Abstract
Granulomatosis with polyangiitis is a systemic vasculitis of unknown etiology, characterized by necrotizing granulomas. It is an autoimmune disease affecting small- and medium-sized vessels of upper and lower respiratory tract, kidneys, and other organs. We described a case of a patient with otitis media with effusion as the first manifestations of granulomatosis with polyangiitis. A 54-year-old female presented as an urgent case with history of a severe otalgia, hearing loss, vertigo, and fever. The patient was treated with diagnosis of otitis media with effusion and acute rhinosinusitis, but without significant success. She developed an acute kidney dysfunction as a sign of glomerulonephritis with rapidly progressive renal failure. Diagnosis of granulomatosis with polyangiitis was confirmed after the histopathological analysis of kidney tissue, not by analysis of middle ear and paranasal sinus mucosa specimens. The patient was treated according to generally accepted protocol, and over time, there was an almost complete recovery.
Keywords
Introduction
Granulomatosis with polyangiitis (GPA) is an autoimmune disease, characterized by necrotizing granulomas and vasculitis affecting small- and medium-sized vessels of upper and lower respiratory tract, kidneys, and other organs. 1 Nasal cavity and paranasal sinuses are the most common sites of involvement (85%–100%). 1 Prevalence of ear involvement varies from 20% to 70% of cases.1–6 In some cases, otologic signs and symptoms may be the first manifestation of this disease, including recurrent serous and purulent acute otitis media, sensorineural hearing loss, as well as facial nerve palsy. 1 It has been reported that cytoplasmic anti-neutrophil cytoplasmic antibodies (c-ANCAs) are highly specific for active GPA.1–6 However, histopathological finding of a granulomatous vasculitis is very important for confirmation of diagnosis.1–8 We described a case of a patient with otitis media with effusion as the first manifestations of GPA.
Case presentation
A 54-year-old female presented with history of a sudden-onset severe otalgia, headache, hearing loss, vertigo, and high temperature (up to 39°C). About 2 months before admission in our hospital, she complained of mild-to-moderate otologic symptoms and was treated with oral antibiotics, but failed to respond to treatment. On admission, laboratory blood analysis showed increase of inflammatory parameters: white blood cell (WBC) count of 24,000/mm3, erythrocyte sedimentation rate (ESR) of 60 mm/h, and C-reactive protein (CRP) of 94 mg/L. Otomicroscopy revealed redness and thickening of the tympanic membranes. Audiology showed a mixed hearing loss on the right ear of 65 dB and a conductive hearing loss on the left ear of 30 dB and flat tympanograms. A computed tomography (CT) scan of the temporal bones showed the presence of fluid in the middle ear spaces and thickening of tympanic cavity and mastoid cellules mucosa, without bone destruction (Figure 1). The patient underwent a myringotomy with grommet insertion and a mastoidectomy and continued to receive intravenous antibiotics. Histopathological analysis of biopsy specimens taken from the mastoid mucosa showed the signs of a mild inflammation. She had a good response to treatment and checked out from the hospital with plan for follow-up.

Axial plane of the CT scan of the temporal bones showing the presence of fluid in the middle ear spaces (arrowheads) and thickening of tympanic cavity and mastoid cellules mucosa, without bone destruction.
However, a month after the treatment, she came to the ear, nose, and throat (ENT) examination again due to the worsening of her general condition and otological complains (otalgia, purulent otorrhea, hearing loss, and vertigo). Otomicroscopy showed that the grommets dropped out with heal of tympanic membrane perforations. Laboratory blood parameters (ESR, CRP, and WBC) were increased. A control CT of the temporal bones identified again the presence of fluid in the middle ear spaces. Revision middle ear surgery was performed. Histology of middle ear mucosa specimens showed a severe inflammatory process. Postoperative course was associated with a bad general status and a facial nerve palsy occurred. The patient could not close eyes properly, but had good taste sensation. Microbiological analysis showed a negative culture of the ear fluid specimens. The patient developed nasal obstruction, pain in the forehead, and vision problems in the form of double vision. CT scan of the paranasal sinuses showed thickening of right ethmoid sinus mucosa, fluid in sphenoid sinus, and the signs of right-sided orbital cellulitis (Figure 2). She underwent the right endoscopic ethmoidectomy and histopathology of excised mucosa showed the signs of severe inflammation. The patient was postoperatively treated with high dose of piperacillin/tazobactam and the signs of orbital cellulitis decreased during the few days. The patient was also tested serologically for antibodies against proteinase 3 c-ANCA (PR3-ANCA), and this titer was high positive (20 IU/mL). However, the patient’s general status was worsened with the development of general edema associated with a systemic disturbance. Blood analysis showed azotemia, proteinuria, and erythrocyturia, suggesting the presence of a rapidly progressive glomerulonephritis.
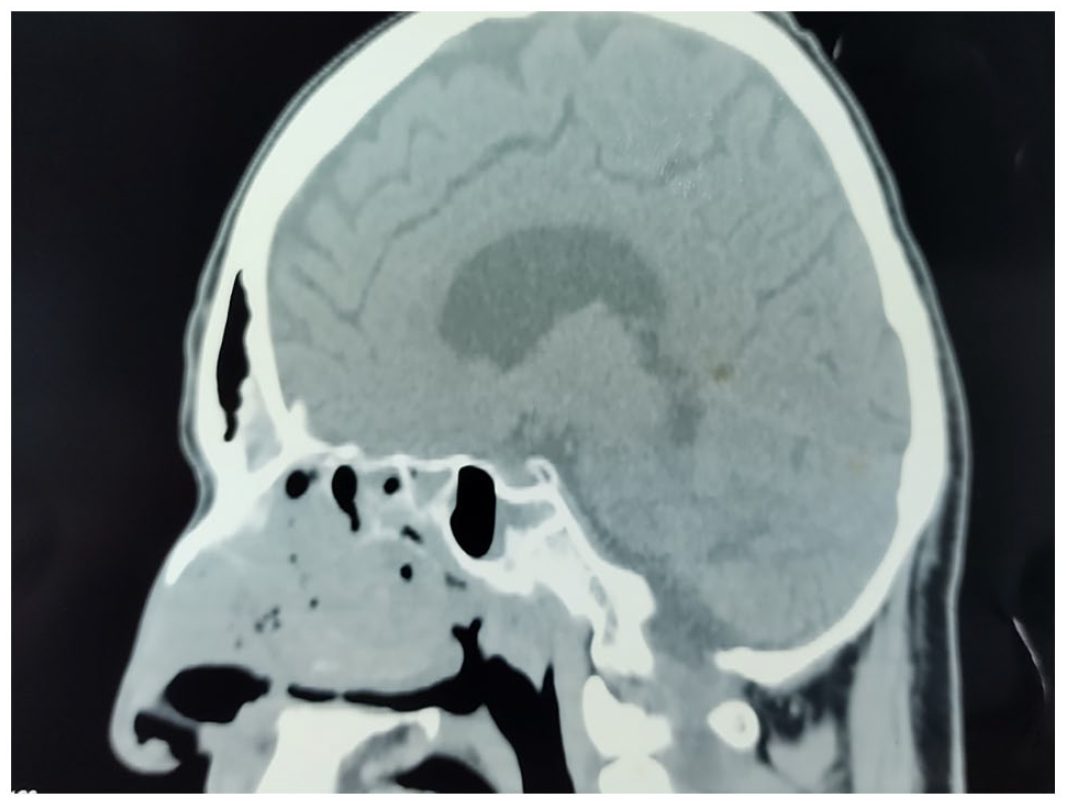
Sagittal plane of the CT scan of the paranasal sinuses showing thickening of right ethmoid sinus mucosa and fluid in sphenoid sinus.
On the admission to nephrology department, the patient had presentation of acute kidney injury with rapidly progressive renal failure (serum creatinine = 250–450 μmol/L) and daily urine output for more than 2000 mL. The patient had signs of systemic bacterial infection, and

Histopathological changes characterized for glomerulonephritis associated with GPA. The figure shows glomerulus (arrowhead) with necrosis affecting more than 50% of capillary tuft with voluminous cellular crescent. Surrounding tubules with flattened epithelium and signs of ischemia with acute tubular necrosis are seen (Hematoxylin & Eosin staining, magnification ×200).
Discussion
GPA was described as a separate syndrome in 1936 by Friedrich Wegener with name “Wegener’s granulomatosis.” 1 Syndrome can be presented in “limited forms” with involvement of the nose and paranasal sinuses, middle and inner ear, as well as subglottic region of the larynx.1–4 American College of Rheumatology, American Society of Nephrology and the European League Against Rheumatism suggested that the name should be changed to GPA.7,8
Etiology and pathogenesis of GPA are not clear. Small- and medium-sized vessels are attacked by circulating pathologic antibodies, named c-ANCA. Classically, GPA affects the triad of upper airway, lower airway, and kidneys, although any organ can be involved. It has an incidence of 1 in 25,000, with a mean age of 41 years. About 90% of the patients are Caucasian. 2 There are no clear diagnostic criteria for GPA, and diagnosis is based on the presence of typical organ involvement, positive c-ANCA serology, and histopathological confirmation of vasculitis, granulomatous inflammation, and necrosis. 9 The presence of circulating c-ANCA has a specificity of 80%–90% for GPA. However, c-ANCA can also be associated with other diseases, especially with rheumatoid arthritis.1,2,7,8 Therefore, some cases of GPA are negative for c-ANCA. A positive identification of c-ANCA is suggestive of GPA, but cannot replace a biopsy.1,2,9–11 Due to the autoimmune nature of disease, combination of corticosteroids and immunosuppressive agents is the mainstay of GPA therapy. Among immunosuppressive agents, cyclophosphamide is the conventional induction-treatment for systemic or diffuse GPA, while methotrexate is an alternative in forms of GPA that are not life-threatening. Rituximab can be used in combination with corticosteroids as a first-line treatment for severe administered in GPA with refractory otolaryngological manifestations. A recent case series reported a good response of refractory otitis media with effusion in GPA after rituximab treatment.3–6,12,13
It is not known why the nasal cavity is the most commonly involved place. Nasal obstruction, crusts and purulent and bloody discharge, facial pain, nasal septum perforation, and progressive nose deformity are the most frequent initial manifestations of GPA. 7 ENT involvement is a common feature in ANCA-associated vasculitis (AAV), particularly in GPA. Otologic manifestations in GPA are common, occurring in 20%–70% of the cases and representing the second most frequent symptoms of head and neck involvement.3–5 The mostly involved site is the middle ear (23%–70%), leading frequently to hearing loss, which could be the presenting symptom of GPA (as in the present case). 6
Less commonly, localized GPA may present only with otologic symptoms, without evidence of other AAV-related organ lesions.12,13 In these patients, not fulfilling the classification criteria for systemic vasculitis, the term “otitis media with AAV” (OMAAV) was proposed.12,13 The most common otologic manifestations include serous and purulent otitis media, due to the presence of the granulomatous tissue in the middle ear or nasal cavity and nasopharynx involvement with secondary Eustachian tube dysfunction. 2 Less common symptoms are sensorineural hearing loss (in 13%–43%), vertigo, and facial palsy. The mechanisms of inner ear damage are poorly understood and possible explanations are deposition of immune complexes in the vessels of stria vascularis, occlusion of labyrinthine artery, and immune complexes deposition in the capillaries of the endolymphatic sac. 2 In our patient, the diagnosis of GPA was confirmed after the histopathological analysis of kidney tissue, not by analysis of middle ear and paranasal sinus mucosa specimens. The technical difficulty of obtaining an appropriate biopsy specimen from the mucosa of the middle ear may be an important factor for impossibility to make histological diagnosis of GPA.1,5 So, it is recommended that biopsy specimen should be taken from the mucosa of the inferior turbinate or paranasal sinus.1,8,10,11 Although the histological evidence of GPA was insufficiently found in the analyses of middle ear or sinus mucosa specimens, surrogate markers for the diagnosis of GPA, such as otitis media or mastoiditis, could be found prior to the development of rapidly progressive glomerulonephritis (RPGN), according to the European Medicines Evaluation Agency (EMEA) algorithm.13,14 Moreover, OMAAV has been suggested as the categorized type of AAV.12–14 In our case, initial symptoms and findings did not suggest the presence of any systemic disease. The treatment was focused on otological and rhinological problems. The patient showed relatively good response to medical and surgical treatment during the first hospital days. However, in spite of clinical attention, the patient’s general condition was worsened with development of acute kidney dysfunction. The patient was treated according to generally accepted protocol and, over time, health problems have improved.
Conclusion
It is clear that otorhinolaryngologists have an important role in diagnosis and treatment of GPA. In this article, the authors described a case of c-ANCA GPA positive patient with middle ear involvement and subsequent renal inflammation. The otolaryngologic symptoms of GPA sometimes might be misdiagnosed in etiology. Thus, rapid recognition and early diagnosis of GPA prevent the risk of irreversible organ damage (e.g. kidney involvement). This article underlies the importance of early recognition and treatment of otolaryngologic manifestations of GPA and the importance of appropriate interdisciplinary management to reduce morbidity in these patients.
Footnotes
Author contributions
D.D., A.P., and M.R. contributed to the concept. D.D. contributed to the design. D.D., A.P., and M.R. contributed to the supervision. D.D., B.P., M.F., A.B., M.B., J.P., and M.R. contributed to the resources. D.D., B.P., M.F., A.B., M.B., J.P., and M.R. contributed to the materials. D.D., B.P., M.F., A.B., M.B., J.P., and M.R. contributed to the data collection and/or processing. D.D., A.P., and M.R. contributed to the analysis and/or interpretation. D.D., A.P., and M.R. contributed to the literature search. D.D., A.P., and M.R. contributed to the writing manuscript. D.D., A.P., B.P., M.F., A.B., M.B., J.P., and M.R. contributed to the critical review.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethics Committee Approval to report this case was received from the Ethics Committee of the Military Medical Academy, Belgrade, Serbia (approval no. 05/2019).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
