Abstract
Mixed adenoneuroendocrine carcinomas are rare tumors that contain both an exocrine and an endocrine component. Since the latest classification by the World Health Organization and with the aid of immunostaining, more mixed adenoneuroendocrine carcinomas are now identified and diagnosed. Nonetheless, our knowledge of these tumors is still limited, notably concerning gastric variants, as the cases reported in the literature are very limited. The clinical and surgical treatment, including the chemotherapy schemes, the prognosis, and recurrence still represent challenges for the medical teams. We present the case of a 62-year-old woman. After an upper endoscopy revealed multiple polyps and a low-grade neuroendocrine tumor, a D2 radical gastrectomy was performed. A low output esophageal anastomotic leak was discovered in the postoperative period and successfully managed. Pathology revealed a gastric mixed adenoneuroendocrine carcinoma, the first case of this kind reported in Ecuador. Patient is doing well and under constant surveillance up until her 13th postoperative month.
Introduction
In 2010, the World Health Organization (WHO) classification of tumors of the digestive system1,2 introduced the new term “mixed carcinomas” to describe tumors with both an exocrine and neuroendocrine component. 1 These uncommon tumors represent a special type of cancerous tissue, as it is composed of a mixture of two malignant histologically different tumors. 3 Gastric mixed adenoneuroendocrine carcinomas (MANECs) are especially rare, 2 making the current knowledge base surrounding these tumors limited and reliant on a small series of case reports.4,5 Consequently, diagnostic and treatment options are a topic of ongoing research. Furthermore, the outcome and prognosis of such aggressive tumors are worrisome. 1
We present the first case of a gastric MANEC reported in Ecuador.
Case report
The patient is a 62-year-old woman with past medical history of rheumatoid arthritis. She complained of dark red rectal bleeding with no associated symptoms. Clinical examination, including rectal, was unremarkable. Thus, a colonoscopy and an upper endoscopy were requested. Colonoscopy was normal, but the upper endoscopy revealed multiple small 0.5 × 0.3 cm polyps that covered the entire greater curvature of the stomach, from the fundus all the way to the antrum. The results of a histological study performed from a biopsy sample revealed a low-grade gastric neuroendocrine tumor. Complementary exams including complete blood count, urinary 5-hydroxyindoleacetic acid (5-HIAA), abdominal tomography, and an octreotide scan appeared normal. Total radical gastrectomy was decided as the best course of action since the polyp location did not allow a subtotal gastrectomy. On laparotomy, the stomach wall appeared normal. The stomach was not enlarged or had any palpable masses; in addition, no ascites or lymph nodes were recognized. A D2 radical gastrectomy reconstructed with an esophagojejunal roux-en-y was performed, a drain was placed near esophagojejunal anastomosis and the remainder of the procedure was achieved without any complications. The gastric mucosa contained 40 small 0.5 × 0.5 cm polyps, covering the fundus, body, and antrum. A grade II, moderately differentiated MANEC (Figure 1), with muscle and vascular invasion, was detected. Immunostaining revealed cytokeratin 20, CD56, and chromogranin A positivity (Figures 2–4, respectively). The resected lymph nodes did not show any signs of malignancy.
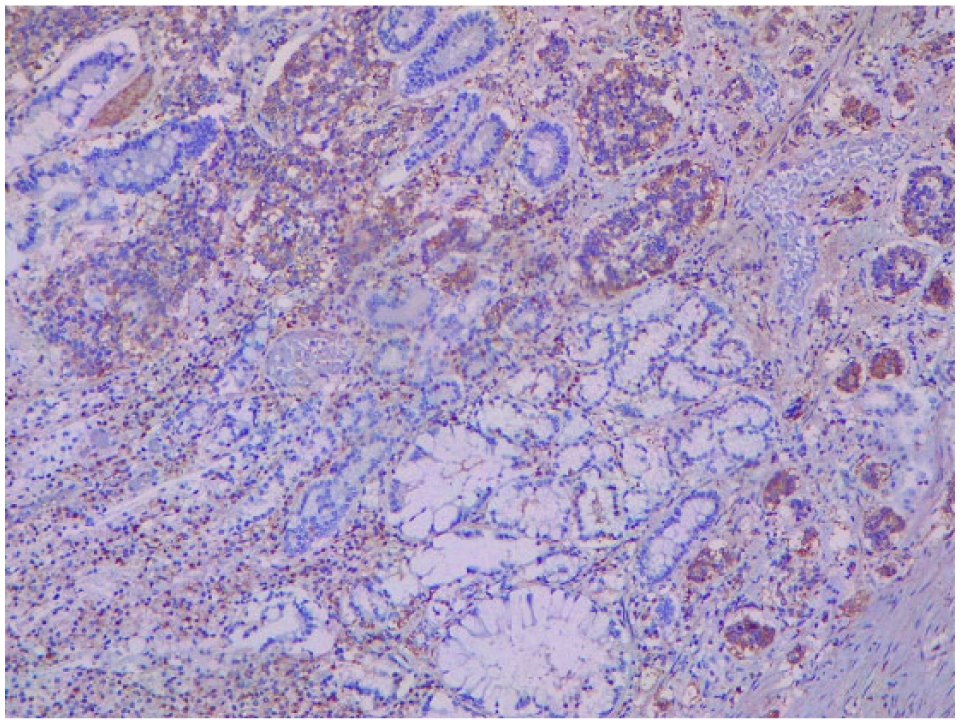
Gastric mucosa with MANEC, atypia in gland cells, and dysplasia in enterochromaffin cells.
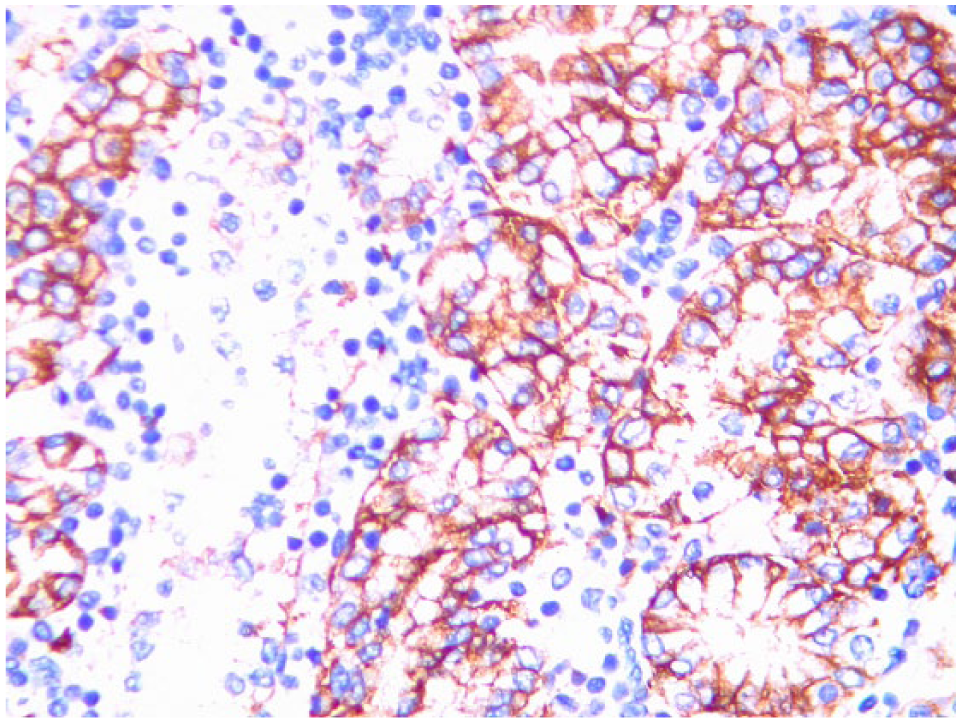
Positive immunostaining for CD56.

Positive immunostaining for cytokeratin 20.

Positive immunostaining for chromogranin 20, within tumor.
During hospitalization, total parental nutrition was removed on the 10th postoperative day; after sips of liquids were initiated and a full diet was instated. However, on the 11th postoperative day, the serous fluid of the drain became purulent. To assess the anastomosis, an esophagogram was performed which ruled out a leak (Figure 5); in addition, a contrast-enhanced abdominal tomography discarded the presence of any abscesses or intra-abdominal collections. Nevertheless, the fluid amylase levels were over the reference range.
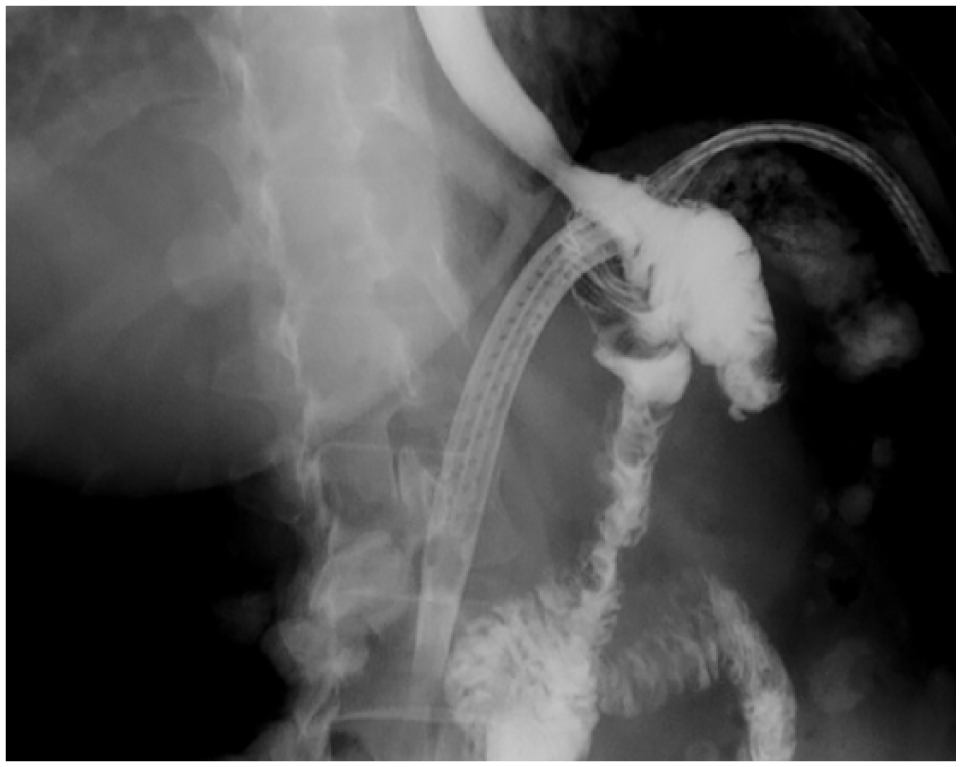
Esophagogram which did not show any leakage or stenosis.
Taking this into account and as the patient remained asymptomatic, tolerating a full diet, and since the output of the drain was low and became serous on the 20th postoperative day, conservative management of the anastomotic fistula was initiated. She was discharged after 25 days and was placed under a close follow-up regimen.
Since the output of the drain persisted for 4 weeks after discharge (Figure 6), an endoscopic intervention with a metal stent was completed to close the fistula (Figure 7). Following this procedure, the patient underwent full recovery.
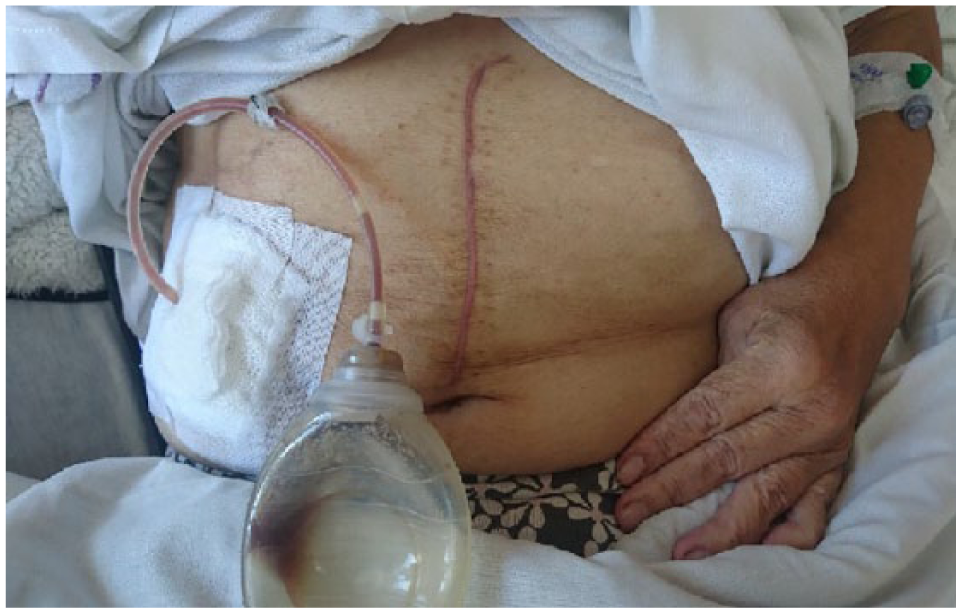
Patient with good clinical condition and fistula.

Metal stent covering the fistula.
Two complete cycles of a combination of etoposide and cisplatin-based chemotherapy were completed; however, before the third cycle could be started, the patient refused any type of treatment mainly due to the adverse effects of chemotherapy. Even after psychotherapy, the patient declined any additional therapy. Eight months after the surgery, at a routine medical check-up, a chest X-ray revealed right basal pulmonary nodules; however, the patient did not allow us to investigate them any further. The patient is in her 13th postoperative month and remains in good condition.
Discussion
Since Cordier et al. published the first description of a gastrointestinal tumor with an exocrine and a neuroendocrine component in 1924, several case reports arose using different names without a consensus. 6 This occurred until the 2010 WHO classification of tumors of the digestive system when the term MANEC appeared. 2 This new category described a mixed neoplasm with a combination of two histologically different tumors: a neuroendocrine carcinoma (NEC) and a gland-forming carcinoma.1,3 An important feature of these tumors is that each component represents at least 30% of the whole lesion. 3 Due to the rarity of this tumor type, few aspects are known about its histogenesis, and many authors agree that they might derive from a multipotent progenitor cell with bidirectional differentiation, opposite to a collision tumor in which the two tumor tissues are believed to originate from independent pathways.3,4 Epidemiological data for MANECs remains inconclusive due to their rare occurrence; however, there are case reports of MANECs occuring in the bowel, pancreas, gallbladder, uterine cervix, and the cecum.2,5 Less than 40 gastric MANEC cases have been reported in the literature.5,7 This is the first reported case of a gastric MANEC in Ecuador, and to our knowledge, no other MANECs have been found in different locations in the country. Clinically, MANECs are considered carcinomas, even though both components are histologically malignant. The tumor behavior is dictated by its most aggressive component, which is usually the neuroendocrine one. 3 The average age of presentation is approximately 60 years,2,4 and males appear to be more prone to developing this type of tumor. Our patient was 62 years at the time of diagnosis. Gastric MANECs usually manifest with nonspecific symptoms, similar to those of conventional gastric cancer. For this reason, most gastric MANECs tend to be detected at an advanced stage. This fact, combined with their aggressive behavior, results in a worse prognosis for most patients. Average survival times are less than 12 months. 2 Our patient presented with rectal bleeding that led to the discovery of a gastric MANEC with vascular and muscular involvement, which suggested a bad prognosis from the start. Macroscopically, MANECs usually appear as polypoid masses or ulcerating stenotic lesions, with a mean size of about 5 cm, 5 as it was described in our case. The identification of the neuroendocrine component within the tumor is difficult, since the rate of immunostaining positivity has been described in 60%–70% for chromogranin, 75%–90% for synaptophysin, and 50% for CD56.4,5 Our case presented immunostaining positivity for all of the aforementioned components. In our case, an NEC was first detected on the greater curvature of the stomach, but the MANEC diagnosis was not achieved until the final pathology analyses; however, it should be noted that a biopsy specimen does not represent the potential heterogeneity of a tumor, and this may explain the low incidence rate of preoperative and postoperative diagnosis, as has been described in previous cases.2,4 Esophageal anastomotic leaks following gastric cancer surgery can be managed successfully and safely with endoscopic therapy, as it is highly effective in achieving healing without the need for surgical repair. 8 For this reason, an endoscopic approach was performed, which successfully sealed the leak in our patient. There is currently no consensus regarding the definitive and optimal treatment of gastric MANECs.4,6 Due to the high risk of metastasis and recurrence rate of MANECs, surgery combined with chemotherapy appears to improve the median survival up to 12 months.1,2,7 Adequate chemotherapy schemes are still undefined; however, since the combination of etoposide and cisplatin has been proved effective in the treatment of small-cell lung cancer, this scheme has been used for MANEC tumors with a response rate of up to 67%.1,7 Alternative regimens that substitute cisplatin or etoposide for irinotecan have also been described.1,7,8 In our case, we could not complete the chemotherapy treatment or investigate her pulmonary nodules, since the patient refused any additional therapy. However, she remained alive nonetheless 13 months after surgery.
Conclusion
Gastric MANECs are a rare kind of tumor without a defined treatment. Most patients, as in our case, are diagnosed at an advanced stage that hinders their survival. More research is needed to fully understand this neoplasm and its proper treatment. We suggest that surgical therapy in combination with adequate chemotherapy can be beneficial for gastric MANECs.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
The authors confirm that the patient has given their informed consent for the case report to be published.
