Abstract
Mixed adenoneuroendocrine carcinomas of the gastrointestinal tract are until today poorly understood and thus very challenging for interdisciplinary therapy. We herewith report the first case series of patients with a primary mixed adenoneuroendocrine carcinoma of the rectum. Both cases were initially diagnosed as adenocarcinoma and only secondarily with mixed adenoneuroendocrine carcinoma and had a poor outcome due to a rapid tumor progression and resistance to chemotherapy. A 65-year-old female presented with local tumor recurrence and hepatopulmonary metastasis 1 year after primary surgery for adenocarcinoma of the rectum and consecutive radiochemotherapy regimen. Fluorouracil (5-FU) was followed by bevacizumab- and capecitabine-based chemotherapy but had to be discontinued due to side effects and progressive disease. Progressive local pain syndrome accompanied by recurrent bleeding episodes led to a local tumor-debulking operation. Afterward, mixed adenoneuroendocrine carcinoma as the underlying diagnosis in the final histopathological examination was detected. The patient died 3 months after the operation in the context of a fulminant tumor progress. A 63-year-old male patient underwent neoadjuvant radiochemotherapy and laparoscopic rectum resection. After 5 months, postoperative oxaliplatin/capecitabine-based adjuvant chemotherapy was switched to carboplatin/etopsid due to a progressive polyneuropathy and biopsy-proven pulmonary metastasis. The patient then had to be switched to local radiation of cerebral metastases and Topotecan due to cerebral bleeding episodes but died 18 months after the initial diagnosis. In conclusion of our case series, mixed adenoneuroendocrine carcinomas of the rectum should be considered as a rare but aggressive tumor entity. An early and detailed histopathological diagnosis is required in order to establish an individual interdisciplinary treatment concept.
Introduction
The first description of a gastrointestinal tumor with a neuroendocrine and an exocrine component was published by Cordier 1 in 1924. As defined by the World Health Organization in 2010, mixed adenoneuroendocrine carcinomas (MANECs) consist of at least 30% of each neuroendocrine and adenocarcinoma tumor cells. Until today, MANECs are poorly understood and thus remain especially challenging for interdisciplinary therapy. Previous case studies have reported MANECs of the esophagus, stomach, ampulla vateri, and colon.2–4 Reports of the characteristics of occurrence, treatment concepts, or outcomes for MANECs of the rectum, however, remain scarce. To the best of our knowledge, we herewith report the first case series of two patients with a MANEC in the rectum with main focus on the interdisciplinary treatment and the further clinical course.
Case reports of two patients
Case report
We herewith present the case of a 65-year-old female patient with a history of 5-FU neoadjuvant radiochemotherapy and consecutive rectum resection with simultaneous atypical liver resection (Segment II) for hepatic metastasized rectal carcinoma (ypT3, pN1 (2/21), pM1, L0, V0, Pn0, R0). Initial surgical therapy was followed by 9 months of adjuvant chemotherapy based on folinic acid, fluorouracil, and oxaliplatin (FOLFOX). The patient was then presented to our tertiary referral center 1 month after the end of adjuvant chemotherapy with a suspected local tumor recurrence accompanied by progressive perianal pain and perianal hemorrhage. Computed tomography (CT) staging also revealed bilateral pulmonary metastases as a new diagnosis. In accordance with our interdisciplinary tumor board, a palliative cetuximab-, irinotecan-, and 5-FU-based chemotherapy was begun but had to be discontinued due to toxic dermatological side effects after completion of the first chemotherapy cycle.
After a chemotherapy-free episode of 1 month, CT restaging revealed a low progression of bipulmonary metastases. A new chemotherapy regimen consisting of bevacizumab and capecitabine was thus begun.
After an uneventful course of 3 months of chemotherapy of this kind, CT examination was repeated and revealed a fulminant local tumor progression as well as progressive pulmonary metastases, new hepatic metastases, and pelvic lymphadenopathy.
Due to a progressive local pain syndrome accompanied by recurrent bleeding episodes we decide to perform a local tumor-debulking operation by means of a Hartmann procedure which revealed for the first time MANEC as the underlying diagnosis in the histopathological examination of the final specimen. After an initially uneventful postoperative course, the patient died 3 months after the debulking operation due to septic multi-organ failure in the context of a fulminant tumor progression.
Histopathological analysis
The final histopathological examination of the debulking operation specimen revealed rpT4 bpN2b (13/16) G3 R1 L1 VI poly-metastasized carcinoma of the rectum and high-grade malignant MANEC. Both components of the carcinoma were represented with 30% (Figure 1).
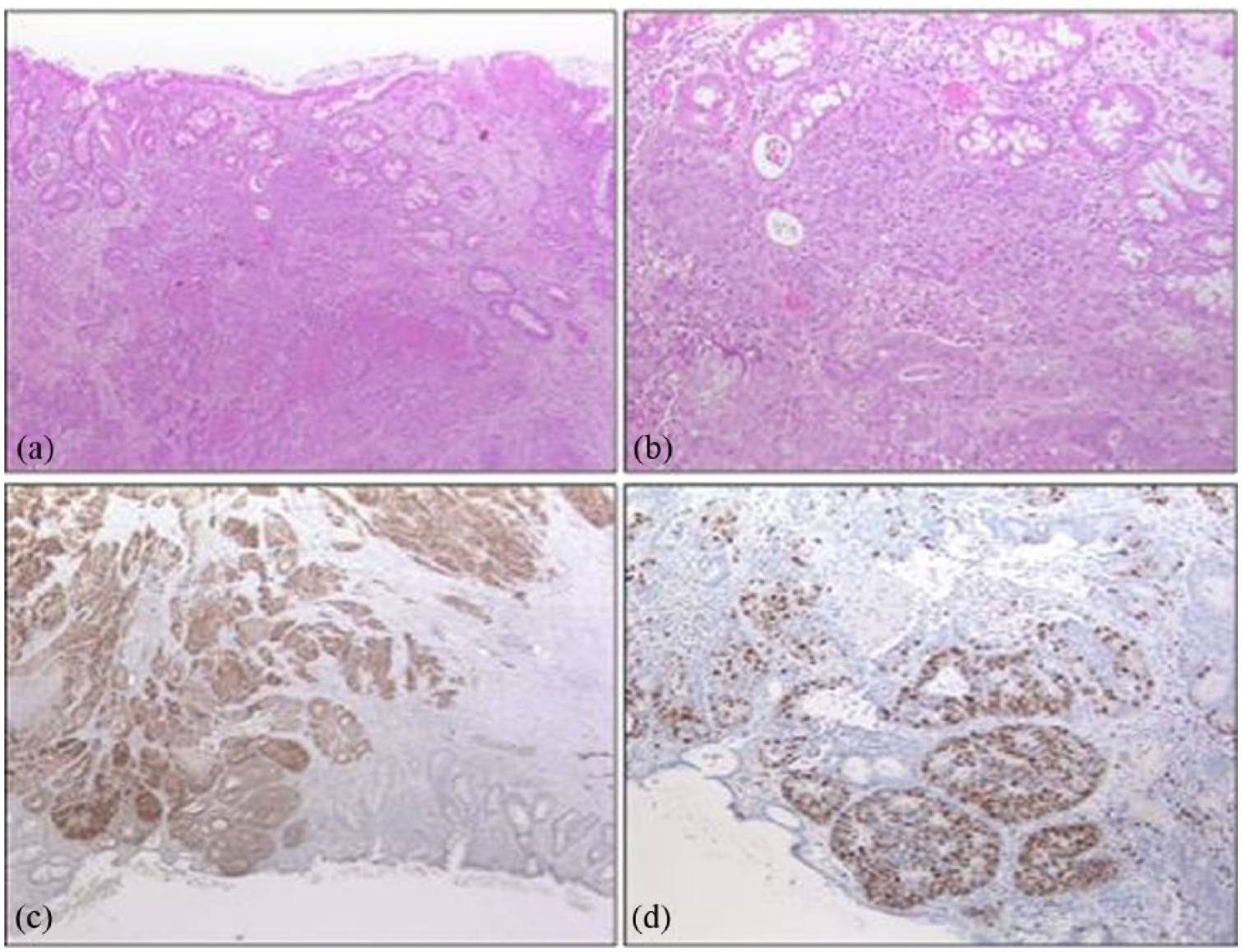
Hematoxylin and eosin (HE) stains and immunohistochemistry (IHC) of a pure neuroendocrine carcinoma (NEC). (a) Low-power view of the NEC centered in the submucosa. (b) Medium-power view of (a) reveals relatively monomorphic cells with stippled (“salt and pepper”) chromatin. Mitotic figures and necrosis are common. (c) NEC cells show strong cytoplasmic immunoreactivity for synaptophysin, while non-neoplastic glands stain negative. (d) Ki-67 IHC reveals a high Ki-67 index (70%), indicating a highly proliferative tumor.
Case report
The second patient was a 63-year-old male who underwent a combined laparoscopic and transanal total mesorectal excision (taTME) after neoadjuvant radiochemotherapy for suspected adenocarcinoma of the rectum. Histopathological examination of the operation specimen again revealed a high-grade malignant MANEC of the rectum. In accordance with our multidisciplinary tumor board, postoperative oxaliplatin/capecitabine-based adjuvant chemotherapy was initiated 4 weeks after the operation.
After 5 months of uneventful adjuvant chemotherapy, CT restaging was highly suspicious of pulmonary and hepatic metastases. A CT-guided pulmonary biopsy was taken and confirmed pulmonary metastases of a MANEC. Due to tumor progression as well as an additional progressive peripheral neuropathy, chemotherapy regimen was switched from oxaliplatin/capecitabine to carboplatin/etopsid. Three cycles of carboplatin and etopsid were administered. The patient then, however, developed cerebral bleeding, and palliative therapy was thus switched to local radiation of cerebral metastases and Topotecan. The patient died 12 months after the operation due to septic respiratory insufficiency.
Histopathological analysis
TNM classification showed a ypT3 pN1a (1/17) M0 R0 L1 V1 as high-grade malignant MANEC. Wild-type KRAS-Gen.
Pulmonary metastasis showed neuroendocrine components of a carcinoma with a MANEC of the rectum. Proliferative activity 70%–80%, positive for synaptophysin, negative for cytokeratin 20, CDX2, and TTF-1 (Figure 2).

Hematoxylin and eosin (HE) stains and immunohistochemistry (IHC) of a mixed adenoneuroendocrine carcinoma (MANEC). (a) HE stains reveal a classic adenocarcinoma component with glandular growth pattern as well as a surrounding neuroendocrine component. (b and c) Using synaptophysin IHC, the neuroendocrine proportion shows strong cytoplasmic staining, while the glands of the adenocarcinoma component stain negative (asterisk). (d) Ki-67 IHC of the neuroendocrine component shows a high Ki-67 index (>90%).
In both patients, staging and restaging examinations were performed in accordance with our national guidelines and included positron emission tomography (PET)–CT scans of the thorax and abdomen, as well as laboratory controls of relevant parameters such as carcinoembryonic antigen (CEA). Relevant findings of the staging examinations are described within the case report for each patient.
Written informed consent for surgery as well as for the use and publication of clinical data were obtained from both patients, as required by our institutional review board.
Discussion
Gastrointestinal tumors which consist of both—an exocrine and a neuroendocrine component—were first described by Cardier 1 in 1924 in “Les cellules argentaffines dans les tumeurs intestinales.” In the following, a different mix of carcinomas described as amphicrine tumors, mixed exocrine–endocrine tumors, and combined and collision tumors were reported and summarized as MANECs.5,6 It took 60 years, however, for a first systemic classification of mixed adenocarcinomas and neuroendocrine tumors by Lewin 7 who differentiated composite/collision neoplasms (neuroendocrine and exocrine components occur in separate areas of the same lesion), combined neoplasms (mixed neuroendocrine and exocrine components), and amphicrine neoplasms (neuroendocrine and exocrine components are present in the same neoplastic cell, which shows a divergent immunophenotype). In 2000, the WHO then defined mixed exocrine–endocrine tumors as consisting of both at least 30% of neuroendocrine and a non-neuroendocrine glandular (adenocarcinomatous) component.8,9 Subsequently, the WHO Classification of Tumors of the Digestive System 2010 introduced the term MANECs which differentiates between adenoma–neuroendocrine tumor (NET), adenoma–neuroendocrine carcinoma (NEC), adenocarcinoma–NET, and adenocarcinoma–NEC depending on the histological appearance of each component.6,10,11 Capella et al. 12 also reported that MANECs may be grouped into different prognostic categories based on the grade of malignancy of each component:
The mixed adenoneuroendocrine neoplasms (MANEC) include three entities: high-grade malignant mixed adenoma/adenocarcinoma–NEC, intermediate grade malignant mixed adenocarcinoma-G1/G2 NET, and amphicrine carcinoma.
The mixed adenoneuroendocrine tumor (MANET) includes three entities: low-grade malignant adenoma–NET, NEC (poorly differentiated neuroendocrine carcinoma), and neuroendocrine tumor (NET). 7
Until now, the exact pathogenesis of MANEC remains unclear. Both a neuroendocrine development out of dedifferentiated adenocarcinomas and a bidirectional differentiation out of multipotential stem cells are being discussed.3,13,14 Vanacker et al. 15 found an identical KRAS mutation as well as analogous somatic mutations in the exome sequencing in both components of MANEC which may support a clonal pathogenic relationship. Also mutations during chromothripsis or inactivation of SMARCA 4 have been reported.16–18
Several origins of gastrointestinal MANECs such as the colon, appendix, or stomach and biliary mixed MANECs of the ampullary region have been described.4,6,19,20 To the best of our knowledge, this is however the first case series of patients with a primary MANEC of the rectum.
An important factor for improving the outcome of MANEC is early and exact histopathological examination. As an example, in our first patient, the diagnosis of MANEC was only confirmed after exact examination of the recurrence specimen 15 months after the initial primary surgery. The importance of immunohistochemistry in the routine diagnostic surgical pathology has been proven in this context.6,21 It remains unclear whether MANECs are driven by the glandular or neuroen- docrine tumor component but they tend to show a high potential for early distant metastasis which needs to be taken into consideration during the primary and further clinical diagnostic pathway in these patients.22–24 Overall prognosis of patients with gastrointestinal MANEC appears to be poor with the reported median survival rates of 7–10 months after initial diagnosis.25,26
Ilett et al. 27 recommended the use of somatostatin receptor scintigraphy (SRI) for diagnosis as well as follow-up, whereas Kadhim et al. 28 reported peptide radionuclide receptor therapy as the second- or third-line therapy in patients with strong somatostatin receptor uptake. La Rosa et al. 29 also recently reported no survival differences between patients with either colorectal NECs or colorectal MANECs.
However, large and/or randomized case series on the treatment and prognosis of MANEC until today remain scarce.
An important step in the improvement of overall outcome in patients with MANEC is an early referral of patients to a high-volume center with a potential multidisciplinary expertise in the diagnosis and treatment of MANEC. The clinical course often appears to be driven by the neuroendocrine tumor component. However, there is a general agreement that the therapy management should be focused on the more aggressive tumor component as defined by the histopathological criteria. Accordingly, MANECs with poorly differentiated neuroendocrine components should be treated like NECs, while MANECs with well-differentiated neuroendocrine components but a rather aggressive glandular tumor appearance should be treated as common colorectal adenocarcinomas.5,30
The National Comprehensive Cancer Network recommends the use of cisplatin or carboplatin with etopsid 31 in such patients in accordance with the “European and North American guidelines for neuroendocrine neoplasms” and also the “Nordic guidelines for neuroendocrine neoplasms in 2014” recommend cisplatin or carboplatin and etopsid.32,33
This first case series of patients with a MANEC in the rectum of course only includes the treatment and clinical course of two patients. It however underlines the aggressiveness of this rather rare tumor entity. An early and detailed histopathological diagnosis is required in order to establish a personalized multidisciplinary treatment concept. Increased awareness in the diagnosis and treatment of MANEC and the uptake of these cases in registers may lead to an accumulation of larger case series and thus hopefully further relevant evidence for treatment options and overall prognosis in patients with gastrointestinal MANEC.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent for surgery as well as for the use and publication of clinical data were obtained from both patients, as required by our institutional review board.
