Abstract
Stent fracture is a rare complication of drug-eluting stent implantation with a reported rate of 0.84%–3.2% in various clinical studies with first-generation drug-eluting stents and 29% in autopsy studies. Sirolimus-eluting stents with their closed cell design were reported to be more prone to fracture compared to paclitaxel-eluting stents. Other risk factors for stent fracture are multiple stenting, longer stent length, chronic renal failure, right coronary artery intervention, and a higher maximal inflation pressure. The role of angiography in diagnosing stent fracture is limited, a fact also questioning the reliability of angiographic data. Image enhancement techniques like StentBoost are widely available in new-generation angiography systems and are used to assess stent expansion, overlap size, or to localize the postdilation balloon. Here, we report a case of zotarolimus-eluting stent fracture at initial implantation diagnosed with StentBoost.
Keywords
Stent fracture (SF) is a rare complication of drug-eluting stent (DES) implantation with a reported rate of 0.84%–3.2% in various clinical studies with first-generation DES1–3 and 29% in autopsy studies. 4 Review of the literature shows that there is increasing concern about SF as a potential cause of stent restenosis and thrombosis.5–7 This can lead to adverse clinical outcomes (recurrent angina, myocardial infarction, and even sudden death). Sirolimus-eluting stents (SES) with their closed-cell design were reported to be more prone to fracture compared to paclitaxel-eluting stents (PES).5–9 Other risk factors for SF are multiple stenting, longer stent length, chronic renal failure, right coronary artery (RCA) intervention, and a higher maximal inflation pressure. 5 Newer generation stents with thinner struts are theoretically more prone to complications such as fracture and longitudinal compression. 7 Endeavor Resolute Zotarolimus-Eluting Coronary Stent System (Medtronic, Minneapolis, MN, USA) uses a cobalt chromium stent platform; a durable antithrombotic, phosphorylcholine-encapsulated coating; and another G1 cell-cycle inhibitor, zotarolimus. Here, we report a case of zotarolimus-eluting stent (ZES) fracture at initial implantation diagnosed with StentBoost.
Case report
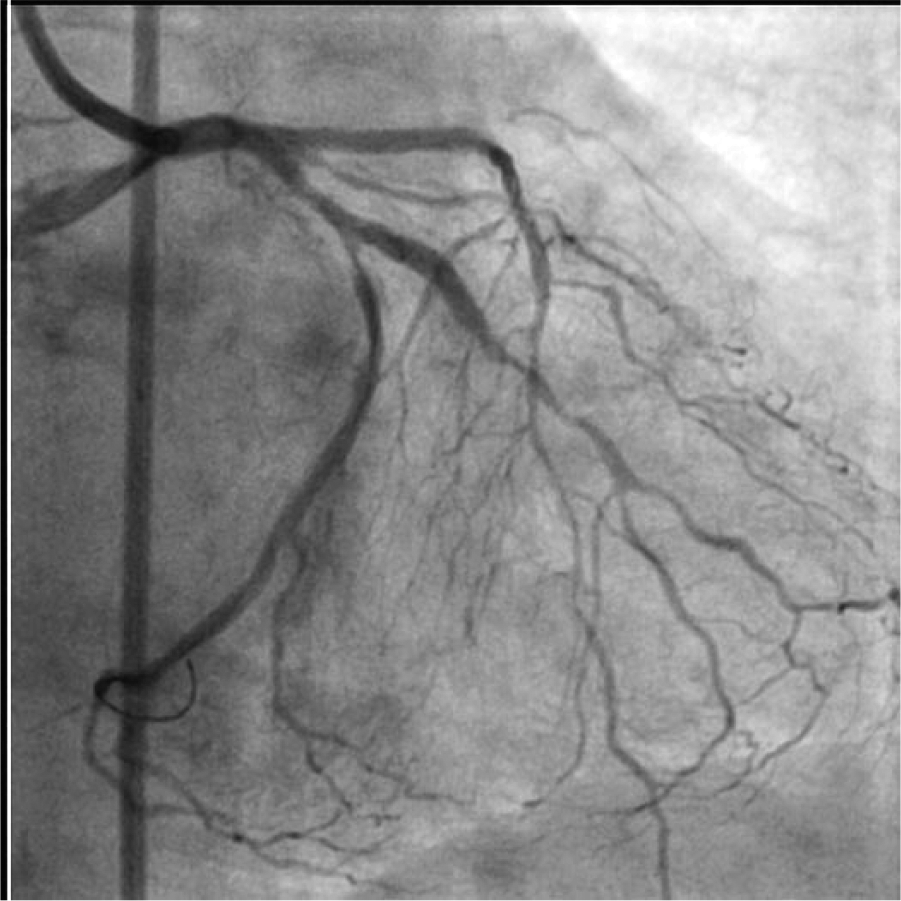
A 58-year-old diabetic woman was admitted to emergency department with unstable angina. After initial medication with acetylsalicylic acid, clopidogrel, enoxaparin, atorvastatin, ramipril, and pantoprazole, she underwent coronary angiography which revealed 90% stenosis at circumflex coronary artery and obtuse marginal branch of circumflex coronary artery (CX-OM) bifurcation (Medina 1-1-1) (Figure 1). A 7 F Extra BackUp guiding catheter was used to engage the left main coronary artery. Both the main branch and the side branch were wired. The obtuse marginal branch (OM) was dilated with a 2.0 × 20-mm balloon dilatation catheter (Simpass; Simeks, Istanbul, Turkey) at 16 atm, and the circumflex coronary artery (Cx) body was dilated with a 2.5 × 20-mm balloon dilatation catheter (Simpass; Simeks) at 18 atm. Thereafter, while parking a balloon in the main branch, a 2.5 × 18-mm stent (Endeavor Resolute; Medtronic) was deployed in the side branch at 12 atm and postdilated with a 2.5 × 12-mm noncompliant (NC) balloon (Simpass; Simeks) at 20 atm. Subsequently deploying the main branch balloon, a mini crush was performed and another 2.5 × 30-mm stent (Endeavor Resolute; Medtronic) was implanted at 20 atm into the Cx body. The OM was rewired and final kissing was performed with NC balloons. Thereafter, localized haziness suggesting thrombosis at first sight appeared in the main-branch stent, both in the proximal part and distal to the OM take off (Figure 2). The patient had no symptom and was hemodynamically stable. StentBoost showed stent fracture and disruption (Figure 3). Another Endeavor Resolute stent was implanted at 14 atm to cover the fracture sites. The final result was satisfactory (Figure 4).

Coronary angiography revealed a Medina 1-1-1 obtuse marginal branch of circumflex coronary artery (CX-OM) bifurcation lesion.
The arrows showing localized haziness appeared in the main branch stent both in the proximal part and distal to the OM take off.

StentBoost shows type V stent fracture (SF).

Endeavor Resolute stent was implanted at 14 atm. The final result was good.
Discussion
Coronary SF was first reported in 2002 after a bare-metal stent (BMS) implantation in a venous bypass graft. 10 The first case of coronary DES fracture was described in 2004, after which several cases were reported. 11 The reported incidence of SF is from retrospective angiographic series.5,6 There are no prospective data. The few case reports are all of patients with restenosis indicating that SF may be the mechanism in selected cases with restenosis. Several mechanisms such as mechanical stress (shear stress, high inflation pressure) and risk factors (long stent, bifurcation stenting etc.) are mentioned to explain SF. 5 Metal fatigue is another proposed mechanism. The retrospective nature of the available data does not allow precise timing of the fracture, but as demonstrated in this case the possibility of fracture at initial implantation is not to overlook. SF was identified in 23.9% of cases of sirolimus-eluting stent-associated restenosis and in 9% of cases of restenosis of PES. 12 There are only a few case reports on ZES fracture in the literature. Venero et al. 13 reported a ZES fracture in an aortocoronary saphenous vein bypass graft (SVG). Recently, Lee et al. 14 described two cases of ZES fracture: one was detected with intravascular ultrasound (IVUS) and the other with conventional coronary angiography.
The role of angiography in diagnosing SF is limited, a fact also questioning the reliability of angiographic data. IVUS is good at assessing stent expansion, malapposition, but has also limitations when assessing SF. Optical coherence tomography (OCT) can also be used to detect SF. Absence of stent struts was the most common morphological feature of SF in OCT. The advantage of OCT over IVUS is that it has better resolution and fewer artifacts. Multi-detector computed tomography (MDCT) also enables the early detection of SF. Standard angiography and IVUS have limited ability to visualize stent struts and their integrity over time. While it is thought that SF is associated with high incidence of restenosis or thrombosis, the true incidence of SF is difficult to ascertain with the above-mentioned current imaging modalities. New methods of digital subtraction imaging in the catheterization lab have been developed to enhance the details of stents at the time of implantation. One such method summates imaging frames around fixed marker bands on balloon catheters which deliver the stents (StentBoost Subtract; Philips Healthcare, Best, the Netherlands). Image enhancement techniques like StentBoost are widely available in new-generation angiography systems and can be easily used to assess stent expansion, overlap size, or to localize the postdilation balloon, but was rarely used until recently. Kim et al. 15 reported a case example of the clinical use of an image enhancement system to confirm the diagnosis of a type IV SF despite lack of IVUS evidence of SF. Rossi et al. 16 reported an intra-aortic coronary SF revealed by StentBoost imaging and confirmed by MDCT.
As demonstrated in our case, image enhancement can be very useful in differential diagnosis of in stent haziness. At first sight, thrombus formation and plaque prolapsus were considered as possible underlying mechanisms, but image enhancement with StentBoost showed a type V SF (Figure 2). The haziness was probably caused by plaque prolapsus/shift at fracture, disruption sites. In our case, aggressive postdilatation with NC balloon after stent implantation or the thin strut structure of the stent may be the cause of the fracture. In the era of newer generation stents with thinner struts, image enhancement should be used routinely to assess stent expansion and possible complications like fracture or longitudinal compression especially when performing complex interventions with multiple stenting.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
