Abstract
Congenital peritoneal encapsulation is a rare entity characterized by an accessory peritoneal membrane that forms during embryonic development. Congenital peritoneal encapsulation is generally asymptomatic but can cause intermittent, colicky abdominal pain related to subacute small bowel obstruction. Diagnosis is made incidentally or upon surgical exploration for chronic abdominal complaints as preoperative imaging is typically nonspecific. We report a case of a 49-year-old male with epigastric abdominal pain, constipation, and superior mesenteric vein thrombosis on imaging. Upon exploratory laparotomy, the small bowel was covered by an accessory peritoneal sac consistent with congenital peritoneal encapsulation. The accessory sac was excised completely, and the patient recovered well. Although rarely causing significant gastrointestinal symptoms, congenital peritoneal encapsulation is an anomaly that requires surgical intervention.
Introduction
Congenital peritoneal encapsulation (CPE) is a rare condition characterized by an accessory peritoneal membrane encompassing a varying degree of the small intestine. 1 Frequently diagnosed at autopsy, CPE may be associated with other congenital abnormalities, such as incomplete situs inversus and intestinal malrotation.2,3 When present, symptoms are vague and recurrent but rarely can cause life-threatening bowel ischemia. 4 The membrane most likely forms during the 10th week of gestation and is secondary to an abnormality during mid-gut reduction from the umbilical stalk.1,3 The mean age of diagnosis is 40 years with a slight male predominance.1,5 Preoperative diagnosis is difficult and rare. 6 Definitive treatment is excising the membrane and addressing other abnormalities.1,7 Histologically, the accessory membrane is identical to the peritoneum.1,4 We present a case of CPE associated with a partial small bowel obstruction, superior mesenteric vein (SMV) thrombus, and a congenital defect in the mesentery associated with the duodenum positioned anterior to the superior mesenteric artery (SMA). Other case reports demonstrate congenital anomalies including malrotation or aberrant vascular structures.2,3 Our case is unusual due to the presence of an SMV thrombus most likely due to venous torsion and subsequent stenosis secondary to a partial volvulus. To our knowledge, no other case reports have demonstrated these anatomic anomalies related to CPE.
Case report
A 49-year-old male with a history of hypertension, gastroesophageal reflux disease, and no past surgical history presented with a 7-day history of vague epigastric abdominal pain and constipation on 30 January 2022. Two days prior, he was seen at an outside hospital with similar complaints, underwent abdominal computed tomography (CT) and was discharged on omeprazole with the diagnosis of gastroparesis. In our emergency department, he reported no fever, chills, melena, or prior surgeries. His physical examination was unremarkable with normal vitals, minimal abdominal distention, and mild epigastric pain. Laboratory data were unremarkable. CT of the abdomen and pelvis revealed the duodenum anterior to the SMA, swirling consistent with a partial mesenteric volvulus, an occlusive thrombus in the SMV extending to the portal confluence, and no evidence for a bowel obstruction (Figure 1). The diagnosis was not clear, heparin, ceftriaxone, and metronidazole were begun, and the patient was admitted to the surgery service and managed expectantly. His pain improved, his abdominal exam remained benign, but he was not able to tolerate liquids on day two. Repeat CT showed progression of the SMV thrombus and increasing mesenteric venous congestion. Subsequently, he was taken to the operating room for an exploratory laparotomy with the presumed diagnosis of a congenital anomaly associated with an internal hernia or volvulus. On opening the peritoneal cavity, the entire small bowel was found to be encased in an accessory thin transparent sac without associated adhesions (Figure 2). The sac was opened and found to be attached to the retroperitoneum, extending from the duodenal-jejunal junction to the terminal ileum. The ligament of Treitz was in the normal anatomic position, the duodenum was anterior to the superior mesenteric vessels, the 2 cm in diameter SMV was filled with clot, and the distal ileum passed posterior to the SMV and mesentery via a congenital defect prior to entering the cecum (Figure 3). The entire accessory peritoneal sac was excised and sent to pathology. As the ileum passed through a narrow mesenteric defect and possibly was responsible for the patient’s symptoms, the distal ileum was transected, transposed, and a stapled anastomosis was performed anterior to the SMV and mesentery. Due to the mesenteric edema and the need for anticoagulation, temporary abdominal closure was utilized and definitive closure was completed 2 days later. Pathology revealed mature fibroadipose tissue with focal mesothelial lining, consistent with peritoneum. His postoperative course was complicated by an ileus and heparin-induced thrombocytopenia that required fondaparinux. He was discharged on postoperative day 14. Outpatient follow-up at 3 months confirmed resolution of his symptoms and the ability to tolerate a regular diet. Retrospective review of the patient’s imaging with radiology confirmed the accessory peritoneal membrane surrounding the small bowel consistent with CPE (Figure 4).
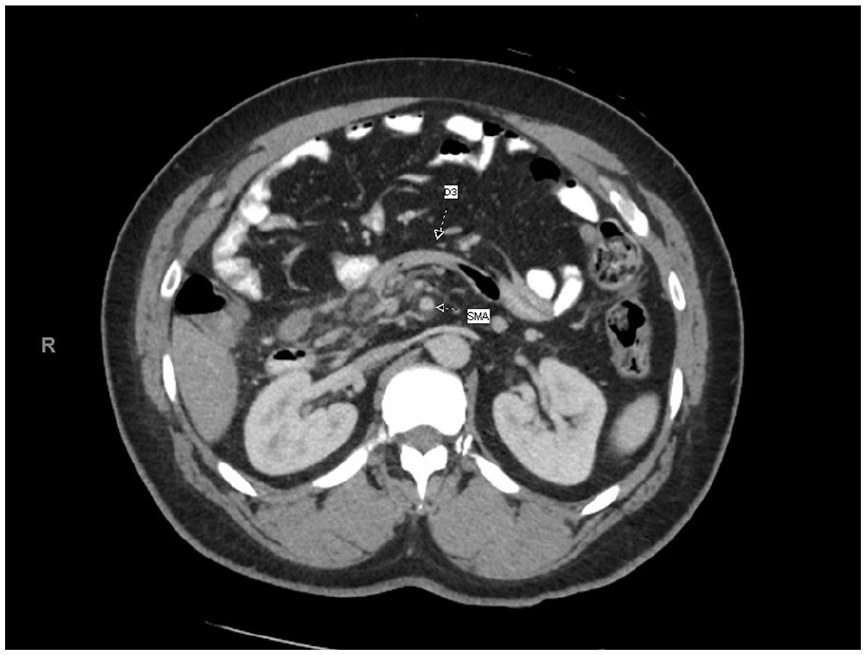
CT showing the duodenum (D3) anterior to the superior mesenteric artery.

Accessory peritoneal membrane (black arrows).

Surgeon’s hand connecting with DeBakey forceps through a defect posterior to the SMV and mesentery.
Accessory peritoneal membrane surrounding the small bowel.
Discussion
CPE is an infrequently encountered congenital abnormality characterized by an accessory peritoneal membrane encompassing the small bowel. 1 The membrane is typically semitransparent, vascularized, thin, and extends variable lengths from the duodenal-jejunal to the ileocolic junction. 3 Fewer than 50 cases have been described in literature since 1868.1,5 The mean age of presentation is 40 (32–46) years with a 5:3 male predominance. 1
A proposed etiology is the abnormal abdominal return of small bowel after physiologic herniation during the 8–10th week of gestation due to aberrant adhesions between the peritoneal lining and the caudal duodenum.1,2 The tethered yolk sac migrates with the intestine rather than remaining at the umbilical pedicle and becomes the accessory membrane.1,5 Other case reports of CPE describe congenital small bowel adhesions causing obstruction in addition to the accessory membrane.2,7,8 These congenital adhesions were not found in our patient. An alternative explanation is related to the timing of the reduction of the bowel during physiologic herniation. Should the distal jejunum and ileum abnormally reduce prior to the proximal jejenum, the proximal jejenum would be pushed inferiorly, to the right, and posterior to the dorsal mesentery, thus forming the abnormal sac. 1 This explanation would be associated with additional congenital abnormalities, consistent with the third portion of the duodenum being anterior to the SMA, as seen in this patient. Similar case reports describe aberrant vascular structures identified at the time of surgery. The anomalous vessels were felt to be contributing to obstruction or a risk for internal hernia and were successfully ligated.2,3 In our case, we did not identify any additional vascular structures but did encounter an enlarged SMV secondary to thrombus. This thrombus was related to the venous stenosis caused by the axial rotation of the SMV secondary to the partial small bowel volvulus. This correlates with the patient’s symptoms as serial CT scans demonstrated progression of the SMV thrombus. Removing the peritoneal encapsulation and restoring the small bowel to its normal anatomic location, along with anticoagulation, adequately addressed this problem.
CPE is frequently asymptomatic or discovered at autopsy. 6 Symptomatic patients present with hours to weeks of varying degrees of nausea, vomiting, constipation, and colicky abdominal pain secondary to acute or subacute bowel obstruction. 9 Vital signs are typically normal. The most uniformly described physical findings include asymmetric abdominal distention and differences in consistency in the abdominal wall, not noted with this patient.2,10 Rarely patients have peritonitis due to strangulated small bowel.
Laboratory data are not helpful in the diagnosis. Radiographic imaging rarely identifies the accessory peritoneal sac. 9 However, CT may demonstrate other congenital anomalies, a pinwheel pattern of small bowel loops known as the Helix Sign, swirling found with a partial volvulus, or a partial small bowel obstruction.2,11,12 The abnormal positioning of the duodenum anterior to the SMA in our patient made us consider a congenital anomaly as the etiology of his symptoms.
The differential diagnosis includes two acquired conditions, encapsulating peritoneal sclerosis (EPS), and abdominal cocoon. EPS may be due to chronic inflammation and is characterized by a thick, grayish white membrane, and is secondary to conditions such as peritoneal dialysis, ventriculoperitoneal shunts, and sarcoidosis. 1 EPS secondary to peritoneal dialysis has been successfully treated with corticosteroids and immunosuppressants. 7 In contrast to EPS, which has a 60% mortality, CPE is usually tolerated well. 7 Abdominal cocoon is also characterized by a dense, adherent membrane. It is a rare disease process in young women thought to be due to retrograde menstruation and gynecologic infections. 8 At the time of surgery, CPE can be differentiated by the thickness and opacity of the membrane, the ability to separate the accessory membrane from the underlying bowel, and the microscopic appearance on final pathology. 11 CPE is histologically identical to the peritoneum while EPS and abdominal cocoon are associated with inflammatory cells (Figure 5).3,7

H&E staining of accessory peritoneal membrane histology at 20× magnification showing mature fibroadipose tissue at the top with a thin mesothelial lining.
Following any necessary resuscitation, the treatment is surgical excision of the accessory peritoneal membrane in its entirety. 1 Care should be taken near the duodenal jejunal junction as the membrane is near the superior mesenteric vessels. Associated adhesions should be taken down and congenital anomalies addressed as appropriate.2,3 The laparoscopic approach may be attempted, but with undiagnosed CPE, the accessory membrane may be missed upon creation of pneumoperitoneum.10,11 In this patient, the membrane was freely mobile, required minimal dissection, and the SMV was quite prominent as it was 2 cm in diameter and filled with clot. The decision to divide the terminal ileum and transpose it superiorly to the SMV and mesentery was intuitive and not previously described. He is doing well at 3 months. His prognosis is quite good as no prior patients have required re-operation following membrane excision.9,10
Conclusion
CPE is an infrequently encountered congenital abnormality characterized by an accessory peritoneal membrane that encircles a variable portion of small bowel. Although the etiology is debated, anatomic abnormalities on imaging should heighten one’s suspicion as the diagnosis is rarely made preoperatively and may be missed during minimally invasive surgery. Treatment with complete peritonectomy of the accessory membrane is straightforward. Prognosis is very good if CPE is recognized and treated early.
Footnotes
Acknowledgements
The authors thank Douglas J. Hartman MD, Associate Professor of Pathology, for reviewing the case and providing histology imaging.
Author contributions
All authors contributed to this paper with conception and design, literature review and analysis, drafting and critical revision and editing, and approval of the final version.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: University of Pittsburgh Medical Center, Department of Surgery.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
