dense deposits in the capillary loop (a; bar = 2 μm). Higher magnification of electron-dense deposits showing organized structures (b; bar = 500 nm).
The immunofluorescence (IF) study revealed predominant IgM (2+) staining. IgG (1+) and C3 (1+) granular staining in mesangium and capillary loops were also observed. Equivalent kappa and lambda staining was found in the capillary loops (1+). C1q and IgA results were negative. Figure 3 displays the results of an electron microscopic (EM) study, which revealed the deposition of organized electron-dense materials at the mesangium and subendothelium. The substructures were composed of short microtubules (~20 nm thickness) with hollow centers. Neomembrane formation and diffuse foot process effacement were also noted.
The positive result for serum cryoglobulin, bilateral skin rash compatible with leukocytoclastic vasculitis, polytypic IgM-dominant deposits in the IF study, and organizing deposits in the EM study help to narrow the diagnosis to cryoglobulinemic glomerulonephritis. The kidney pathology did not show any light-chain restriction since there was equal intensity of kappa and lambda staining, suggesting mixed cryoglobulinemic glomerulonephritis. The underlying cause of cryoglobulinemia was then investigated in this elderly patient.
Common causes of mixed cryoglobulinemia such as HCV and HBV were ruled out by negative serology results. The results of age-appropriate cancer screenings including colonoscopy, mammography, and pelvic examination were all negative. However, serum protein electrophoresis revealed a monoclonal spike of 0.35 g/dL. Serum immunofixation showed IgM kappa monoclonal gammopathy. The immunofixation of cryoprecipitate was not done at that time due to the logistic issue in the out-patient setting. Serum kappa was 248.31 mg/dL (3.3–19.4 mg/dL) and serum lambda was 40.84 mg/dL (5.71–26.30 mg/dL) with a kappa-to-lambda free light-chain ratio of 6.08. These findings raised the possibility of lymphoproliferative disorder. However, bone marrow aspiration and biopsy showed normocellular trilineage marrow without evidence of abnormal clonality, lymphoma, or plasma cell disorders.
Based on all evidence, particularly the detectable monoclonal IgM kappa gammopathy in the patient’s serum despite not revealing lymphoproliferative disease, a diagnosis of MGRS with non-hepatitis-associated mixed cryoglobulinemic glomerulonephritis was suspected. After a multidisciplinary team discussion and informing the patient of all the possibilities, clone-directed therapy was initiated, including a 3-week cycle of oral cyclophosphamide at 200 mg/day and dexamethasone. After six cycles, the bilateral leg edema and rash were completely resolved. Serum creatinine was decreased to 0.83 mg/dL, and proteinuria was 0.49 g/g creatinine. The monoclonal protein level was decreased to 0.13 g/dL, and the serum kappa-to-lambda ratio was 1.3. The patient is still in remission as of 3 years after the completion of treatment.
Discussion
This report discusses a case of detectable serum IgM kappa monoclonal gammopathy with non-hepatitis-associated mixed cryoglobulinemic glomerulonephritis. The diagnosis of mixed cryoglobulinemia was made based on serum cryoglobulin and kidney pathology findings, which indicated MPGN, organized microtubular substructural deposits, and polytypic IgM-dominant staining in the IF study. The patient was treated with clone-directed therapy and has been in renal and hematological remission.
Two important points were raised from the presenting case. The first is the awareness of secondary causes of mixed cryoglobulinemic glomerulonephritis. Previous studies showed that the mean age of patients with essential or non-malignancy-related mixed cryoglobulinemia was 50–60 years.6,7 The new onset of cryoglobulinemic glomerulonephritis in our 71-year-old patients emphasized the necessity of exploring other causes, particularly hematologic malignancy, which has been reported to relate with type I and type II cryoglobulinemia. 8 The use of clone-directed therapy was associated with favorable outcome in cryoglobulinemic glomerulonephritis cases with hematologic conditions. 5 The patient might have received an inadequate treatment if the diagnosis of essential cryoglobulinemia had been made. Second, compared with monoclonal gammopathy detected in cryoprecipitate, the evidence of monoclonal gammopathy that can be detected in serum of patient with type II cryoglobulinemia has been less established, which could serve as another sign for MGRS-related cryoglobulinemia.
The definition of MGRS was established by the International Kidney and Monoclonal Gammopathy (IKMG) research group in 2017 8 as any B-cell or plasma cell clonal lymphoproliferation that does not cause tumor complications or meet any current hematological criteria for specific therapy, with one or more kidney lesions that are regulated to the produced monoclonal immunoglobulin. Kidney lesions associated with MGRS can be classified according to the presence of monoclonal immunoglobulin deposits and the organization of the deposits. The differential diagnosis of MGRS-associated lesions with microtubular substructures, as presented in our case, includes immunotactoid glomerulonephritis and cryoglobulinemic glomerulonephritis. Type I cryoglobulinemic glomerulonephritis is known to be associated with monoclonal gammopathy and lymphoproliferative disorders. Type II cryoglobulinemic glomerulonephritis involves a mixture of monoclonal and polyclonal immunoglobulins and can also be the initial presentation of MGRS-associated lesions. 8
To date, there is still controversy about the role of serum monoclonal gammopathy in type II cryoglobulinemic glomerulonephritis. It is known that type II cryoglobulinemia consists of monoclonal and polyclonal immunoglobulins (which can be demonstrated by the immunofixation of cryoprecipitate). However, unlike cryoprecipitate, the monoclonal protein is not universally detectable in the patient’s serum.9,10 The true prevalence of detectable monoclonal proteins in the serum of type II cryoglobulinemic patients has yet to be identified. A previous study reported a prevalence of 20% for monoclonal protein in the serum of patients with noninfectious mixed cryoglobulinemic glomerulonephritis. 9 More recent study showed a prevalence of 76% for positive serum protein electrophoresis/immunofixation in non-hepatitis-associated cryoglobulinemia patients (combining type I, II, and III). 5 Moreover, an association has yet to be determined between the detectable serum monoclonal antibody and the clinical outcome or prognosis. For example, evidence is lacking whether the patients with detectable level of serum monoclonal antibody might be more related to an underlying hematologic malignancy or worse prognosis, compared with the patients with only detectable monoclonal antibody in the cryoprecipitate.11,12 The response to clone-directed therapy might also be different between these two groups.
Table 2 summarizes a review of the literature of the noninfectious mixed (type II) cryoglobulinemic glomerulonephritis, in which the association with serum monoclonal gammopathy was determined.9,13–18 Interestingly, all of the cases of the noninfectious mixed cryoglobulinemia with positive serum monoclonal gammopathy were linked with serum IgM kappa monoclonal antibody. We speculated that the IgM monoclonal gammopathy might have high autoantibody activity that allows for the formation of an immune complex, as also observed in Waldenström macroglobulinemia. 19 Chauvet et al. 20 reported a case series of any kidney diseases associated with IgM monoclonal gammopathy, in which 5 of 15 cases tested positive for type II cryoglobulin. Almost all of the serum immunofixation results indicated monoclonal IgM kappa antibody (12 of 15 cases). Higgins et al. 21 also reported that type II cryoglobulin is more common than type I cryoglobulin among patients with IgM monoclonal gammopathy associated with glomerulopathy.
Literature review of cases with non-hepatitis-associated mixed (type II) cryoglobulinemic glomerulonephritis in which serum monoclonal gammopathy was evaluated.
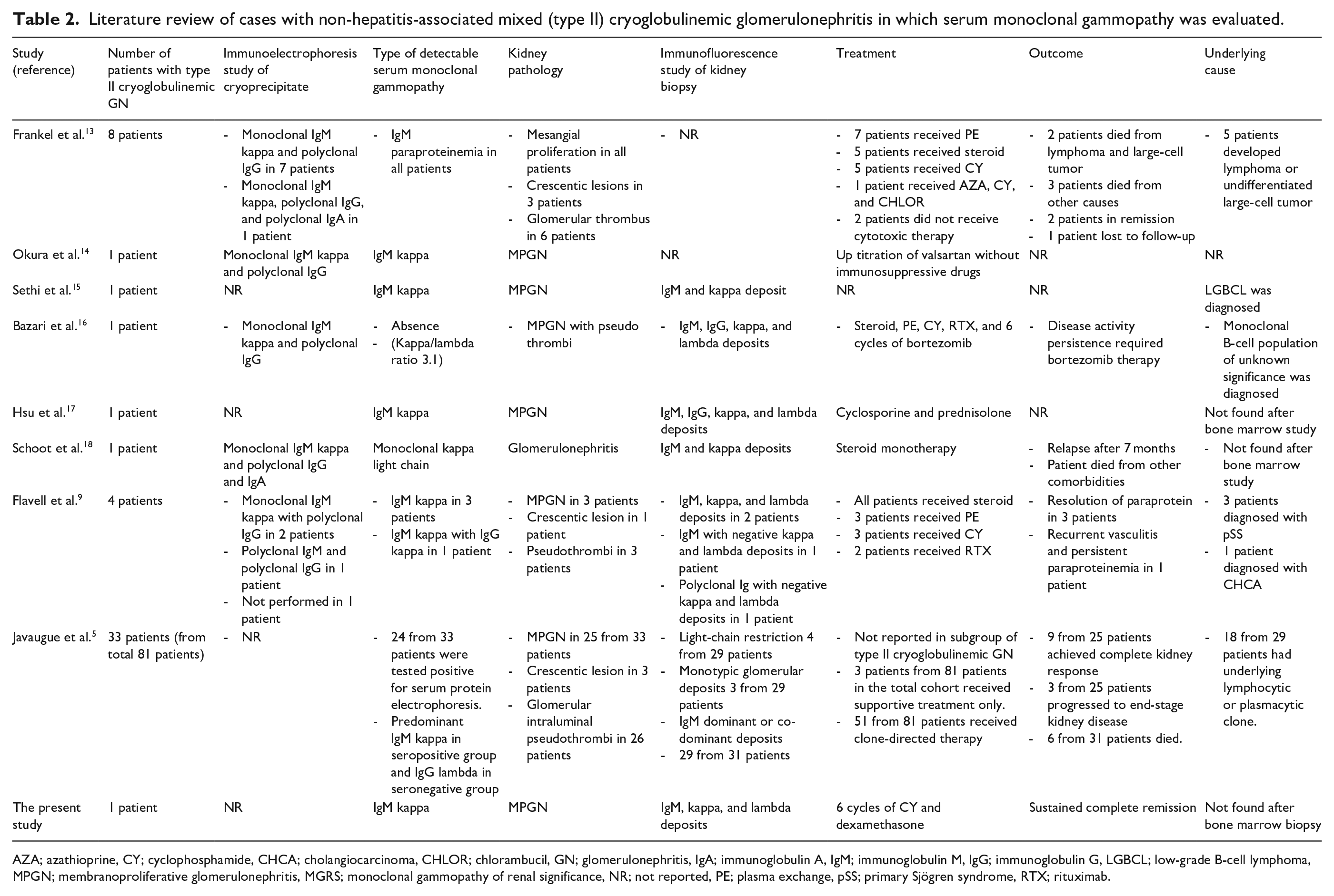
AZA; azathioprine, CY; cyclophosphamide, CHCA; cholangiocarcinoma, CHLOR; chlorambucil, GN; glomerulonephritis, IgA; immunoglobulin A, IgM; immunoglobulin M, IgG; immunoglobulin G, LGBCL; low-grade B-cell lymphoma, MPGN; membranoproliferative glomerulonephritis, MGRS; monoclonal gammopathy of renal significance, NR; not reported, PE; plasma exchange, pSS; primary Sjögren syndrome, RTX; rituximab.
The kidney pathology in our case showed an MPGN pattern with dominant IgM and equivocal kappa and lambda light-chain immunostaining. Although MGRS-associated kidney pathology should have a light-chain restriction, one might speculate another possibility of the immune complexes in MGRS-associated type II cryoglobulinemic glomerulonephritis consisting of mixed components (both monoclonal and polyclonal antibodies). The polyclonal part of the immune complexes could then result in a lack of light-chain restriction, which was also presented in previous reports of patients with lymphoproliferative disorders with monoclonal gammopathy and type II cryoglobulinemic glomerulonephritis.11,16 IF study after pronase digestion of the paraffin section can serve as an alternative method to reveal the masked monoclonal protein. 22
Recognition of MGRS or monoclonal gammopathy in patients with mixed cryoglobulinemia is essential because clone-directed therapy may provide better outcomes compared with the standard immunosuppressive therapy in these cases, although this idea has not been tested in randomized control trials.5,23 The aim of clone-directed therapy includes the preservation of renal function and prevention of MGRS recurrence, especially after kidney transplantation. In our case, clone-directed therapy was administered with six cycles of cyclophosphamide and dexamethasone. Complete renal response and normalization of monoclonal protein were noted. No overt hematological malignancy was detected as of 3 years after treatment completion. However, the data on clone-directed therapy in MGRS are limited. Benefits over conventional immunosuppressive drugs were observed in the retrospective studies of clone-directed therapy in C3 glomerulonephritis 24 and proliferative glomerulonephritis with monoclonal immunoglobulin deposits associated with MGRS. 25 Recently, a retrospective study showed that patients with non-hepatitis-associated cryoglobulinemic glomerulonephritis were associated with hematologic conditions and responded well to clone-directed therapy. 5 There has been no evidence whether the use of clone-directed therapy in patients with monoclonal gammopathy-related mixed cryoglobulinemia but without hematologic malignancy (such as patients with chronic HBV or HCV infection)26–28 could result in better disease control and prevent malignancy transformation.
Conclusion
Detectable monoclonal gammopathy can be found in the serum of patients with type II cryoglobulinemic glomerulonephritis. Although lymphoproliferative disorders are more closely related with type I cryoglobulinemia, it is essential to exclude monoclonal gammopathy in patients with mixed cryoglobulinemic glomerulonephritis to reveal hidden lymphoproliferative disease before the diagnosis of essential cryoglobulinemia can be made. The detection of MGRS or underlying clonal disorders can lead to appropriate treatment that might preserve renal function and prevent recurrence. However, more studies are needed to confirm the benefit of clone-direct therapy over conventional immunosuppression in patients with MGRS-associated mixed cryoglobulinemia.
Footnotes
Acknowledgements
The authors thank to fellows and nurses at the Division of Nephrology and Division of Hematology, Chulalongkorn University for the comprehensive care of this patient.
Author contributions
S.F. involved with the first draft of manuscript, study design, and case reviewing. N.U. was the attending hematologist and involve with the case reviewing. K.I. was the attending pathologist. S.U. was the attending nephologist and involved with manuscript writing, manuscript editing, and study design.
Data availability
The data used during the current case report are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
