Abstract
Scleromyxedema is a rare, chronic mucinosis characterized by widespread skin fibrosis and an associated monoclonal gammopathy. Therapeutic options remain limited. We present the case of a male in his 50s with biopsy-confirmed scleromyxedema and IgG λ monoclonal gammopathy who initially responded to intravenous immunoglobulin but later progressed despite full-dose maintenance. Due to severe pruritus and recent evidence implicating Type 2 cytokines in scleromyxedema pathogenesis, dupilumab was added to ongoing intravenous immunoglobulin. Pruritus improved within 4 months, but fibrosis remained unchanged and M-protein levels continued to rise, reaching 35.3 g/L by month 12. Dupilumab was discontinued due to lack of disease modification. Bone marrow biopsy confirmed smoldering multiple myeloma. The patient subsequently responded well to daratumumab, cyclophosphamide, bortezomib, and dexamethasone chemotherapy and autologous stem cell transplant. This case illustrates the potential for IL-4Rα blockade to control symptoms such as pruritus in scleromyxedema, but underscores the need for plasma cell-directed therapy to address the underlying disease process.
Keywords
Introduction
Scleromyxedema (SMX) is a life-threatening disorder characterized by mucin deposition and fibrosis of the skin and internal organs.1,2 SMX is typically associated with IgG λ monoclonal gammopathy; however, IgG κ cases have also been reported.2,3 The pathogenic role of these immunoglobulins remains incompletely understood. 4 Both Type 1 (e.g. TNF-α, IFN-γ) and Type 2 (e.g. IL-4, IL-13) cytokines have been implicated in myofibroblast activation and matrix deposition in SMX and related fibrosing disorders.4,5 Recent immunophenotyping studies suggested an increased frequency of IL-4-producing T cells in SMX, raising the possibility that Type 2 cytokine inhibition may offer symptomatic benefit. 6 We report a case of treatment-resistant SMX in which dupilumab was trialed for symptom control (pruritus) but did not alter fibrosis or gammopathy progression. The patient ultimately responded to plasma cell-directed therapy and autologous stem cell transplant (ASCT).
Case
A male in his 50s first presented to a center overseas with diffuse skin thickening and waxy papules. Histopathology confirmed the diagnosis, and this was later corroborated by a second pathologist at Johns Hopkins. At the time of diagnosis, serum studies revealed an IgG λ monoclonal gammopathy (M-protein 22 g/L), while bone marrow biopsy and whole-body MRI showed no evidence of multiple myeloma.
Over the following years, the patient was treated with topical corticosteroids, phototherapy, lenalidomide (discontinued early due to recurrent infections and lack of efficacy), methotrexate, and cyclosporine, with limited or no success. From 2015 to 2020, intravenous immunoglobulin (IVIG; 2 g/kg every 3–4 weeks) induced initial disease control, but a taper in 2017 led to worsening symptoms. Despite the resumption of full-dose IVIG, progressive disease developed by 2022.
Upon referral to our center, joint pain, limited range of motion, restricted mouth opening, difficulty swallowing, and pruritus (7/10) were reported. Physical examination revealed erythematous, xerotic, waxy, shiny, thickened skin over >50% body surface area with a characteristic leonine face, doughnut, and Shar-Pei signs. Small monomorphic papules were seen on his neck and retroauricular skin (Figure 1). Dermatology Life Quality Index (DLQI) was 22. 7 Repeat testing showed an IgG λ M-protein of 23.6 g/L. Given ongoing pruritus and new mechanistic data implicating IL-4 in SMX, dupilumab was initiated as an adjunct to IVIG (per atopic dermatitis posology) for symptom relief. Four months later, the itch resolved, minimal/no improvement of skin thickening and fibrosis were noted, and DLQI decreased to 17. After 12 months, functional limitations worsened, DLQI rose to 25, and M-protein had increased to 35.3 g/L. Bone marrow biopsy revealed 20%–30% λ-restricted plasma cells, consistent with smoldering multiple myeloma, and whole-body MRI ruled out focal lytic lesions or soft tissue involvement. Dupilumab was discontinued due to a lack of disease-modifying efficacy. The patient then received four cycles of daratumumab, cyclophosphamide, bortezomib, and dexamethasone (Dara-CyBorD) from January to April 2024, followed by ASCT in July 2024. By August 2024, M-protein had decreased to 1.1 g/L, and ~50% improvement in skin infiltration was observed (Figure 2).

Patient’s presentation before initiating dupilumab treatment. Cutaneous fibrosis and infiltration manifesting as (a) leonine face. (b) Donut sign. (c) Waxy shiny thickened and fibrotic skin on the patient’s back with Shar-Pei sign. (d) Small monomorphic linear papules on the neck and retroauricular skin.
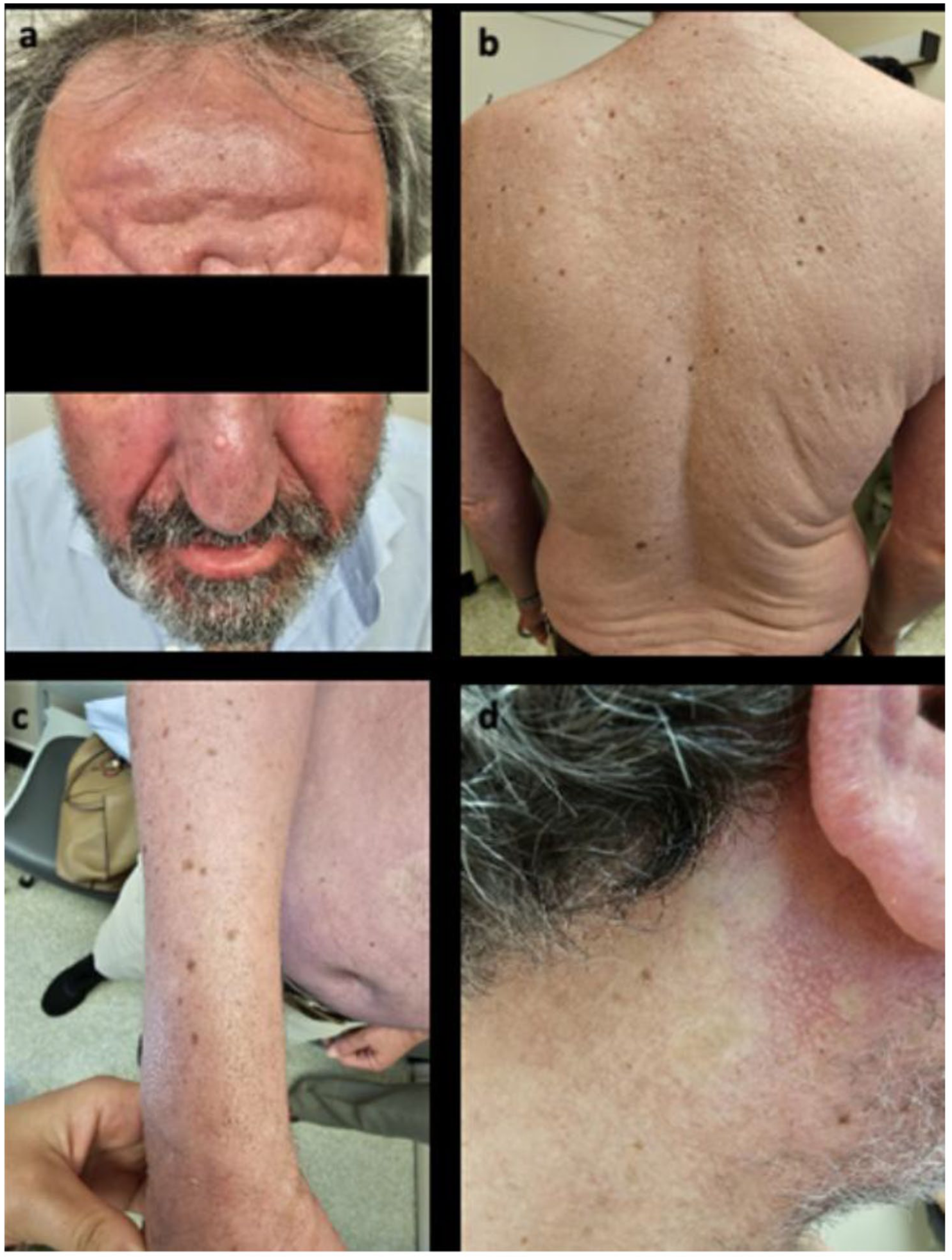
Patient presentation 12 months after dupilumab initiation. Persistence of (a) leonine face. (b) Shar-Pei sign. (c) Waxy shiny fibrotic skin on limbs. (d) Monomorphic linear papules on the neck and retroauricular skin.
Discussion
This case highlights the diagnostic and therapeutic complexity of advanced SMX. While IL-4Rα inhibition via dupilumab provided transient relief of pruritus, it had no measurable impact on fibrosis or progression of the underlying monoclonal gammopathy. The patient’s disease ultimately responded to plasma cell-directed therapy and ASCT, supporting the central role of the gammopathy in SMX pathogenesis.
Recent studies have implicated Type 2 cytokines, including IL-4 and IL-13, in fibroblast activation and matrix remodeling, suggesting a potential role for Type 2 pathway inhibitors in fibrosing skin diseases.4,5 In SMX, Kalli et al. reported increased IL-4-producing T cells in lesional skin. 6 However, while this may explain symptom burden such as pruritus, it does not appear sufficient to drive disease remission in the absence of hematologic control. Our patient’s DLQI and pruritus improved within 4 months, but fibrosis and M-protein levels continued to worsen, and 20%–30% λ-restricted plasma cells were eventually identified on bone marrow biopsy.
The therapeutic ladder in SMX typically begins with IVIG, often used as first-line therapy for cutaneous disease. 8 In patients with systemic or refractory involvement, corticosteroids, thalidomide, bortezomib-based regimens, and stem cell transplant are commonly employed.9,10 Recent literature supports the use of anti-CD38 antibodies such as daratumumab in monoclonal gammopathies, even in the absence of overt myeloma, given their favorable safety and efficacy profiles.2,3,11,12
This observation aligns with our findings. Dara-CyBorD led to a rapid biochemical response, and ASCT further consolidated skin and laboratory improvement. While dupilumab was trialed for symptomatic relief, we refrain from attributing any disease-modifying role, particularly in the context of rising M-protein and evolving plasma cell burden. Whether earlier use or combination with cytotoxic therapy would yield different outcomes remains unknown.
Due to the rarity of SMX, treatment is often guided by case series and pathophysiologic rationale rather than randomized trials. Our findings underscore the need for further translational studies to clarify the contribution of Type 2 cytokines to fibrosis and to define appropriate roles for biologics versus cytotoxic regimens.
Conclusion
In this case of refractory SMX, dupilumab provided relief of pruritus but had no impact on cutaneous fibrosis or progression of the underlying monoclonal gammopathy. Disease control was ultimately achieved with plasma cell-directed chemotherapy and ASCT. These findings underscore the importance of addressing the hematologic component in SMX and suggest that IL-4Rα inhibition may offer symptom relief in selected cases, but further clinical and translational studies are needed to define its role in disease modification.
Footnotes
Consent to participate
The patient has provided signed written consent to the submission of the case report for submission to this journal.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: E.N. has been a speaker or consultant, or has received investigator-initiated research funding from AbbVie, Arcutis, Bausch Health, Boehringer Ingelheim International, Bristol Myers Squibb, Galderma, Janssen, LEO Pharma, Medexus, Novartis Pharmaceuticals, Pfizer, Sanofi Genzyme, Sun Pharmaceuticals, Eli Lilly, and UCB. E.N. is the founder of the Montreal Derm FilEZ website, which is a non-profit educational resource. A.N. has been a speaker and consultant for Janssen, AstraZeneca, AbbVie, Karyopharm, and Kite Pharma. Other authors declare no conflicts.
