Abstract
We report the severe mercury poisoning of a 4-year-old child by a so far unknown route of exposure, namely, by skin-to-skin contact. The child was admitted to the hospital with episodic pain in his extremities, tachycardia, hypertension, increased sweating, behavioral changes and weight loss. Extensive examinations eventually revealed an acute mercury poisoning. The initial mercury levels were 19 µg/L in urine (reference level 0.4 µg/L) and 37 µg/L in blood (reference level 0.8 µg/L). A facial cream bought online, containing approximately 18% mercury, was identified as the primary source of intoxication. The symptoms improved after disposal of the cream and chelation therapy. Further analyses, home visits and interviews suggested that the child was accidently intoxicated by skin-to-skin contact with the mother, although other routes of exposure such as dust ingestion and surface-to-skin contact cannot be excluded. The mercury levels in urine and blood samples of the child and other family members as well as in domestic dust samples decreased considerably over time.
Introduction
Acute mercury poisoning is a rare condition in European countries nowadays. However, multiple cases of mercury poisoning due to the use of skin-whitening creams have been reported. These creams are used to achieve a lighter skin-tone, a desired ideal of beauty in many countries. Although not all skin-lightening creams contain substantial amounts of mercury, recent studies showed that mercury is regularly added to such creams.1–4 In vitro studies showed that inorganic mercury species such as mercurous chloride (calomel), mercurous oxide, mercuric chloride or ammoniated mercury, which are predominantly used in skin-whitening creams, could penetrate the skin. 5
The case reports associated with skin-lightening creams frequently describe enormous diagnostic difficulties connecting the symptoms to mercury poisoning. This can be explained by the rarity of such poisonings in developed countries as well as by the unspecific symptoms that are often attributed to other diseases. Our case, which involved a mercury-containing facial cream that was not advertised as a skin-lightening cream, reflects the considerable challenges of diagnosing a mercury intoxication and reveals novel exposure pathways.
Case
In Summer 2018, a 4-year-old boy was admitted with loss of weight (2 kg in 4 weeks), weakness, fatigue, strong sweating, arterial hypertonia (114/104 mmHg) and tachycardia (>150 bpm). Furthermore, he complained about non-specific abdominal pain in stomach. He complained about non-specific joint pain in his ankles, without any swelling, warming or redness, but severe enough that he demanded to be carried, instead of walking himself. The boy’s behavior was shy and uncooperative. According to his parents, he had been a lively and happy child before. Such a mixed symptomatology is uncommon in children, and arterial hypertonia and weight loss in particular merited detailed further analysis.
Blood tests were mainly unremarkable, except for elevated aldosterone levels. He was discharged and 3.75 mg Amlodipine per day was prescribed to control his blood pressure. However, the blood pressure continued to be increased, and the other symptoms remained as well, resulting in further diagnostic tests. A chest X-ray and whole-body magnetic resonance imaging showed no signs of tumors, but nonspecifically enlarged lymph nodes were noted in the axilla and under the carotid sheath. Microbiological studies and standard laboratory tests were negative. Due to the ongoing hypertonia and tachycardia, 3.75 mg Propranolol three times per day was added to his medication.
Six weeks after his first admission, the patient’s blood was tested for mercury, as one of the authors remembered a similar case years ago. The leading symptom for the differential diagnosis of a mercury intoxication was the arterial hypertension. 6 The mercury levels (Table 1) were extremely elevated compared to reference values and significantly above the HBM-II (danger level) value proposed by the Human Biomonitoring Commission of the Federal Environmental Agency (UBA).7,8 The mercury levels 6 weeks after the first admission were in urine 19 µg/L (reference level 0.4 µg/L; HBM-II level 25 µg/L), in urine corrected for creatinine 90 µg/g creatinine (no reference level; HBM-II level 20 µg/L) and in blood 37 µg/L (reference level 0.8 µg/L; HBM-II level 15 µg/L).
Urinary and blood mercury levels of all family members shortly after the discovery of the cream as the source of intoxication (start) and after 6 months (end).

Reference and threshold levels are provided by the German Federal Environmental Agency.7,8 n.a.: data not available; HBM: human biomonitoring.
Children (aged 6–14 years).
Adults (aged 18–69 years).
Soon after diagnosis, efforts were made to identify the source of exposure. Asked about any unusual household remedies connected to the family’s origin (Balkan), the mother explained that she had started using a face cream from Kosovo, which she had bought online. Laboratory analyses of the cream revealed a mercury content of ~18%. Mercury levels in comparable ranges have been previously reported for skin-lightening creams.2,4 However, the cream was not advertised as a skin-lightening cream, but for achieving a better skin appearance. According to the manufacturer, the cream has an “immediate effect and success in problems such as: Acne, pimples, facial scars after birth, sunspots, signs after acne, etc.” 9 Obviously, mercury was not listed as an “active” ingredient. Although we do not know the exact composition of the cream, Hg was likely added in the form of inorganic salts such as Hg2Cl2 or HgNH2Cl.10,11 The mother insisted that she had never used the cream on her children. Nevertheless, blood and urine samples from all family members were also analyzed (Table 1). As expected, the mother`s mercury levels were extremely high due to the use of the cream over several months. The patient’s 9-year-old brother also showed elevated mercury levels. Surprisingly, both mother and brother showed no clinical signs of mercury poisoning. The father’s mercury levels were only partially available and relatively low.
To exclude any other sources of exposure, the family’s apartment was thoroughly inspected. Ambient air monitoring revealed no elevated mercury levels compared to local background levels (~0.1 µg/m3). Analyses of dust samples from the family’s vacuum cleaner showed values of up to 68 µg/g dust, which is significantly higher than the reference value (0.15 µg/g) for indoor dust samples. 12 Repeated analyses of indoor dust samples showed a decrease in mercury levels over time (Figure 1). Dust samples from the parent’s bedroom, taken approximately 4 months after the identification of the source, showed higher levels compared to the other rooms (8.4 µg/g vs 3.3 µg/kg in the living room and 5.4 µg/g in the children’s bedroom).

Time course of mercury levels in the mother’s hair and indoor dust samples from the apartment. The age of the hair was calculated by assuming an average growth of 1 cm/month. Day zero is the date of the patient’s initial mercury biomonitoring.
In addition, hair samples (~30 cm long) from the mother were taken about 4 months after she had stopped using the cream. The hair was cut into 2-cm-long segments and analyzed. The results are shown in Figure 1. The lowest mercury levels were found closest to the scalp. Mercury levels increased with growth distance from the skin reaching up to 89 µg/g hair (alert level 1 µg/g hair). 13 Until 14 cm length, mercury levels increased exponentially. Thereafter, the increase was linear. The levels found in the mother’s hair were higher than what has been previously reported for the use of skin-lightening creams. 4
The patient immediately received oral chelation therapy with 40 mg Dimaval® (2,3-Dimercapto-1-propanesulfonic acid) twice per day. His condition improved rapidly and he could be discharged. Oral chelation therapy was continued until mercury levels were below the HBM-I value (Figure 2).
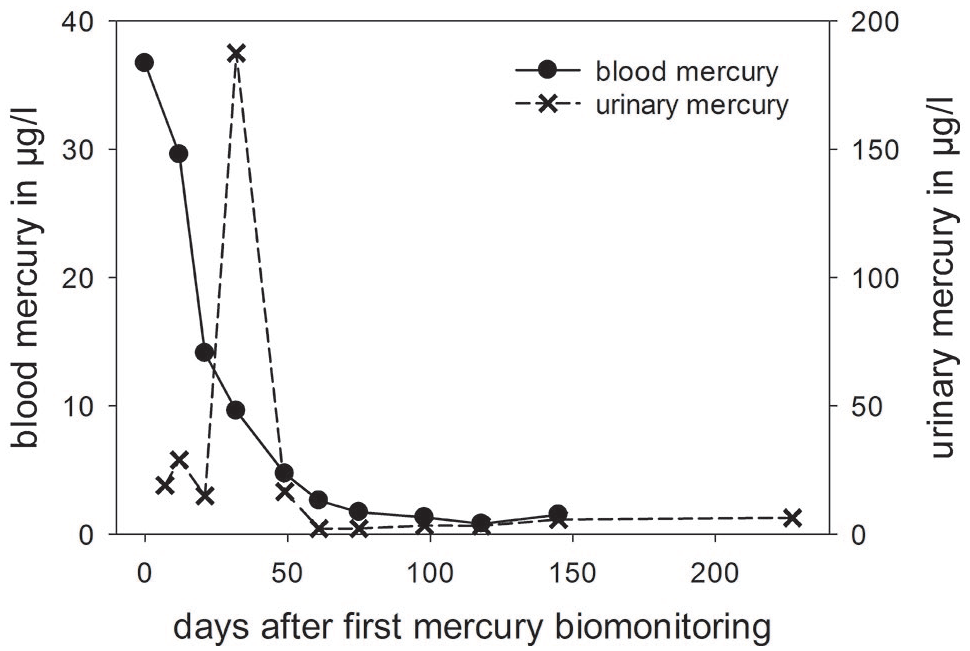
Time course of mercury levels in the patient’s urine and blood.
Six months after hospital discharge, the patient’s urinary and blood mercury levels decreased significantly (Table 1) and his symptoms had disappeared. Follow-up visits were by now performed for 18 months since treatment. No potential re-exposure was identified, no long-term effects were observed.
Discussion
We report the case of a child with a serious mercury poisoning due to his mother’s use of a mercury-containing facial cream. As described in similar case reports, the diagnosis was extremely challenging due to nonspecific symptoms that could be associated with other diseases.6,14 Unclear symptoms, especially in arterial hypertension, should be reason enough to think of mercury as a differential diagnosis and to have mercury determined in blood or urine. In this case, the final diagnosis was made approximately 6 weeks after the first symptoms occurred. The patient was immediately treated with an oral chelating agent. Consequently, urinary mercury excretion dramatically increased after the treatment started, but decreased rapidly after a couple of days along with the blood mercury levels (Figure 2). In contrast to the patient, both mother and brother did not show any clinical signs of mercury poisoning, although Hg levels in urine and blood were also significantly elevated. This may be explained by different physiological and genetic factors that could not be addressed in this case report.15,16 Unfortunately, urine and blood samples from the father were only taken 6 months after the incident. Consequently, an increased exposure to mercury can neither be confirmed nor excluded, although urinary levels of Hg were above the reference value.
Mercury-containing creams are known to be responsible for intoxications. However, the published reports likely represent a fraction of the actual cases. Most likely, consumers and physicians do not instantly associate occurring adverse health effects with mercury poisoning and the use of such creams. At first, it was unclear, how the child was poisoned. After the exclusion of other mercury sources, it was evident that the facial cream directly or indirectly caused the intoxication. The ingestion of the cream or mercury-contaminated dust caused by the cream was considered a possible route of exposure. However, the cream was kept away from the children and we excluded a severe mercury intoxication by dust ingestion as the apartment was very clean and only little visible dust formation was noticed. Therefore, the absorption of mercury via skin remained the most plausible cause of exposure. Although mercury in creams is readily absorbed into the skin, 5 residual amounts may remain on the skin surface or other surfaces such as the bed linen for some time. In our opinion, the mercury-containing cream was likely transferred by skin-to-skin contact. According to the mother, she had used the cream on herself before she went to bed. The patient commonly slept in the parent’s bed, implying frequent and enduring skin contact. Depending on the retention time of the cream on the skin, transmittal of mercury could have also occurred during daytime via cuddling. His older brother, who did not sleep in the parent’s bed and spent less time at home due to his school attendance, had probably less skin contact, explaining his lower mercury levels and asymptomatic intoxication.
The temporary presence of the cream on the mother’s skin was confirmed by the analysis of mercury in her hair (Figure 1). Based on our initial hypothesis that the mercury deposition in the hair was caused by systemic mercury exposure, we expected the maximum levels to be around the time the mother has stopped using the cream (day 0). In contrast, mercury accumulated over time. Furthermore, the levels in hair are significantly higher to what has been reported for people with comparable blood mercury levels in artisanal and small-scale gold mining areas.17,18 Therefore, her hair must have absorbed mercury due to contact with the cream, either during application (hairline) or sleep (all hair). The high concentration of mercury in hair, together with skin shedding, may also explain the levels of mercury in dust. We advised the family to regularly vacuum-clean the apartment and avoid any dust formation as the mercury levels in dust remained relatively high.
Conclusion
In summary, this is, to the best of our knowledge, the first case of mercury intoxication due the transmission of a mercury-containing cream by skin-to-skin contact. Furthermore, the cream was advertised as a facial cream and not, as in other case reports, as a skin-lightening cream. Although very little is known about the retention of mercury-containing creams on the skin and the transmission of mercury by skin-to-skin contact, this case clearly demonstrates that this route of exposure may lead to severe mercury intoxications and that the consumers of mercury-containing creams not only threaten their own health, but also, although unknowingly, endanger their families and friends.
Footnotes
Acknowledgements
Blood, urine, cream and dust samples were all analyzed by the Medical Laboratory Bremen (Bremen, Germany, ![]() ) and the Institute and Clinic for Occupational, Social and Environmental Medicine of the University Hospital LMU Munich (Munich, Germany). Mercury monitoring of ambient air was conducted by the Institute and Clinic for Occupational, Social and Environmental Medicine of the University Hospital LMU Munich in Munich, Germany. The authors would like to thank Isak Qorolli and Stefan Gröbmair from the Institute and Clinic for Occupational, Social and Environmental Medicine, University Hospital, LMU Munich, for their support during the analysis of mercury in ambient air, blood, urine and hair and Dr James Rooney for discussing and editing the manuscript with them.
) and the Institute and Clinic for Occupational, Social and Environmental Medicine of the University Hospital LMU Munich (Munich, Germany). Mercury monitoring of ambient air was conducted by the Institute and Clinic for Occupational, Social and Environmental Medicine of the University Hospital LMU Munich in Munich, Germany. The authors would like to thank Isak Qorolli and Stefan Gröbmair from the Institute and Clinic for Occupational, Social and Environmental Medicine, University Hospital, LMU Munich, for their support during the analysis of mercury in ambient air, blood, urine and hair and Dr James Rooney for discussing and editing the manuscript with them.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
