Abstract
Chronic-contained rupture of an aortic aneurysm is a rare subset of ruptured aneurysms. The presentation is unusual, and the diagnosis is frequently delayed. Here, we describe a case of contained rupture of abdominal aortic aneurysm that presented with signs and symptoms of femoral neuropathy. Clinical and radiological findings were initially misinterpreted. The correct diagnosis was formulated belatedly, causing a progressively increased risk of fatal events. Surgical aortic repair was performed and the postoperative course was uneventful. In conclusion, in the presence of a retroperitoneal mass, a diagnosis of chronic-contained rupture of an abdominal aortic aneurysm should be considered.
Introduction
Chronic-contained rupture of an aortic aneurysm is a rare subset of ruptured aneurysms, and it is found in 1.5%–3% of abdominal aortic aneurysms (AAAs). 1 Patients with chronic-contained rupture present with atypical symptoms for aneurysmal rupture and they are hemodynamically stable. The clinical presentation occasionally results in misdiagnosis, including carcinoma or infection. 1
The purpose of this case report is to emphasize the importance of a careful evaluation of patients with chronic back pain, combining clinical and radiological findings.
Case report
A 64-year-old Caucasian man presented to our hospital with a 5-year history of low back pain and functional impairment of the left leg. The patient had undergone multiple computed tomography (CT) scan and other diagnostic procedures in another hospital, as discussed below. The first angio-CT was performed in 2015 in another hospital and the report described a soft-tissue mass measuring 8.0 × 4.0 cm with peripheral enhancement extended in the paravertebral space from the fourth lumbar vertebral body to the fifth vertebral body. The mass caused lysis and destruction of the fourth and fifth lumbar vertebrae and the intervening disk and was not visualized separately from the aorta, the inferior vena cava, and the left psoas muscle. The CT finding was interpreted as retroperitoneal tumor. Subsequently, the patient underwent CT-guided aspiration of lesion, and hemorrhagic cells mixed with cellular debris were obtained. No neoplastic cells were evident on cytological examination of this material. The patient was consequently discharged from the hospital without a clear diagnosis.
One year later, the patient underwent emergency CT scan in the previous hospital for acute foot pain. The new radiological report described the presence of an AAA that extended from 7 cm below the renal arteries to the aortic bifurcation (approximately 5.8 × 5.0 cm in diameter). The CT scan also confirmed the presence of the already described retroperitoneal mass with increased diameter (12.0 × 8.0 cm vs 8.0 × 4.0 cm). The posterior wall of the aorta was not clearly separated from the mass. Reactive lymph nodes were visualized in the surrounding area. Magnetic resonance (MR) imaging of the abdomen was then performed to further characterize the lesion, and it confirmed the presence of a solid mass of 10 × 5.7 cm with peripheral and inner septal contrast enhancement, suggesting a lymphoproliferative lesion as the most likely diagnosis. Consequently, the patient underwent oncologic evaluation with no clear response.
From 2015 to 2021, the patient continued to suffer from low back pain radiating to the left leg.
In July 2021, he seeked medical attention in our Hospital. On our examination, a large pulsatile swelling was seen in the back of the patient on the left side. The patient presented clear symptoms of femoral neuropathy with functional impairment of the left leg and inability to extend the knee. He referred also an intense lumbar pain. The patient was hemodynamically stable. After a new CT scan, the images showed the presence of an AAA with a maximum diameter of 17 cm but no clear delimitation because of its extension along the paravertebral spaces where it formed a mass with features consistent with a blood clot (the same mass that previously was considered a retroperitoneal tumor) (Figure 1). An erosion of the anterior surface of the L3 and L4 vertebral bodies was also noted (Figure 2).
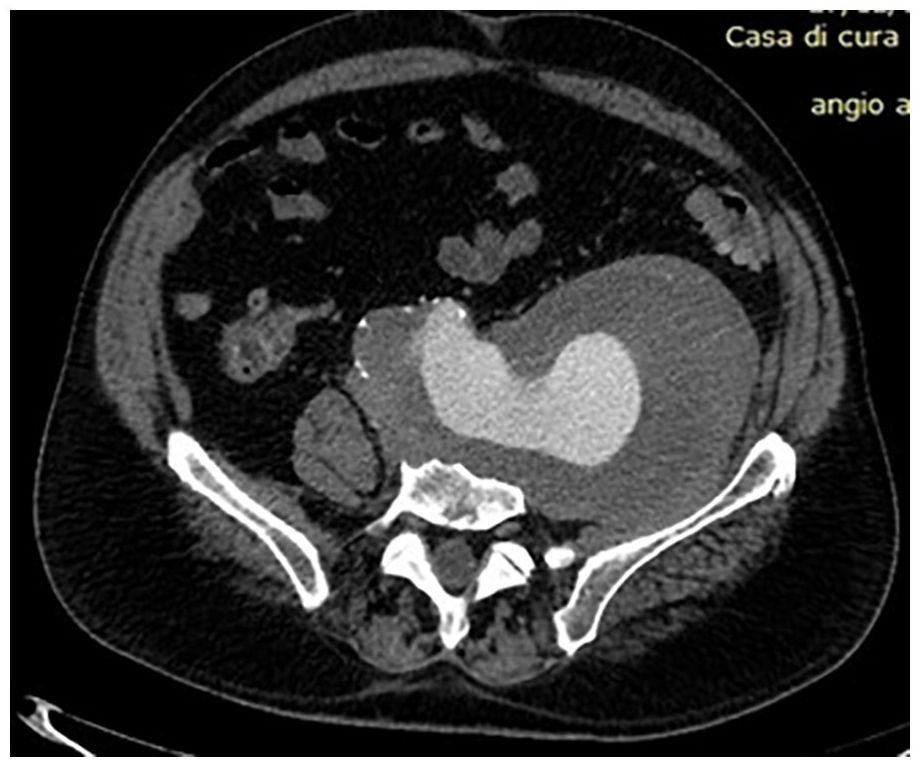
Contrast-enhanced axial multidetector CT angiography image reveals chronic-contained aneurysm rupture of the aorta within left ilio-psoas muscle (white arrow).
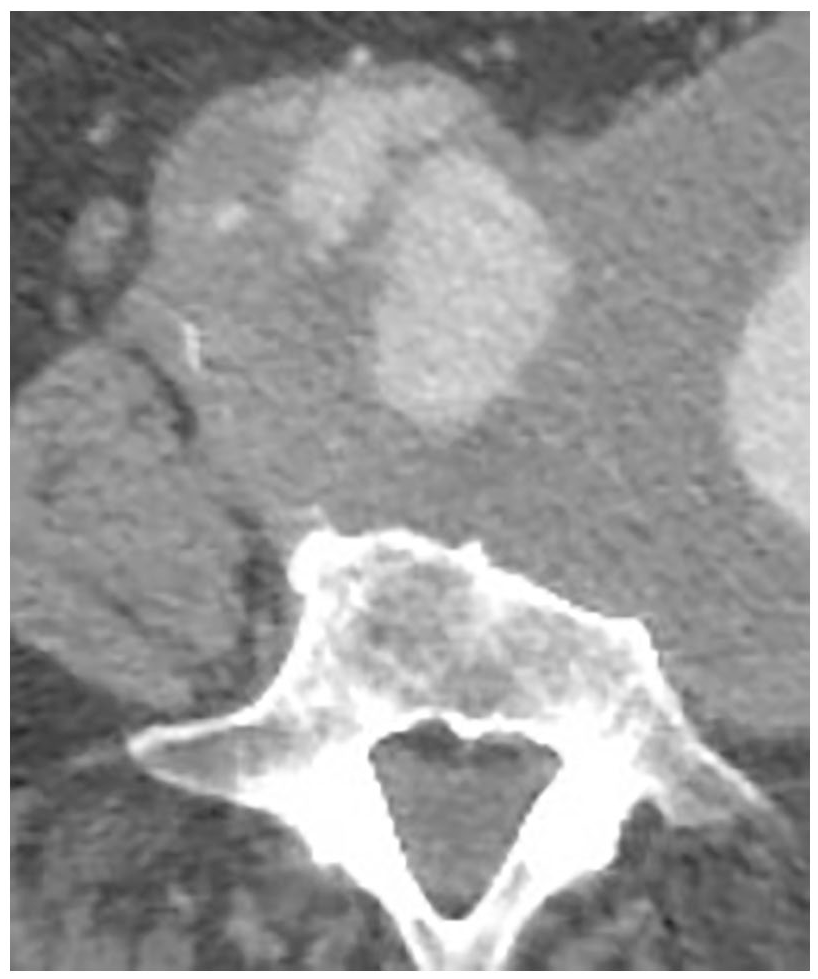
Computed tomography showing chronic-contained rupture of an abdominal aortic aneurysm with erosion into the L3 vertebral body (white arrows).
A diagnosis of chronic-contained rupture of AAA was formulated by our vascular surgery team.
The patient underwent surgical exclusion of the aneurysm by median laparotomy. The aortic neck of the aneurysm was isolated and clamped at the infrarenal level, while clamping of the common iliac arteries was performed by endoclamping using a Foley bladder catheter. After aortic cross-clamping and opening of the aneurysm, large clots of blood were evacuated from the paravertebral region; these clots corresponded to the mass seen in CT and MR imaging. We found that the L3–L4 vertebrae were almost completely eroded (Figure 3). An aorto-bisiliac bypass with a knitted Dacron graft (16 × 9 mm) was performed, without peri-procedural complications. The patient had no back pain at discharge. No pathologic organisms grew on the cultures of the clot evacuated during surgery. After a rehabilitation training program, the functional impairment of the left leg improved.
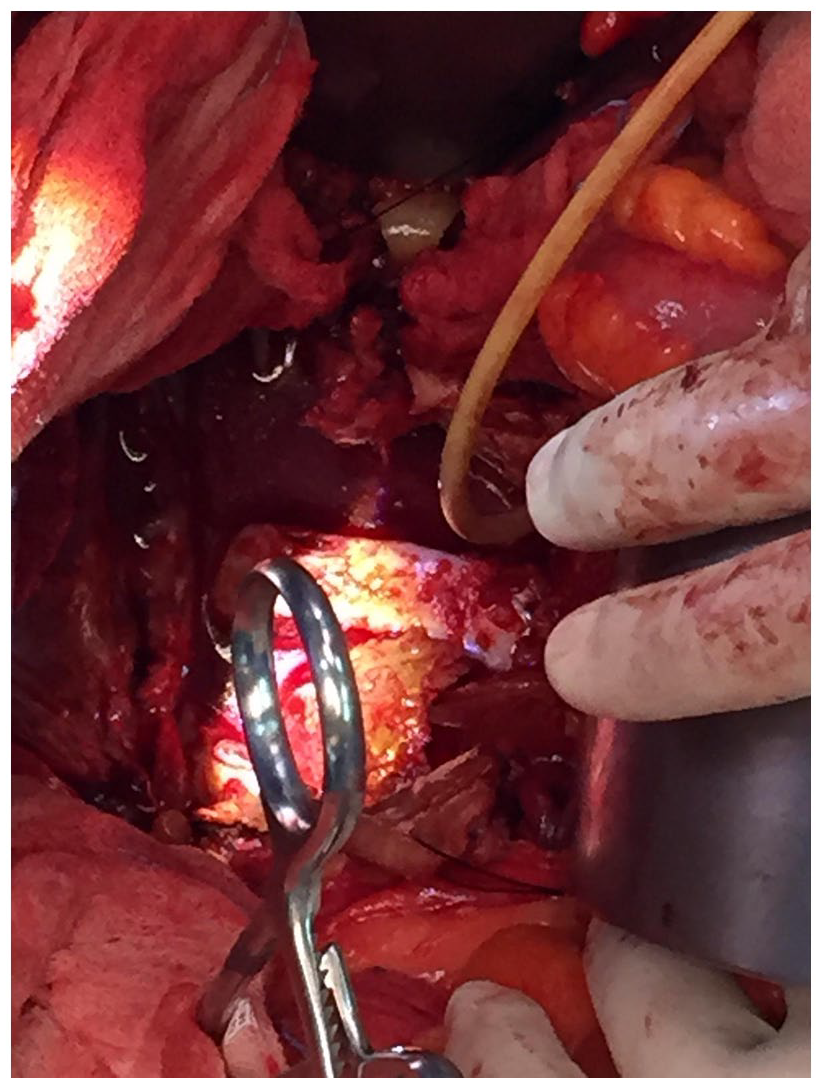
Intraoperative picture of the contained rupture.
Discussion
Chronic-contained rupture of aortic aneurysm was first described by Szilagyi et al. 2 and Galessiere et al. 3 Since the initial description, less than 100 similar cases have been documented worldwide. 4 Its presentation is distinctly different from that of acute rupture of aortic aneurysm.
The typical triad of acute abdominal or lumbar pain, hypotension, and shock is not present. 5 Patients with chronically contained rupture of aneurysms are hemodynamically stable and have a long history of back or loin pain with symptoms attributable to compressive or erosive effect. 6 In fact, as in this case, the rupture may occur within a contained space such as the retroperitoneum or the psoas muscle space, leading to the patient being stable at presentation and often for longer. 5 Booth and Galland 7 reported that the clinical features of contained AAA rupture are vague with back pain in 64% of cases, abdominal pain in 20%, groin pain in 14%, femoral neuropathy in 8%, sepsis in 10%, and asymptomatic in 8%. 4 The clinical presentation may masquerade as metastatic carcinoma, infectious spondylitis, retroperitoneal infection, psoas abscess, and, rarely, obstructive jaundice. 8 The mechanism of chronically contained AAA rupture is not clear. Moreover, the site of sealed AAA rupture has been reported as posterior in 62.5% of cases, suggesting that vertebrae can stop the rupture effectively because of their inherent strength. 9 Transmitted arterial pulsation and vertebral compression of 80–90 times/min may produce extensive bone destruction with time, involving nerve roots or fibers that pass around muscles.10–12
It is also possible that the large volume and significant extent of the bleeding may irritate the left lumbosacral trunk as a whole. According to Jones et al., 13 chronic-contained ruptures should meet the following criteria: well-known AAA, previous pain symptoms that may have resolved, stable hemodynamic status with a normal hematocrit, CT scans showing retroperitoneal hemorrhage, and pathologic confirmation of organized hematoma. 14
Abdominal X-ray and ultrasound are less sensitive than CT scan when used to diagnose contained aortic rupture, with a reported diagnostic accuracy of 50%. CT is more reliable because it establishes the extent of bone destruction, helping in early detection of signs of rupture. 8 Other CT signs include anterior displacement of the adjoining viscera, extravasation of contrast material, fluid collection, and hematoma within the posterior pararenal, peritoneal, or pleural space, free intraperitoneal fluid, and perirenal “cobwebs.”15,16
In addition to these features, a chronic-contained leak may lead to laminated mural calcification or periaortic mass of mixed or soft-tissue attenuation. A positive “aortic drape” sign is highly suggestive of a contained leak. It consists of a posterior aortic wall that cannot be identified distinctly from adjacent structures, with the posterior aorta closely applied to the spine, beyond the contour of the vertebral column.1,17
In the presence of a retroperitoneal mass not clearly divided from the vascular axis, with radiological characteristics of uncertain interpretation, a diagnosis of AAA should be suspected. Indeed, conditions with a high mortality rate, such as a contained ruptured aortic aneurysm, should be diagnosed at an early stage. This would significantly improve patients’ prognosis, since there is a concrete risk of sudden rupture of the aneurysm and a consequent massive bleeding.
Conclusion
Contained rupture of an aortic aneurysm is at risk of frank rupture and should be identified early at presentation.
Footnotes
Acknowledgements
The authors thank the patient for giving them consent to publish her de-identified clinical information and photos. In addition, the authors thank Prof. Savino Cilla for helping in article preparation.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
All procedures performed in studies involving human participants or on human tissue were in accordance with the ethical standards of the institutional and/or national research committee and with the 1975 Helsinki declaration and its later amendments or comparable ethical standards. Our institution does not require ethical approval for the present case report.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
