Abstract
Endoleaks are a common complication of endovascular aortic aneurysm repair and often lead to aneurysm rupture. We report a complex case of a Type IIIA endoleak in a patient with suspected cholangitis. Immediate surgical measures to manage both the cholangitis and endoleak were performed while minimizing the risk of graft infection.
Introduction
Endovascular aortic aneurysm repair (EVAR) is a frequently used surgical approach for abdominal aortic aneurysm (AAA) repair that offers several benefits when compared to an open approach; such as lower morbidity and mortality rates, less blood loss and postoperative pain, reduced risk of ileus and/or gastrointestinal injury, and earlier discharge from the hospital. 1 Endoleaks are a complication of EVAR and can lead to aneurysmal rupture. An endoleak is continued blood flow in an aneurysm sac outside of a stent graft.1,2 Endoleaks are classified as direct (Type I and III) or indirect (Type II, IV, and V). 2 Classification of endoleaks is necessary to determine both treatment modality and prognosis. Type III endoleaks are due to dysfunction at the junction of stent graft segments and are at an exceptionally high risk of rupture due to persistent systemic pressure.2,3
Acute cholangitis is a bacterial infection of the bile ducts commonly due to obstruction in the biliary tree. 4 Emergent treatment is required because it is associated with mortality rates as high as 10%. 4 The presence of a Type III endoleak and cholangitis in a patient can lead to graft infection and life altering complications such as sepsis and aneurysm rupture. For that reason, Type III endoleaks are serious adverse events that require prompt intervention to prevent further increases in aneurysm sac size and eventual rupture or dissection of the aneurysmal sac.
Case report
A 66-year-old female with a past medical history of hypertension, hyperlipidemia, cholecystectomy in 2019, and AAA status post-EVAR in 2016 at a separate facility presented to the emergency department with complaints of worsening epigastric abdominal pain radiating to her back, fever, and jaundice for 5 days. During initial workup, her labs revealed a normal white blood cell count with elevated neutrophils at 82.6% and elevated liver enzymes, including a total bilirubin of 9.8 mg/dL. A right upper quadrant ultrasound revealed biliary ductal dilation and a common bile duct (CBD) measuring 20 mm. A CT scan of the abdomen and pelvis with intravenous contrast was completed confirming the sonographic findings as displayed in Figure 1(a). A large Type III endoleak of her aorto-iliac endograft secondary to a complete disconnect of the left iliac limb was also discovered, with a maximum aortic sac diameter of 6.1 × 6.0 cm as shown in Figure 1(b).

CT scan (a) showing significant biliary ductal dilation. (b) Demonstrating abdominal aortic aneurysm with separated left iliac stent consistent with a Type III endoleak.
Upon initial examination, the patient was febrile to 102.2°F, tachycardic with epigastric tenderness, and jaundiced. It was unclear when the patient had developed the Type III endoleak due to the lack of follow-up with her vascular surgeon for over 5 years, and there were concerns about a rapid expansion and impending rupture. After discussion with the gastroenterology team, the decision was made to take the patient urgently for an endoscopic retrograde cholangiopancreatography (ERCP) and aortography to treat both the cholangitis and endoleak.
Preoperatively, blood cultures were sent, and a 3-day course of broad-spectrum antibiotics was initiated. Blood cultures showed no growth. The ERCP revealed a distal CBD stricture measuring 3 mm in length located 2 cm above the ampullary orifice with cystic dilation suggesting a Type III choledochal cyst demonstrated in Figure 2. A subsequent sphincterotomy and duct sweep with a stent placed in the CBD were performed. Next, the vascular surgical team performed an aortography via access from the right femoral artery which demonstrated extravasation of contrast into the aneurysm sac, as can be seen in Figure 3(a). Once the catheter was confirmed within the endograft main body, a 16 mm × 16 mm × 124 mm limb was placed via left-side access from the start of the flow divider down to the iliac limb to allow for adequate overlap with the existing iliac limb. A complete aortography was performed that revealed satisfactory placement with visualization of the renal arteries, complete exclusion of the aortic sac, and no endoleaks, as shown in Figure 3(b). The patient was continued on antibiotics while her bilirubin normalized, and she was discharged home on postoperative day 3 with a 10-day course of amoxicillin-clavulanate and hepatobiliary surgery outpatient follow-up for the suspected choledochal cyst.
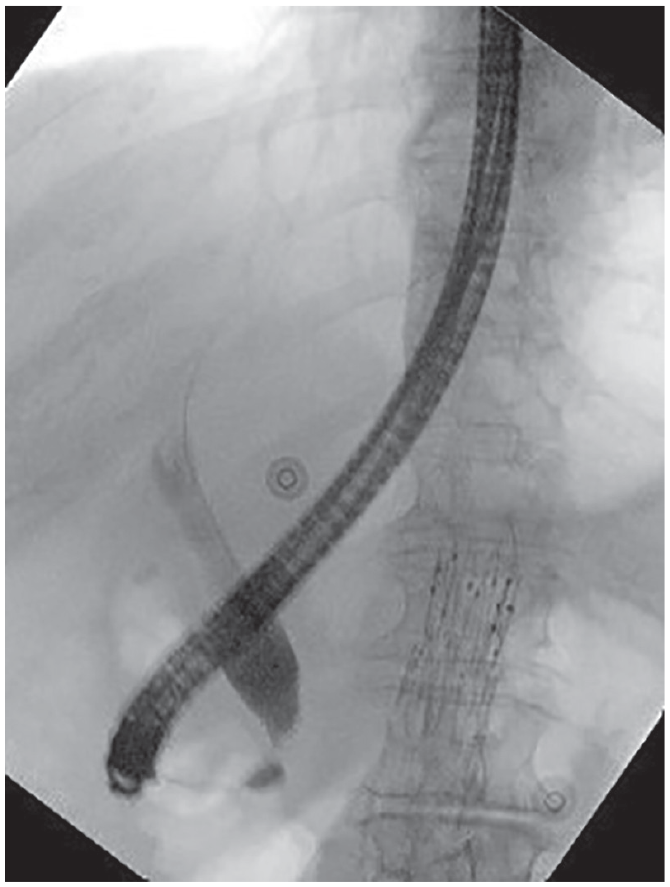
ERCP, imaging of common bile duct; consistent with a distal stricture.
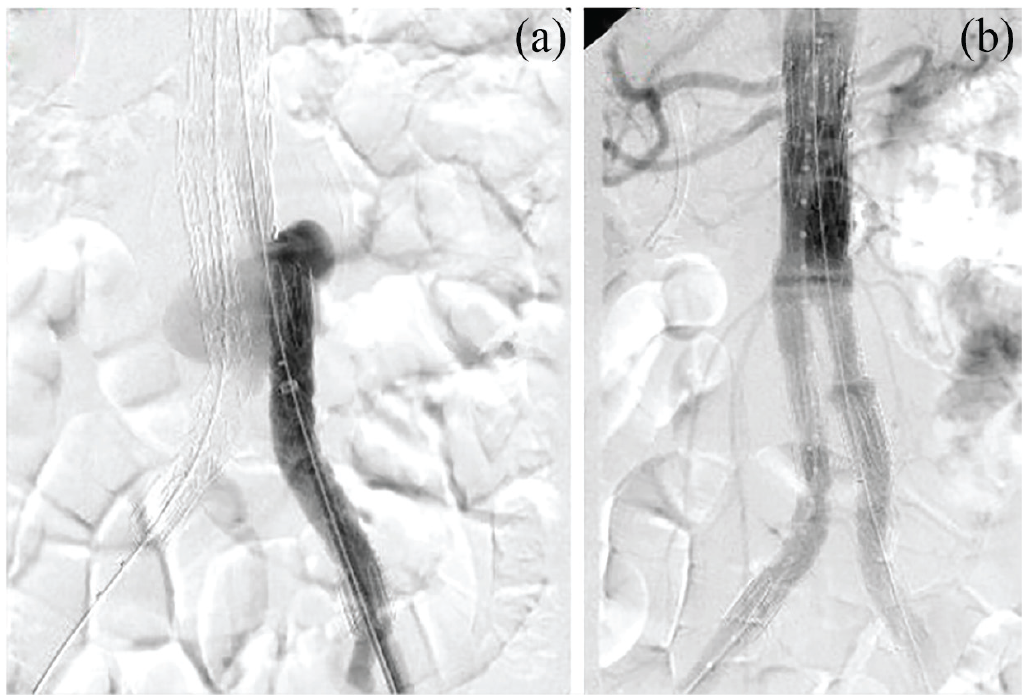
(a) Aortography demonstrating the Type III endoleak by showing contrast going into the aneurysm sac. (b) Completion aortogram showing preservation of renal arteries and no endoleaks.
The patient underwent outpatient endoscopic ultrasound and fine needle aspiration and was diagnosed with biliary ductal adenocarcinoma. A successful pancreaticoduodenectomy was performed. At the patient’s follow-up appointments at 1 and 12 months after her endoleak repair, there was a reduction in the size of the AAA from 6.1 cm to 5.2 cm.
Discussion
An AAA is a pathologic condition with progressive abdominal aortic dilatation of 3.0 cm or more that predisposes the abdominal aorta to increased risk of rupture. 5 Most AAA are asymptomatic and are identified incidentally on imaging with the size of the aneurysm being the most critical factor related to the risk of rupture.5,6 An aneurysm with a diameter of 5.0–6.0 cm has a 5% yearly chance of rupture. 7 Rapidly expanding aneurysms at risk for rupture can present with symptoms such as abdominal pain that radiates to the back, flank, and groin. 5 This nonspecific presentation makes the diagnosis even more complex in the setting of suspected cholangitis.
Patients lost to follow-up have a significantly higher incidence of complications, such as endoleaks, after EVAR. 8 A multicenter study including 965 patients showed that Type III endoleaks occur in 1.2% of aortic aneurysm repair cases. 9 Type III endoleaks are divided into three subtypes: Type IIIA leaks occur from separation of graft limb components which causes aortic sac pressurization, Type IIIB leaks originate from a fabric tear or fracture, and Type IIIC leaks are due to a defective side branch component. 3 Type III endoleaks can be treated by placement of a covered stent, insertion of an endograft limb extension, aorto-uni-iliac endograft, or proximal aortic cuff.2,9
Stent graft infections are a rare complication after an EVAR, with studies showing an overall incidence to be 0.2%–0.7%. 10 They can occur via perioperative contamination, mechanical erosion, or hematogenous seeding from an existing infection. 11 The time from EVAR to presentation with a graft infection occurs either in the early postoperative period or after 12 months with Staphylococcus aureus being the most common organism identified under microscope. 10 The increased risk of perioperative graft infection due to acute cholangitis made management of this patient’s Type III endoleak complex as no standardized practices exist. Additionally, the presence of an endoleak increases the likelihood of sepsis as an incompletely excluded but perfused hematoma may serve as a nidus for bacteria. 12
The findings from the CT scan suggest that the patient’s presentation was the result of a periampullary mass. Biliary tract dilatation is a key diagnostic element in acute cholangitis. 13 Obstruction in the biliary tract leads to infection, translocation of the bacteria into the bloodstream, and eventually septicemia. 14 Biliary decompression through ERCP with antibiotic coverage—the preferred treatment for cholangitis—was performed on our patient to reduce the risk of graft infection and septicemia.
There is literature available that shows transient bacteremia from gastrointestinal, genitourinary, and dental procedures is an uncommon cause of vascular graft infections. 15 Additionally, our literature review found no consensus guidelines for managing endoleaks in the setting of cholangitis.
Conclusion
In conclusion, follow-up surveillance plays a vital role in patients after undergoing an EVAR for various reasons. It allows providers the opportunity to monitor the patient’s recovery progress and detect potential complications or endoleaks that may arise over time. 8 Providers can promptly address complications and initiate appropriate treatment to improve patient outcomes. Coexistent cholangitis and endoleaks demand an individualized approach to patient management. Cholangitis can result in secondary infection of endovascular grafts and is associated with significant morbidity and mortality in the setting of endoleaks and eventual aneurysmal rupture. Early medical therapy, appropriate antibiotic coverage, and adequate follow-up are recommended to avoid graft infection and ensure proper recovery.
Footnotes
Acknowledgements
The authors are grateful to the patient for the learning opportunity, as well as our colleagues from the gastroenterology department for providing crucial support in managing this case.
Author contributions
All authors contributed to the conduct of this research. All authors have read and approved the final version of the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
