Abstract
Mucormycosis is an acute and aggressive fungal infection usually, but not exclusively, occurring in immunocompromised individuals. Lack of knowledge and awareness in developing countries hinders timely management. This case highlights the importance of a prompt index of suspicion for the timely and aggressive intervention of mucormycosis to the Tanzanian community. We report a case of fatal rhino-orbital-cerebral mucormycosis in a diabetic patient presenting at a tertiary hospital in the Kilimanjaro region, Tanzania. Nasal deformity, proptosis, chemosis and left-sided hemiplegia were evident on physical examination. Nasoendoscopy revealed extensive necrosis of the nasal septum, inferior turbinate and involvement of the frontal recess. Computed tomography and magnetic resonance imaging of the paranasal sinuses and head revealed necrosis and ischemic changes due to fungal invasion. The patient deceased due to intracranial complications of advanced fungal invasion. Late presentation and unacquainted with mucormycosis our patient had an unfavourable outcome. Early diagnosis, extensive surgical debridement and a multidisciplinary approach to treatment are of the essence in favour of a better prognosis.
Introduction
Rhino-orbital-cerebral mucormycosis is an acute, fulminating and highly fatal fungal infection requiring an urgent multidisciplinary approach. 1 Mucormycosis describes infections caused by fungi of the order Mucorales. The most frequently reported pathogens in mucormycosis are Rhizopus spp, Mucor spp and Lichtheimia spp (formerly of the genera Absidia and Mycocladus), followed by Rhizomucor spp, Cunninghamella spp, Apophysomyces spp and Saksenaea spp. Mucormycosis is classified based on its anatomic site and clinical manifestation as rhinocerebral, pulmonary, cutaneous, gastrointestinal, disseminated and uncommon rare forms, such as endocarditis, osteomyelitis, peritonitis and renal infection. 2 No epidemiological data exist for rhinocerebral mucormycosis in Tanzania. 3 In this report we describe a fatal case of rhinocerebral mucormycosis in an individual with diabetes type 2. This is the first case to have been identified and confirmed at our institution. We intend to advocate the identification of danger signs and urgency for early treatment.
Case presentation
A 49-year-old male, with a poorly controlled type 2 diabetes mellitus, presented with an acute onset of severe right-sided headache, nasal bleeding, purulent nasal discharge, facial swelling and facial pain 14 days before seeking medical attention. He was diagnosed with type 2 diabetes mellitus 2 years earlier with poor compliance with the use of glycaemic medications.
Upon arrival at the emergency department, the patient was fully conscious, febrile with a temperature of 39.2°C, pulse rate of 118 beats per minute, respiratory rate of 21 cycles per minute, oxygen saturation of 97% in room air and random blood glucose of 17 mmol/L.
Physical examination revealed a saddle nose deformity and an ulcerative lesion on the nasal alar, proptosis, periorbital oedema, chemosis (Figure 1) and loss of sensation in the ophthalmic and maxillary branches of the trigeminal nerve. Upon ophthalmologic review, a frozen eyeball with no perception of light in the right eye was evident. Nasoendoscopy by our ENT team showed yellow-brown tenacious and sticky mucopurulent discharge, tissue necrosis of the inferior turbinate and nasal septum. A computed tomography (CT) scan of the paranasal sinuses (Figure 2) revealed mucosal thickening with multiple air-fluid levels in the maxillary, ethmoid and sphenoid sinuses bilaterally suggestive of acute sinusitis. Cavernous sinus thrombosis and orbital apex syndrome were considered as differential diagnoses.
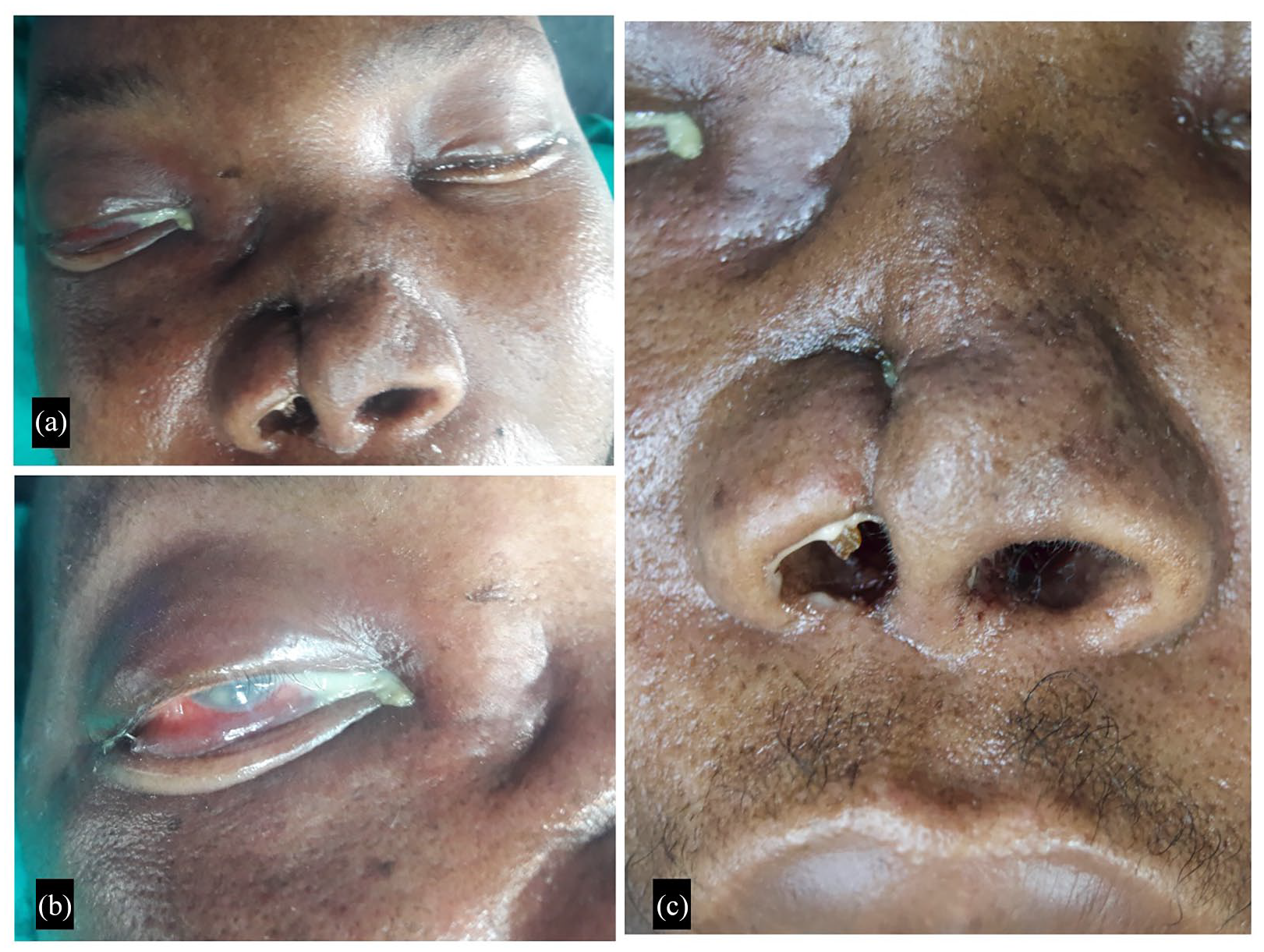
Patient features at the time of presentation demonstrating deformity of the nose with rhinorrhoea and crusting (a and c); chemosis, periorbital oedema and purulent discharge from the right eye (b).

(a) Axial CT of the paranasal sinuses demonstrate mucosal thickening with multiple air-fluid levels in the maxillary, ethmoid and sphenoid sinuses bilaterally. (b) Axial MR images of the brain show right temporal and basifrontal lobe abscesses (black arrowheads) with meningeal enhancement along the right frontoparietal convexity (white arrowhead).
The patient was started on intravenous meropenem and fluconazole in the initial course of treatment for acute invasive fungal sinusitis. On the third day of admission, he developed signs of meningism. Blood culture for bacterial growth was negative. Nasal swab for culture was positive for Klebsiella pneumoniae sensitive to aminoglycosides and carbapenems. Cerebral spinal fluid revealed elevated protein suggestive of meningitis. At this point, the patient’s blood glucose was now stabilized; however, his fever persisted. The patient was deemed unsuitable for surgical intervention.
Once stabilized, the patient was taken for endoscopic sinus surgery. Extensive debridement involving removal of the cartilaginous septum, inferior and middle turbinate and medial maxillary sinus wall was performed (Figure 3). Ethmoid, sphenoid and frontal sinuses consisted of necrotic mucosa which exposed dead bone of the perpendicular plate of the ethmoid and anterior skull base. The left nose was in view through the septal defect. There was minimal involvement of the left lateral nasal wall. Grocott–Gomori methenamine silver (GMS) staining of tissue biopsies obtained for histopathology analysis was confirmatory for mucormycosis (Figure 4).

Endoscopic images of the nose showing necrosis of the inferior turbinate (IT) and nasal septum (NS) of the right nose (a), fungal spores (b, white arrowhead); exposed and necrosis of the perpendicular plate of the ethmoid and anterior skull base (c and d, black arrowhead and asterisks). The left middle turbinate (MT) and lateral nasal wall mucosa appear of normal consistency.
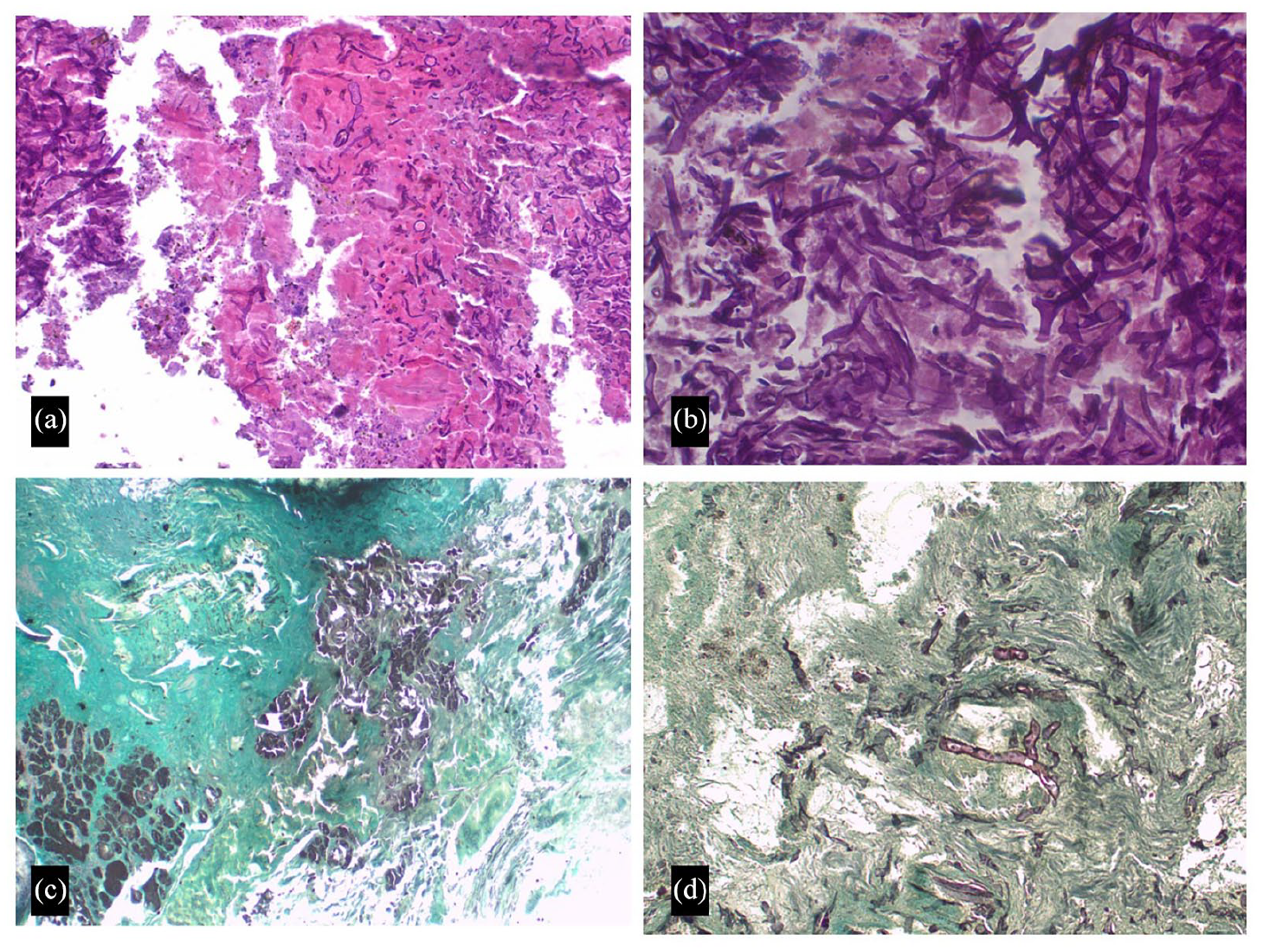
Hematoxylin and eosin stain photomicrographs (a and b) show a lesion with bony and cartilaginous tissue, areas of extensive necrosis and inflammatory cells infiltration. There are areas with aggregates of broad non-septate fungal hyphae branching at 90° with few conidia. Grocott–Gomori methenamine silver stain (GMS) shows the grey and black appearance of mucormycosis organisms with peripheral outline (c and d).
In the subsequent seven postoperative days, the patient deteriorated to develop left-side hemiparesis. A magnetic resonance imaging (MRI) of the brain revealed right temporal and basifrontal lobe abscesses with meningeal enhancement along the right frontoparietal convexity. The right temporal abscess compresses the right internal carotid artery in the cavernous sinus (Figure 2).
The patient’s health status continued to decline while neurosurgery was consulted. However, the family decided to proceed with home care given the poor prognosis anticipated. The patient deceased 4 days later.
Discussion
Many fungi can cause acute invasive fungal rhinosinusitis (AIFRS). Common isolates include Aspergillus, Mucor, Rhizopus and Rhizomucor. 4 Mortality is high while poorer outcomes are linked with immunosuppression, mucormycosis, orbital and intracranial involvement. Diabetes mellitus is associated with 36%–88% of cases as a predisposing condition.1,5,6 Prevalence of glucose impairment in northern Tanzania is estimated to be 21.7% of which diabetes accounts for 5.7% of the population while awareness and medical treatment are low. 7
Mucormycosis infections are characterized by extensive angioinvasion that results in vessel thrombosis and subsequent tissue ischemic necrosis consequently hindering the delivery of leucocytes and antifungal agents to the foci of infection making eradication of the fungus difficult.8–10 In an event where the cavernous sinus is involved, direct cerebral seeding may occur via the ethmoidal and orbital veins draining this area. 11 Inhalation of sporangiospores is the main route of infection in immunocompromised individuals which predisposes them to pulmonary infection. Rhino-orbital-cerebral mucormycosis that originates in the paranasal sinuses with an incursion into the eye, orbit and brain is mainly seen in diabetic patients.12,13
As seen in our case, fulminant progression of symptoms highlights the severity of the condition. Cranial nerve palsy, diplopia, mid-facial pain, proptosis, periorbital oedema, orbital apex syndrome and nose and palate ulcer should raise a high index of suspicion in diabetic patients. 14
A multidisciplinary and multimodal approach to the treatment of mucormycosis is emphasized for better management. In developing countries, such services can only be available in tertiary care centres.12,15 Nasal endoscopy is ideal in early recognition of features suggestive of necrosis and aid in biopsy for histopathology to confirm the diagnosis. In diabetic patients, high chance of fungal infection is associated with uncontrolled diabetes. Diabetic ketoacidosis favours rapid invasion of the fungus due to the high affinity of the Mucorales to the acidic environment.11,12 An MRI is the imaging of choice in suspicion of orbital or brain involvement. Recommended treatment includes aggressive debridement of all involved tissues and adjuvant liposomal amphotericin B. Craniofacial resection and repair of the skull base may be required. Early intervention must be addressed to minimize intradural and intracerebral extension. Sufficient reversal or control of the immunosuppression favours a good prognosis.6,16
Conclusion
Early detection and aggressive management are of critical importance in eradicating mucormycosis. Late diagnosis is associated with catastrophic outcomes. Although histopathology services are available in limited-resource settings, untimely dissemination of results hinders expedited treatment as it was in our case. We highly consider this report to be enlightening to the health practitioners of symptoms encompassing the nose, eye and brain in diabetic and other immunocompromised patients to be a high index of suspicion of an invasive mucormycosis infection. Rarely this condition can also occur in immunocompetent individuals.
Footnotes
Acknowledgements
We register our sincere appreciation to the family of the deceased patient for providing informed and written consent to publish this report. The effort of health workers who have contributed to the management of the patient despite the unfortunate outcome is recognized and acknowledged.
Author contributions
D.C.C. conceptualized and wrote the first and final versions of the manuscript. M.K., M.M., A.S., P.A. collected and summarized patient’s data. D.K., P.S., P.M. read and contributed to the revision of the draft. All authors read and accepted the final version for submission.
Consent
This case follows the World Medical Association Declaration of Helsinki. Written informed consent was obtained from the patient’s family representative before inclusion in this report. A copy of the written informed consent is available for review by the Editor-in-Chief upon request.
Data availability
We have not shared patients’ hospital records as they contain personal identification information.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical approval
Institutional ethical approval is not required for reporting individual cases and or case series.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
