Abstract
In recent years, various devices have been approved for peripheral artery disease with femoropopliteal lesions. However, treatment of long, calcified, and diffused lesions is still challenging because these lesions are associated with restenosis. This report described the case of an 82-year-old man with bilateral severely calcified and diffused long lesions in the superficial femoral artery that was treated using polymer-coated paclitaxel-eluting stent and interwoven nitinol stent. After 6 months, in-stent restenosis was observed at the implantation site of the interwoven nitinol stents. Polymer-coated paclitaxel-eluting stents were deployed at the in-stent restenosis site. After another 6 months, angiography and intravascular ultrasound imaging revealed no restenosis at the polymer-coated paclitaxel-eluting stent site. Optical coherence tomography was also performed, revealing that the stent struts were well covered by neointima, which was very thin at approximately 0.1 mm. This representative case demonstrated substantial differences in the effects of devices; in other words, the superiority of polymer-coated paclitaxel-eluting stent in treating long, diffuse, and calcified lesions indicated that its implantation is a reasonable option when the initial gain was obtained following sufficient vessel preparation.
Keywords
Introduction
To date, various devices have been approved for endovascular therapy (EVT) of peripheral artery disease (PAD) with femoropopliteal lesions. However, treatment of long, calcified, and diffused lesions is still challenging because these lesions are associated with restenosis.1,2 Recently, the Eluvia™ polymer-coated paclitaxel-eluting stent (PC-PES) (Boston Scientific, Marlborough, MA, USA) and the Supera™ interwoven nitinol stent (IW-NS) (Abbott Laboratories, Abbott Park, IL, USA) were approved for the superficial femoral artery (SFA) in Japan.
Herein, we describe a case in which the PAD in the bilateral SFA was treated using both PC-PES and IW-NS. Six months after the intervention, in-stent restenosis (ISR) was observed only at the IW-NS implantation sites. From this experience, we also discuss appropriate EVT strategies for long and calcified lesions to maintain long-term patency.
Case presentation
The patient was an 82-year-old man on hemodialysis. In July 2019, he presented with bilateral intermittent claudication (Rutherford classification 3). His ankle-brachial index (ABI) was 0.64 in the right side and 0.66 in the left side. Computed tomography revealed long, calcified, and diffused lesions in bilateral SFA (Figures 1(a1) and 2(a1)). Despite medication therapy, the claudication did not disappear, so EVT was performed. Pre-procedure angiographic findings are shown in Figures 1(a2) and 2(a2). The right SFA was pre-dilated by a 6-mm non-compliant balloon. Moreover, a 5.5 × 80-mm IW-NS, 6.0 × 80-mm PC-PES, 6.0 × 80-mm IW-NS, and 7.0 × 80-mm PC-PES were implanted from the distal to the proximal site to cover all SFA lesions after ballooning (Figure 1(a3)). The left SFA was pre-dilated by a 5-mm non-compliant balloon. Moreover, a 5.5 × 120-mm IW-NS, 7.0 × 120-mm PC-PES, and 7.0 × 80-mm PC-PES were implanted from the distal to the proximal site to cover all SFA lesions after ballooning (Figure 2(a3)). For both SFA lesions, we selectively deployed IW-NSs for calcified lesions, which were detected using fluoroscopy and intravascular ultrasound (IVUS) (circumferential grade of calcium plaque quantification ⩾ Grade 33). All IW-NSs were deployed at a length that was the same as the reference length, that is, neither elongated nor shortened. After the procedure, intermittent claudication was resolved, and the ABIs were improved to 1.10 (right side) and 1.10 (left side).
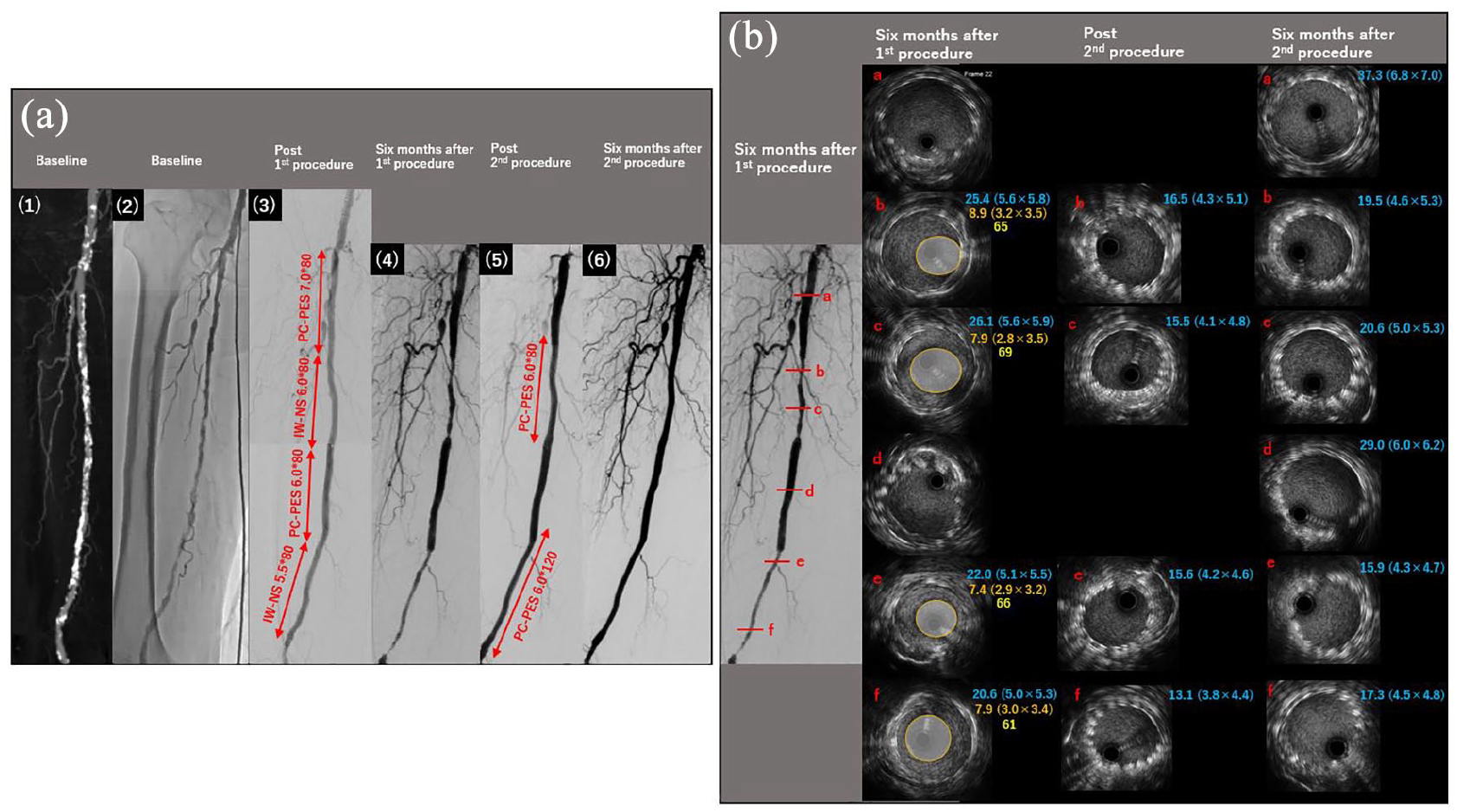
CT, angiographic, and IVUS findings of the right SFA. (a) CT and angiographic findings of the right SFA: (1) baseline CT, (2) baseline angiography, (3) after the first procedure, (4) 6 months after the first procedure, (5) after the second procedure, and (6) 6 months after the second procedure. (b) IVUS findings of the right SFA. Blue text: stent area (mm2) (short diameter (mm) × long diameter (mm)); orange text: lumen area (mm2) (short diameter (mm) × long diameter (mm)); and yellow text: % stenosis (calculated as 100 − lumen area/stent area × 100).

CT, angiographic, and IVUS findings of the left SFA. (a) CT and angiographic findings of the left SFA: (1) baseline CT, (2) baseline angiography, (3) after the first procedure, (4) 6 months after the first procedure, (5) after the second procedure, and (6) 6 months after the second procedure. (b) IVUS findings of the left SFA. Blue text: stent area (mm2) (short diameter (mm) × long diameter (mm)); orange text: lumen area (mm2) (short diameter (mm) × long diameter (mm)); and yellow text: % stenosis (calculated as 100 − lumen area/stent area × 100).
However, 6 months after the first procedure, his symptom recurred and ABI decreased to 0.60 (right side) and 0.65 (left side). Angiography revealed ISR at the site where IW-NSs were deployed (Figures 1(a4) and 2(a4)). IVUS findings are shown in Figures 1(b) and 2(b). IW-NSs were relatively well expanded based on the reference diameter of each stent; however, significant lumen loss was observed. The stent area (stent diameter), lumen area (lumen diameter), and % stenosis of each lesion where IW-NSs were implanted are also shown in Figures 1(b) and 2(b). On the contrary, PC-PESs did not show significant restenosis and were kept well expanded. We hypothesized that neointima hyperplasia was the main reason for the restenosis of IW-NS because all IW-NSs were deployed at a length that was similar to the reference length and were relatively well expanded. Moreover, calcification or significant thrombus was not detected at areas of ISR, and dual antiplatelet therapy had been continued. For the restenosis at the right SFA, a 6-mm scoring balloon and a 6-mm non-compliant balloon were used for pre-dilation. Thereafter, a 6.0 × 120-mm PC-PES was implanted to the distal ISR site, and a 6.0 × 80-mm PC-PES was implanted to the proximal ISR site (Figure 1(a5)). For the restenosis at the left SFA, a 6-mm scoring balloon and a 6-mm non-compliant balloon were also used for pre-dilation. Then, a 6.0 × 120-mm PC-PES was implanted to the ISR site (Figure 2(a5)). Implanted stents were post-dilated by a 6-mm non-compliant balloon. Then, IVUS imaging showed that the newly implanted PC-PESs were relatively well expanded (Figures 1(b) and 2(b)). After the second procedure, intermittent claudication resolved and the ABIs were improved to 1.10 (right side) and 1.13 (left side). As a result, claudication has not recurred. Angiography was performed 6 months after the second procedure. Restenosis was not observed in the stent implantation sites of the bilateral SFA lesions (Figures 1(a6) and 2(a6)). We reperformed IVUS assessment at the same sites of the first and second procedures. PC-PESs that were implanted 1 year ago had well-maintained expansion, and the lumen area was kept sufficiently. PC-PESs that were implanted 6 months ago in the implantation site of IW-NSs also had well-maintained expansion, and the lumen area was kept sufficiently (Figures 1(b) and 2(b)). Moreover, we performed optical coherence tomography (OCT) to assess neointimal proliferation in detail. The axial resolution of OCT is 10 times higher than that of IVUS; therefore, OCT imaging provides a much clearer view of the area between the lumen and plaque/neointima surface. 4 Representative OCT images of each stent are shown in Figure 3. Stent struts were entirely covered by the neointima, and the neointima was very thin (approximately 0.1 mm), which did not affect the lumen area.

OCT findings of the right SFA: (a) 7 × 80-mm PC-PES, (b) 6 × 80-mm PC-PES in 6 × 80-mm IW-NS, (c) 6 × 80-mm PC-PES, and (d) 6 × 120-mm PC-PES in 5.5 × 80-mm IW-NS.
Discussion
Although many useful devices have been approved, treatment of long, calcified, and diffused lesions in the femoropopliteal artery is challenging. Moreover, hemodialysis is also one of the predictors of restenosis after EVT for femoropopliteal artery lesions.1,2
As our EVT strategy, we chose primary stenting because this patient had a high risk of restenosis if treated with drug-coated balloon (DCB) alone.5,6 To maintain long-term patency after EVT, chemical mechanical supports were needed. Chemical supports include the drug effects of paclitaxel and fluoropolymer, and mechanical support was achieved using a scaffold.
Several studies have shown that the 1-year late lumen loss and the neointimal volume in the PC-PES group were significantly lower than that in the polymer-free paclitaxel-coated stent (PF-PCS) group.7,8 Indeed, we performed OCT for the assessment of PC-PES. The neointimal proliferation of the PC-PES was relatively thin, and there was no difference in the thickness of the neointima between periods of PC-PES implantation. A previous study showed that OCT results at 90 days demonstrated significantly lower neointimal areas with PC-PES than with PF-PCS and bare nitinol stent (BNS) in a swine model. 9 To our knowledge, we are the first to report OCT findings of PC-PES implanted at different timings in the human body.
In general, restenosis after BNS in SFA lesions peaks at 1 year. 10 PC-PES has a biocompatible fluoropolymer that enables continuous paclitaxel delivery for at least 6 months. 11 Hence, PC-PES could greatly inhibit long-term neointima proliferation than BNS in our case.
For mechanical support in long, calcified, and diffused lesions, balloon angioplasty and DCB are not favorable to maintain long-term patency.5,6 Moreover, these lesions need certain vessel preparation with balloon angioplasty to optimize EVT. Hence, to prevent restenosis, a back-up scaffold using a nitinol stent is needed for calcified lesions. Tomoi et al. 8 reported that the PC-PES group tended to have a larger change rate of the stent volume than in the PF-PCS group at 1 year, suggesting that the PC-PES was likely to be more expanded over time. Although the relationship between different stent platforms and clinical outcomes is poorly investigated, the expansion capacity of the stent can be considered as one of the determinants of clinical outcomes.
Vessel preparation is mandatory to obtain optimal results of EVT. After we confirmed the initial gain in which the stent can expand sufficiently, deployment of PC-PES might be reasonable for long and calcified lesions. Our findings are supported by previous reports showing that patients with complete lesion coverage using PC-PES had better patency compared with those who were treated with a combination of PC-PES + DCB or PC-PES + bare metal stent/balloon angioplasty. 12 To our knowledge, this is the first case report that compared the clinical progress of PC-PES and IW-NS in the same patient. In the present case, PC-PESs showed good clinical performance after 6 months and 1 year. However, long-term follow-up is necessary to confirm the clinical performance of PC-PES.
Conclusion
PC-PES, a new-generation drug-eluting stent, can be the first choice for treatment of long and calcified lesions in the femoropopliteal artery because this device can suppress restenosis by providing both scaffold and drug support. Thus, a better understanding of the unique characteristics of each device and careful patient follow-ups are needed. Furthermore, selected devices should be well suited for each lesion in order to maintain long-term patency.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
