Abstract
Objectives:
Invasive fungal sinusitis is associated with substantial morbidity and mortality influenced by multiple factors; however, evidence regarding prognostic determinants of survival remains limited. This study aims to evaluate survival outcomes and identify prognostic factors in patients with invasive fungal sinusitis to support evidence-based treatment planning.
Methods:
A retrospective study was conducted on patients with invasive fungal sinusitis treated between February 2000 and June 2018, with follow-up ending on 30 April 2021. Survival curves were analyzed using the Kaplan–Meier method and log-rank test, while Cox proportional hazards models were employed for univariate and multivariate analyses of prognostic factors.
Results:
One hundred sixteen patients were diagnosed with invasive fungal sinusitis, and these patients were further classified into an acute sinusitis group (80 patients (69.0%)) and a chronic sinusitis group (36 patients (31.0%)). The overall 1-year survival rate was 49.0% (median survival time of 9.8 months). Among acute invasive fungal sinusitis patients, the 1-year survival rate and median survival time were significantly shorter than those of chronic invasive fungal sinusitis patients. Compared with
Conclusions:
Invasive fungal sinusitis still has a high mortality rate, particularly in patients with chemosis, altered consciousness and leukopenia, necessitating aggressive treatment.
Introduction
Invasive fungal sinusitis has a lower incidence than the overall incidence of rhinosinusitis, and it is categorized into acute and chronic forms. This condition is associated with high morbidity and mortality, especially in cases of acute invasive fungal sinusitis is particularly fulminant, characterized by rapid angioinvasion, vascular thrombosis and extensive tissue necrosis, 1 with an overall mortality rate of approximately 64.9%, 2 and a disease-specific mortality rate of approximately 51.4%. 2 Conversely, chronic invasive fungal sinusitis follows a more indolent yet destructive course and has a 1-year overall mortality rate of 52.0%. 3 Despite clear differences in their progression and patterns of tissue invasion, both acute and chronic invasive fungal sinusitis are associated with poor survival outcomes because these opportunistic fungi can rapidly spread from the nasal or paranasal sinuses to the orbit and intracranial structures once infection is established. 4 Therefore, recognizing the clinical patterns is essential for early management to reduce morbidity and mortality. In addition, factors that influence disease progression should also be carefully considered.
The main factors influencing the survival of invasive fungal sinusitis patients include disease-related variables, patient-related factors and treatment-related factors. First, disease-related variables include the type of pathogenic fungal organism, the duration of the illness, the extent and rate of disease progression and the degree of tissue invasion at presentation. These factors influence the severity of local destruction, the likelihood of orbital or intracranial extension and ultimately the patient’s prognosis. Second, patient-related factors include underlying health conditions and the immune status of the individual. Recent decades have seen a global rise in invasive fungal sinusitis, attributable to the increased prevalence of diabetes mellitus, expanding immunosuppressed populations and the widespread application of chemotherapy, immunosuppressant after transplant and prolong steroids therapies.5,6 Finally, treatment-related factors include the timeliness with which medical attention is sought and the specific therapeutic interventions provided. Diagnostic delays remain a major challenge, as the early clinical manifestations of invasive fungal sinusitis often resemble uncomplicated rhinosinusitis. Definitive diagnosis requires histopathologic confirmation of fungal invasion; however, sampling limitations and overlapping morphologic features may impede early organism identification, further postponing treatment initiation. Management typically involves urgent surgical debridement in conjunction with systemic antifungal therapy, yet treatment is often complicated by the need for repeated debridements, difficulties in achieving complete removal of devitalized tissue, antifungal toxicities and organism-specific drug resistance. Consequently, patient outcomes are shaped by a complex interplay of disease-related, host-related and treatment-related factors. Reported prognostic determinants include the extent of disease at presentation, underlying immunocompromised status, timeliness and adequacy of surgical intervention and the causative fungal organism.
The survival rate of invasive fungal sinusitis patients has been addressed in previous studies. Cho et al. 7 conducted a study involving 45 patients with acute invasive fungal sinusitis and reported an overall survival rate of 53%. Turner et al. 8 performed a systematic review and reported an overall survival rate of approximately 46% among patients with acute invasive fungal sinusitis. Most previous studies have mainly focused on acute invasive fungal infections, whereas data on chronic invasive fungal infections remain limited. Humphreys et al. 3 analyzed the 1-year overall survival rate among 38 patients with chronic invasive fungal infections, and they reported a rate of 48%.
Regarding the relationship between pathogenic fungal organisms and survival rates, Foshee et al.
9
conducted a retrospective study involving 27 patients diagnosed with invasive fungal sinusitis and reported an overall 1-year mortality rate of 57.7%. The study revealed that mucormycosis was associated with a significantly severe mortality rate, with an average time to death of 32.8 days, compared with 148 days for aspergillosis. Moreover, Trief et al.
10
reported that the mortality rate of invasive fungal sinusitis patients in the mucormycosis group was 71.4%, whereas that in the aspergillosis group was 28.5%. However, these previous studies did not mention the relationship between fungal organisms and survival rates of patients with acute and chronic forms of the disease because both
According to the available literature review, both
Materials and methods
A retrospective study was conducted to review the medical records of patients with invasive fungal sinusitis between February 2000 and June 2018. The end of the follow-up was indicated on 30 April 2021. All of the patients were confirmed to have a diagnosis through histopathological examinations; however, patients infected with unknown fungal organism or those with insufficient data, including date of death and last status prior to June 2018, were excluded.
Survival analysis 13 was conducted to calculate sample sizes, and the prevalence of invasive fungal sinusitis was estimated to be 0.54 on the basis of the study of Chen et al. 14 The relative risk was determined to be 2.84, 8 with a probability of an uncensored observation of 1 and a correlation of 0.8 with other covariate factors; therefore, a sample size of 81 patients was determined to be needed for the study, with a 95% confidence interval and a 2% error. However, to ensure a comprehensive evaluation of the management of invasive fungal sinusitis, all medical records of patients who met the eligibility criteria were reviewed.
In this study, well-controlled diabetes mellitus was defined as blood glucose levels consistently within the target range (80–130 mg/dL) or an HbA1c <7%, without frequent hypoglycaemic episodes or acute metabolic complications. Chronic kidney disease was defined as either decreased kidney function (estimated glomerular filtration rate (eGFR) <60 mL/min/1.73 m2) or evidence of kidney damage (such as persistent proteinuria/albuminuria, structural abnormalities, or abnormal urine sediment) persisting for more than 3 months. Organism identification was confirmed histopathologically by demonstrating fungal hyphae on routine Hematoxylin and Eosin (H&E) staining and on special stains, including Grocott’s Methenamine Silver (GMS) and Periodic Acid-Schiff (PAS). These stains highlighted morphologic characteristics consistent with
Regarding treatment at our institution, surgery remained a key component of management. The surgical approach was individualized according to the severity and anatomic extent of disease as assessed by nasal endoscopy and imaging. Localized disease confined to a single sinus was managed with targeted endoscopic procedures, including maxillary antrostomy or sphenoidotomy. In contrast, pansinusitis required a more comprehensive approach, including full-house endoscopic sinus surgery, to achieve adequate debridement and drainage. When the disease extended into critical regions involving the orbit, skull base, hard palate, or other extrasinus compartments, more aggressive or combined surgical strategies (i.e. orbital exenteration and palatectomy) were required to achieve adequate disease clearance and protect vital structures. In addition, most patients were initiated on empirical amphotericin B. Once clinically stabilized, they were transitioned to targeted antifungal therapy based on culture or histopathologic results, typically voriconazole for 6–12 weeks for
After treatment, rehabilitation played an important role in restoring function and improving quality of life. Patients with orbital defects often required an orbital prosthesis, while those with palatal defects benefited from the use of an obturator to aid in speech and swallowing. These rehabilitative measures contributed significantly to overall functional recovery following disease control.
The study was conducted in accordance with the Declaration of Helsinki and was approved by the Local Human Ethics Research Committee (HE621212). Given the retrospective nature of the study, a waiver of written informed consent was granted and was applied consistently throughout the manuscript.
Data analyses
Descriptive data are presented as percentages and means ± standard deviations. Survival curves were analyzed via the Kaplan–Meier method, and statistical significance was assessed via the log-rank test. Univariate and multivariate analyses for prognostic factors were conducted via Cox proportional hazards models. A value of
Results
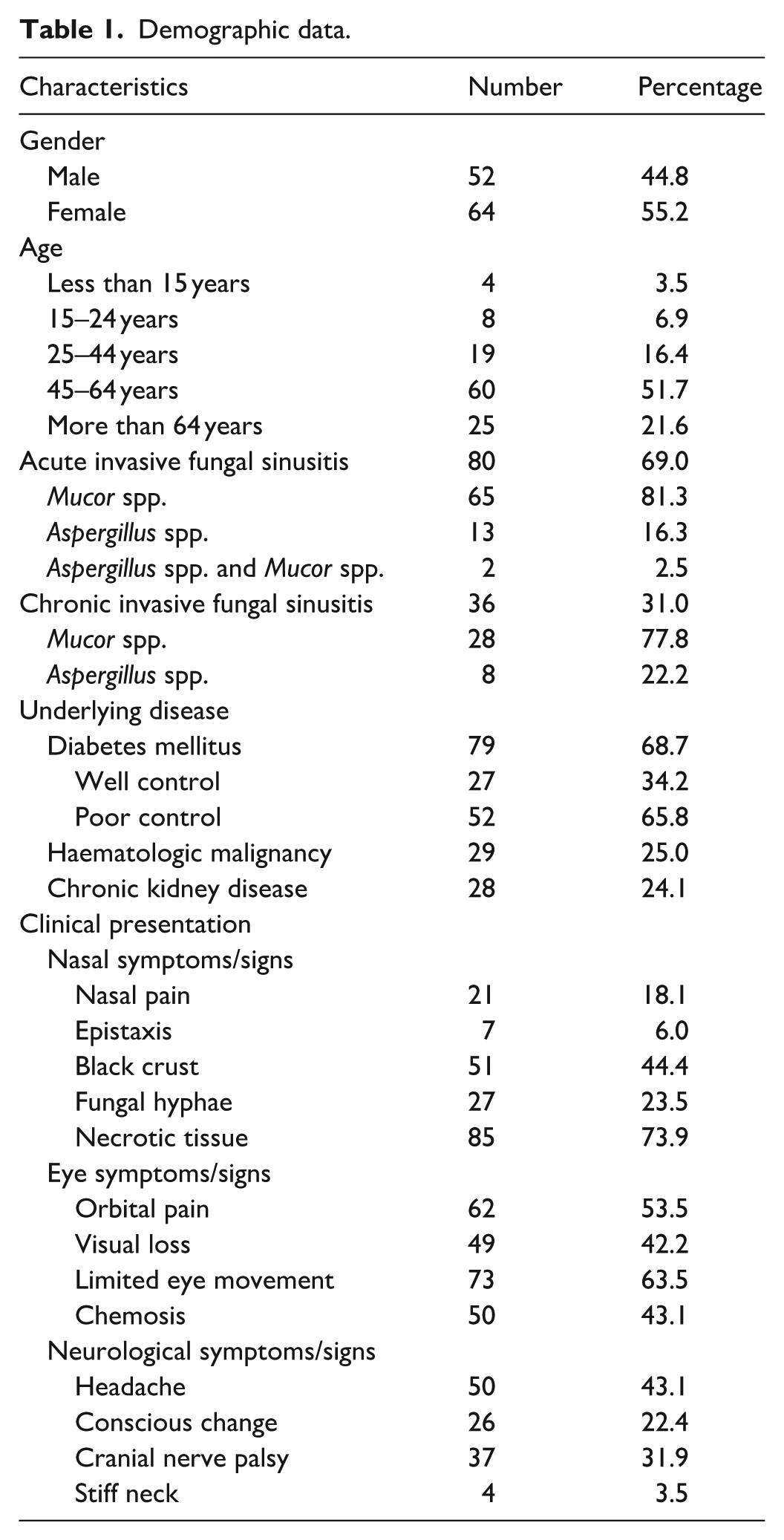
One hundred sixteen patients (52 males and 64 females) met the eligibility criteria for review of their medical records. The 45- to 64-year-old age group represented half of our patient population. Acute invasive fungal sinusitis was diagnosed in 80 patients, with
Demographic data.
The computed tomography findings revealed that the maxillary sinus was the most common site of sinus involvement, with 53.0% of the other surrounding structures involving the orbit, 36.0% involving intracranial extension and 9.0% involving the palate. Most patients received treatment within 2 weeks after diagnosis; however, seven patients experienced delays due to the patients’ unstable clinical statuses. At the end of the study, 28 patients survived, while 88 had died (Table 2).
Investigation and treatment outcomes.

CT: computed tomography.
The 1-year survival rate for all patients with invasive fungal sinusitis was 49.0%, with a median survival time of 9.8 months (Figure 1(a)). Notably, the overall survival analysis revealed that patients with chronic invasive fungal sinusitis had better outcomes than those with acute fungal sinusitis (Figure 1(b)).

The 1-year survival rate of invasive fungal sinusitis patients (a) was 49.0% (95% CI: 39.7–57.8), and the median survival time was 9.8 months. Additionally, the 1-year survival rates of patients with acute and chronic invasive fungal sinusitis were analyzed and were 41.2% (95% CI: 30.4–51.7) and 66.6% (95% CI: 48.7–79.4), respectively. The median survival time for patients with acute invasive fungal sinusitis was 4.9 months (95% CI: 2.0–12.5), whereas the median survival time for those with chronic invasive fungal sinusitis was 22.0 months (95% CI: 9.7–77.2). The differences in survival between patients with acute and chronic disease were statistically significant (
A survival analysis was conducted according to the organisms identified in patients with acute and chronic invasive fungal sinusitis. In the acute disease group, patients with

One-year survival rates were assessed on the basis of the fungal organisms that were detected in patients with acute and chronic sinusitis. Acute aspergillosis sinusitis patients had a 1-year survival rate of 53.9% (95% CI: 24.8–76.0) and a median survival time of 13.4 months (95% CI: 2.0–21.2), whereas acute mucormycosis sinusitis patients had a 1-year survival rate of 42.3% (95% CI: 40.3–53.8) and a median survival time of 4.9 months (95% CI: 1.6–13.3) (a). With respect to patients with chronic sinusitis (b), patients with aspergillosis had a 1-year survival rate of 62.5% (95% CI: 22.9–86.1) and a median survival time of 18.2 months (95% CI: 1.5–40.2), whereas patients with mucormycosis had a 1-year survival rate of 68.8% (95% CI: 48.6–82.4) and a median survival time of 24.8 months (95% CI: 9.7–106.1).
Furthermore, prognostic factors were evaluated using univariate Cox regression analysis. Altered consciousness and a white blood cell (WBC) count <4000 cell/mm³ emerged as significant predictors, with hazard ratios of 2.1 and 1.8 (
Univariate and multivariate analysis of variables associated with overall survival.

WBC: white blood cell; VA: visual acuity; EOM: extraocular movement; 95% CI: 95% confidence interval.
Discussion
Invasive fungal sinusitis is an opportunistic infection associated with a significant mortality rate, particularly among immunocompromised patients. Therefore, increasing awareness of patients’ underlying clinical status is critical for timely diagnosis and intervention. In our cohort, a substantial proportion of patients had predisposing comorbidities, with diabetes mellitus present in 68.7% of cases. Notably, poorly controlled diabetes accounted for 50.9%, highlighting a major driver of impaired host immunity. This differs from previous studies that identified haematologic malignancies as the predominant underlying condition.2,15 In our study, haematologic malignancies were the second most common comorbidity, occurring in 25% of patients. Both conditions profoundly compromise immune function because diabetes reduces neutrophil activity and microvascular function, 16 while haematologic malignancies weaken immune responses through the disease process and treatments. 17 These factors reduce the host’s ability to control opportunistic pathogens.
The variation in predominant underlying diseases compared with the literature may reflect differences in regional epidemiology, referral patterns and the prevalence of chronic diseases in each setting. It also highlights the importance of understanding local risk factors when developing diagnostic pathways or resource allocation strategies. Despite early initiation of surgical debridement and antifungal therapy, the mortality rate remained high. This suggests that outcomes are affected not only by the timing of treatment but also by the degree of underlying immunosuppression, the rapid progression of acute invasive forms and possible delays in recognizing early symptoms. These findings reinforce the need for a high level of vigilance in high-risk groups and emphasize the importance of multidisciplinary care, optimization of comorbidities such as glycaemic control, and improved strategies for earlier detection.
The 1-year survival rate for our patients with invasive fungal sinusitis was 49%, with a median survival duration of 9.8 months. Additionally, when we analyzed the 1-year survival rate of patients with acute and chronic forms of the disease, we found that patients with acute disease had a shorter survival rate than did those with chronic disease. These data indicate that the acute form is associated with more severe outcomes than the chronic form. Moreover, the acute form had a survival rate of 41.2%. This finding is similar to that of Cho et al.,
7
who reported a survival rate of 47%. In our cohort, the survival rate of patients with
With respect to chronic disease, we observed a 1-year survival rate of 67.5%, which exceeds the 48.0% reported by Humphreys et al.
3
This discrepancy may be attributed to variations in patient characteristics, underlying conditions and the specific fungal organisms that are involved. Although mucormycosis typically demonstrated a highly aggressive behaviour that resulted in an acute clinical presentation, and chronic mucormycosis had been documented,20,21 albeit infrequently. Reported cases indicated that chronic forms tended to occur in individuals with partial or intermittent immunosuppression, in whom the disease progressed in a more indolent manner compared with the classic acute presentation. In our cohort, chronic invasive fungal sinusitis was identified in 36 patients, of whom 28 were infected with
Prognostic factors were analyzed via multivariate Cox regression, which demonstrated three significant factors, including chemosis, altered consciousness and a WBC count of less than 4000/mm³. Chemosis exhibited a hazard ratio of 1.8. This finding was consistent with the studies conducted by Trief et al. 10 and Ngaotepprutaram et al., 15 who reported an increased mortality rate when the fungal infection involved the orbit. Altered consciousness had a hazard ratio of 2.5, which is comparable to the findings of Turner et al., 8 who reported that patients presenting with altered mental status had the highest risk of death, with a survival rate of only 9.1%. Furthermore, when the disease progressed to the brain, the survival rate decreased to 25%. Finally, a WBC count of less than 4000/mm³ had a hazard ratio of 3.1, thus indicating a similar trend of low WBC counts as that reported in the study by Cho et al. 7 and indicating the association of this trend with increased fungal infection severity.
Invasive fungal sinusitis is a rare condition, resulting in a limited number of patients being available for analysis, particularly for subgroup analyses. In our cohort, we identified 116 patients with invasive fungal sinusitis; however, only 8 of these patients had chronic aspergillosis sinusitis. This small sample size may be insufficient for meaningful comparisons with patients with other types of invasive fungal sinusitis. Recent literature has also suggested a possible association between COVID-19 and an increased risk of invasive fungal sinusitis, potentially related to elevated iron levels and the widespread use of systemic corticosteroids in affected patients.22–24 However, cases diagnosed after 2018 could not be included in our study because complete and reliable clinical and follow-up data were unavailable, and incorporating these cases would have compromised the consistency and validity of the analysis.
Additionally, the limitations of this study include the inability to retrieve all certain data due to its retrospective design. Furthermore, we lacked fungal culture data to substantiate the diagnoses, which may impact the robustness of our findings.
Conclusion
Overall, survival analysis of patients with invasive fungal sinusitis revealed that survival was not high, especially for patients with acute disease, which was more aggressive than chronic disease.
Footnotes
Acknowledgements
The authors thank Mr. Chalongpon Santong for assistance with data analysis.
Ethical considerations
Ethical approval for this study was waived by Khon Kaen University Ethics Committee in Human research (Approval number: HE621212/ID: IRB00001189). Due to the retrospective nature of our study, patient information was obtained from the patients’ previous medical records. Consequently, the Ethics Committee approved a waiver of the written informed consent requirement.
Consent to participate
Given the retrospective nature of the study, the Local Human Ethics Research Committee’s waiver of written informed consent is applied consistently throughout the manuscript.
Author contributions
PM, PK and PV contributed to the study design, data collection, analysis and interpretation of the data. PM and PK prepared the article draft and revised it critically. CT, PU, SS and WR assisted in revising the article. All authors read and approved the final version of the manuscript.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
