Abstract
An aberrant right subclavian artery (ARSA) is an extremely rare congenital anomaly that forms during aortic arch development. Most reports of thoracic endovascular aortic repair (TEVAR) described an ARSA in the right aortic arch, but it is rare in the left aortic arch. We present the case of a 66-year-old man who underwent total arch replacement because of acute type A aortic dissection. An outpatient follow-up examination revealed that the aortic diameter enlargement exceeded 60 mm because of false lumen entry from the ARSA. Therefore, surgical intervention was planned. TEVAR, ARSA embolization, and bilateral axillary bypass surgery were successfully performed for a chronic dissecting aortic aneurysm for which the ARSA was the inflow route. He was discharged 12 days after surgery. Four years later, no enlargement of the aneurysm diameter was observed. TEVAR is a minimally invasive and useful treatment option for chronic type B dissections with an ARSA associated with the left aortic arch; however, patients with an ARSA have fragile blood vessels and require careful follow-up.
Introduction
An aberrant right subclavian artery (ARSA) is a congenital anomaly that occurs during the development of the aortic arch; with an ARSA, the right subclavian artery branches directly from the aorta as the fourth branch of the aortic arch. 1 It is extremely rare, with a prevalence of only 0.4%–2.0% in the left aortic arch. 1 Thoracic endovascular aortic repair (TEVAR) for an ARSA associated with the left aortic arch results in better outcomes because the arch angle is not steep.2,3 We report a case of a chronic dissecting aortic aneurysm with an ARSA as the inflow route that was managed with TEVAR, ARSA embolization, and bilateral axillary bypass surgery.
Case
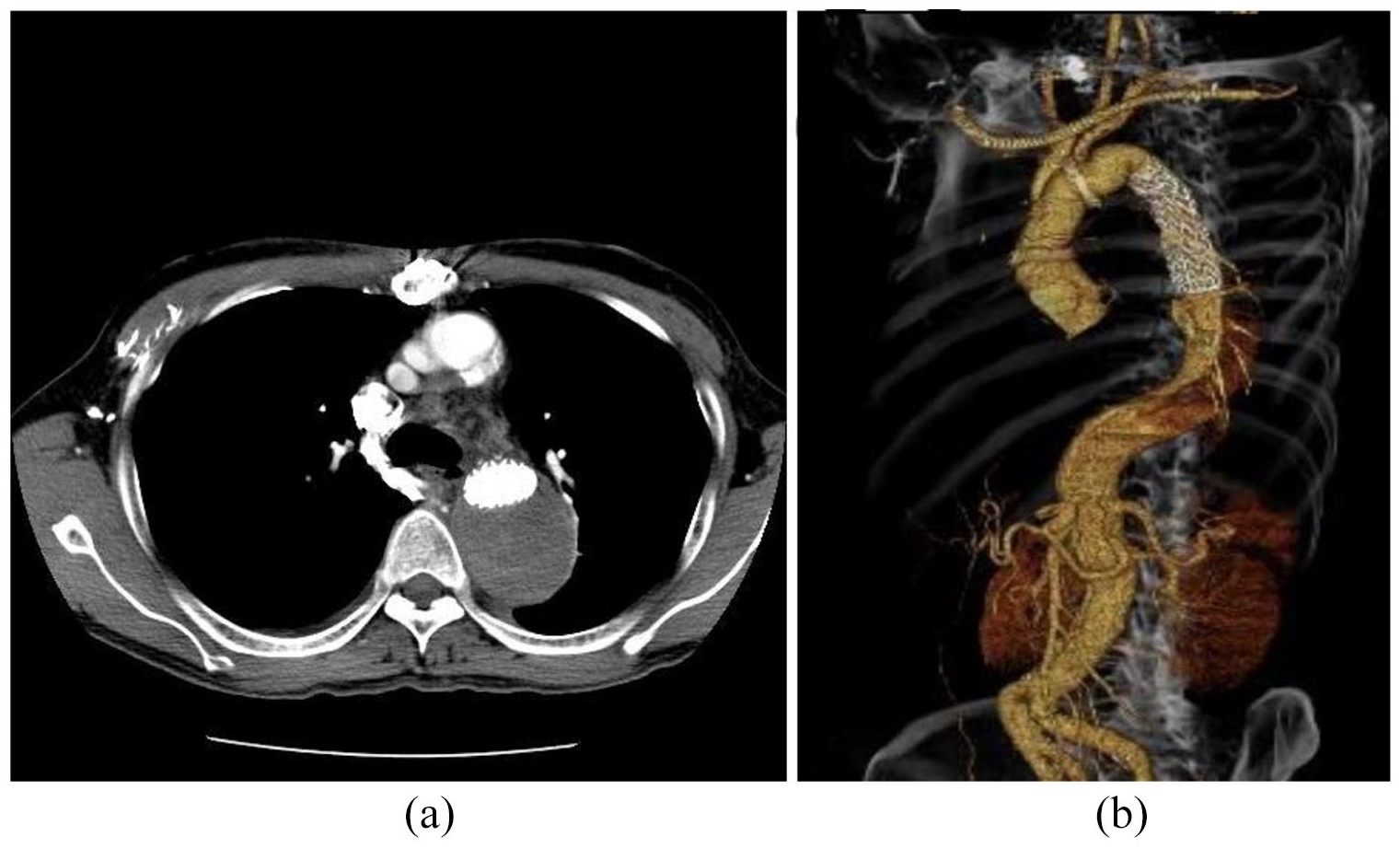
The patient was a 66-year-old man who presented to our emergency hospital for chest pain. Examination revealed an acute type A aortic dissection originating from the aortic arch. An ARSA was identified that started from the descending aorta, circulated on the dorsal side of the esophagus, and connected to the right axillary artery. The origin of the ARSA was 17 mm in diameter and emanated from the true cavity (Figure 1(a)). The aortic arch was branched in the following order: the right common carotid artery, left common carotid artery, left subclavian artery, and right subclavian artery. The inferior mesenteric artery originated from the false lumen, whereas the other branches originated from the true lumen. Emergency surgery was performed and included total arch replacement with a 22 mm artificial blood vessel and a 9 cm fresh elephant trunk graft. The postoperative course was uneventful, and the patient was discharged 25 days postoperatively.

(
Follow-up computed tomography (CT) was performed 4 years later, revealing that the maximum minor axis of the descending aorta had expanded to 60 mm because of inflow from the ARSA to the false lumen (Figure 1(b)). The patient had no dysphagia or notable blood test or cardiac echocardiography findings; however, carotid echocardiography revealed mobile plaques in both carotid arteries. At admission, his blood pressure (in the upper left arm) and pulse were 138/80 mmHg and 82 beats/min, respectively. No differences were noted after palpation of the radial arteries between both sides. Because the ARSA was thought to cause the false lumen enlargement, we embolized the ARSA. TEVAR and bilateral axillary bypass surgery were performed. A spinal drain was placed on L4/5 the day before surgery to prevent a spinal cord injury. Surgery was performed under general anesthesia. The bilateral axillary arteries and right common femoral artery were secured. Contrast imaging was performed from the ARSA to confirm blood flow into the false lumen (Figure 2(a)).

(
An AMPLATZR Vascular Plug II (16 mm; AGA Medical Corporation, Plymouth, MN, USA) and an Interlock-35 Coil (10 mm × 40 cm; Boston Scientific Co., Marlborough, MA, USA) were placed in the lower part of the ARSA (Figure 2(b)). From the right femoral artery, a GORE TAG (26 mm × 10 cm; W.L. Gore & Associates, Flagstaff, AZ, USA) was placed in the fresh elephant trunk graft. No endoleak was observed on contrast imaging (Figure 2(c)). Only the joint with the fresh elephant trunk graft was adjusted with a balloon. The bilateral axillary arteries were bypassed with a 6 mm artificial blood vessel before surgery was completed. The operative time was 3 h and 42 min. No postoperative anticoagulant therapy was administered. Because no paralysis was observed, the spinal drain was removed after the patient returned to the intensive care unit. Contrast-enhanced CT performed on day 7 postoperatively revealed that the false lumen of the descending aorta was caused by a thrombotic occlusion, and that the blood flow in the right upper limb was good. The maximum minor axis of the aortic dissection was 60 mm and did not change (Figure 3). The postoperative course was good, and the patient was discharged 12 days postoperatively. At 4 years postoperatively, no observable aneurysm diameter enlargement had been reported.
(
Discussion
Approximately 60% of all ARSAs form aneurysms at the origin; these formations are called Kommerell diverticula (KDs). 4 Furthermore, 80%, 15%, and 5% of ARSAs pass on the dorsal side of the esophagus, between the trachea and the esophagus, and on the dorsal side of the trachea, respectively. 5 Although most cases are asymptomatic, a surgical intervention may be necessary if the esophagus is compressed and causes dysphagia lusoria (DL), or if the KD expands by 30 mm or more.6,7 Although there is no consensus regarding the optimal approach for DL, Dieffenbach et al. reported that the supraclavicular approach alone led to a permanent improvement in dysphagia of patients without KD (but not in that of patients with KD). 8
In this case, the onset of the ARSA occurred with acute type A aortic dissection. The ARSA originated in the true lumen, and no aneurysm was observed. Therefore, no surgical intervention for the ARSA was performed during the initial surgery. Kawashima et al. reported that an ARSA was reconstructed using the median sternotomy approach, which is usually implemented for patients with a thoracic aortic aneurysm and dysphagia. When using this method, it is necessary to exfoliate the ARSA at the right edge of the trachea; however, caution is required because the right vagus nerve runs in front of the ARSA. 9
Furthermore, Cinà reviewed 32 cases of KD associated with a right aortic arch, and most involved TEVAR for ARSAs in the right aortic arch. 6 The right aortic arch has a shorter radius and a more acute curvature than the left aortic arch, which has a gentle curve; therefore, proximal endoleak caused by severe angulation may require additional intravascular treatment.2,3 TEVAR was performed for four cases of KD associated with the left aortic arch, resulting in a mortality rate of 0% during follow-up. 10 ARSA cases associated with the left aortic arch have better outcomes because the arch angle is not steep.
In our case, chronic type B aortic dissection was noted and enlargement of the aortic diameter (secondary to blood flow from the ARSA) exceeded 60 mm; therefore, TEVAR was selected. Because the proximal side is placed in an artificial blood vessel, a sufficient landing can be secured; however, the distal side is placed in the dissection cavity, making repeat dissection at the peripheral end of the stent graft edge difficult. Therefore, the graft diameter on the peripheral side was 0% of the true lumen trace. Furthermore, embolization was performed at the descending part of the ARSA to avoid compression of the esophagus. A right carotid-to-right subclavian artery bypass, which is the standard debranching technique that involves a short bypass and allows for future sternotomy, is usually performed. Because our patient had plaques in the carotid artery, a left axillary artery-to-right axillary artery bypass was performed to avoid cerebral infarction. Postoperatively, no anticoagulant therapy was administered to promote thrombosis of the aortic dissection cavity.
TEVAR is a minimally invasive and useful treatment method for chronic type B dissection with an ARSA. Blood vessels are fragile, and a follow-up CT examination is necessary. Surgeons must focus on the enlargement of the diameter of the false cavity and repeat dissection because of the stent edge.
Conclusion
TEVAR and embolization of the ARSA allowed successful treatment of chronic type B aortic dissection for which the ARSA was the inflow route. TEVAR is a minimally invasive and useful treatment option for aortic dissection; however, it is associated with aneurysm enlargement and repeat dissection. Therefore, careful follow-up is required.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases series.
Informed consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
