Abstract
Chronic, non-healing venous ulcers of the lower extremity are often limb-threatening conditions. Their management is characterized by a prolonged and frequently frustrating clinical course that represents an economic burden to both the patient and healthcare system. During the last two decades, thermal ablation of underlying incompetent venous systems has been extensively utilized to treat chronic venous insufficiency. Despite successful correction of venous hypertension, a substantial subgroup of patients remain affected by non-healing venous ulcers, thus posing a significant clinical challenge. In this case report, we detail quantitative and qualitative wound treatment course in a patient refractory to standard interventions, by treatment with a combination of cell-free amniotic fluid and dehydrated amniotic membrane following successful thermal ablation of refluxing veins.
Introduction
Chronic venous insufficiency (CVI) affects an estimated 25% of women and 15% of men 1 with approximately 1%–4% of the US adult population affected by advanced stages of the disease, including chronic, non-healing venous ulcers (class C6; Clinical Severity, Etiology or Cause, Anatomy, Pathophysiology (CEAP) classification). 2 – 8 This advanced stage of disease significantly reduces patients’ quality of life (QoL) and imposes significant economic burden via direct healthcare costs and decreased productivity,9,10 with an estimated annual cost for the management of venous ulcers between $1.9 and $2.5 billion in the United States alone.11,12 This cost will likely rise with increased life expectancy and an aging population. 12
Managing the advanced stages of CVI with non-healing venous leg ulcers (VLUs) can be a complex and frustrating clinical course.13,14 This is due, in large part, to the paucity of treatment modalities used concurrently with or immediately following the correction of the pathophysiological hemodynamics of the underling venous reflux disease. Consequently, a number of these patients are unsuccessfully treated and viability of the affected extremity may be jeopardized. Thus, there is an unmet need for the treatment of non-healing VLUs that do not respond to current therapies.
Relatively novel agents in regenerative medicine have led to improvement in the management of chronic wounds. Recent clinical results using placental tissues and fluids to treat diabetic foot ulcers (DFUs) have shown promise, 15 indicating these placental biologics are a safe and effective alternative for the treatment of chronic wounds. However, to our knowledge, the safety and efficacy of combinatorial therapy using both amniotic membrane (AM) and cell-free amniotic fluid (AF) have yet to be evaluated in the management of CVI-C6 patients. Here we present a case of chronic, non-healing VLUs treated effectively with cell-free AF and AM, following endovenous thermal ablation (ETA). In this patient, ETA treatment with radiofrequency ablation (RFA) coupled with standard-of-care compression therapy was ineffective. However, following subcutaneous AF injections combined with topical AM, substantial wound healing and wound closure was achieved.
Case report
Patient history
A 65-year-old male with CVI (C6; CEAP Classification)3,6 presented to the Wound Care service following a 3-year history of persistent medial and lateral non-healing VLUs affecting the left leg. The patient’s past medical history was significant for left hip replacement, avascular necrosis of left hip, osteoarthritis, peripheral neuropathy, degenerative disk disease, hypertension, benign prostatic hyperplasia, gastro-esophageal reflux disease, and alcohol abuse. There was no history of previous deep vein thrombosis (DVT), diabetes mellitus (DM), peripheral arterial disease (PAD), and/or hypercoagulability disorders. Family history for CVI and/or DVT was negative.
Treatment was initiated 3 years prior to presenting to the Wound Care clinic and consisted of RFA for correction of superficial reflux disease affecting the great saphenous vein (GSV) and small saphenous vein (SSV), using standard RFA protocols. 12 The patient underwent successful GSV (17 cm segment) RFA occlusion below the knee followed by 1% polidocanol (2 mL) foam sclerotherapy of the wound bed, 21 stab phlebectomies, and liquid 1% polidocanol sclerotherapy for associated varicosities approximately 5 months prior to presenting to the Wound Care clinic. At that time, venous duplex ultrasonography of the left lower extremity was performed. No evidence was found of acute DVT and/or superficial vein thrombosis or of valvular incompetence (Table 1). The left common and external iliac veins and their tributaries were patent and there was no evidence of acute DVT (Table 2). Post-procedural duplex evaluation and venous mapping of the left lower extremity showed successful correction of the underlying venous reflux. Other previous treatments of the patient’s lower limb wounds consisted of an Unna boot and silver-based dressing, as well as multiple surgical debridements under local anesthesia. Despite correction of superficial venous hemodynamics, the VLUs failed to improve. At the time of presentation, intermittent drainage was noted in the left lower medial VLU and lateral perimalleolar region VLU. The wound biopsy (performed at two wound sites) showed no evidence of malignancy.
Venous duplex ultrasound results from measurements on lower left limb.
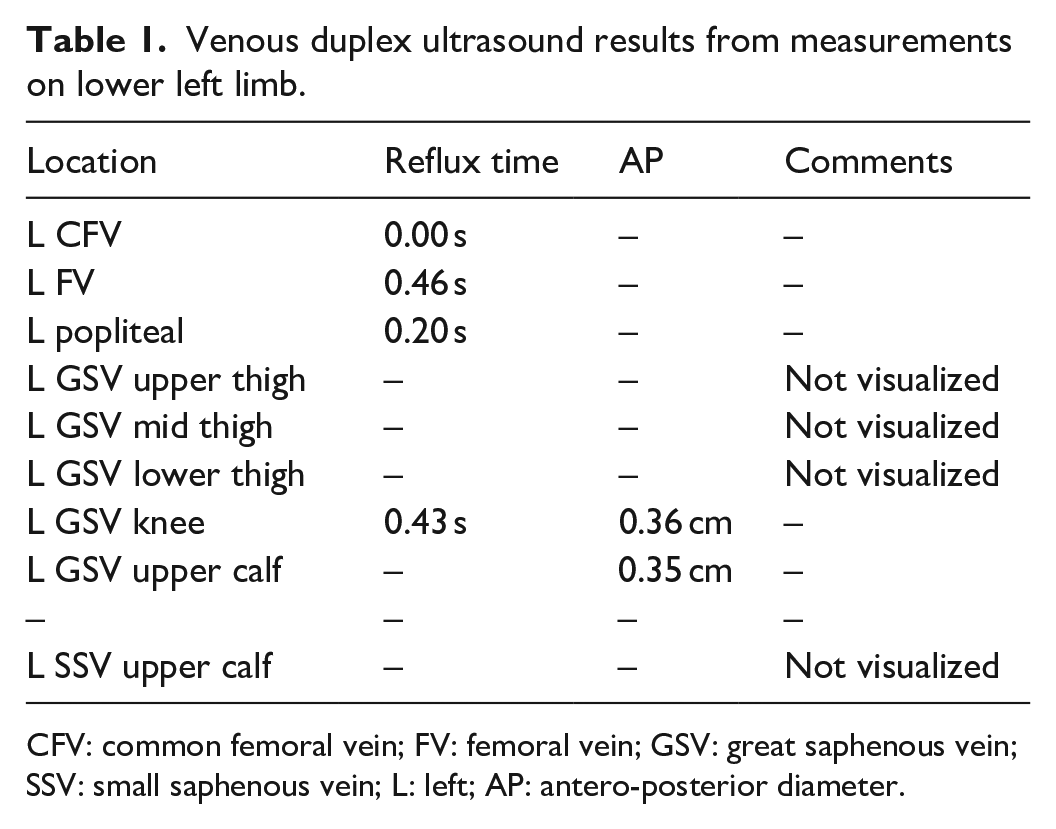
CFV: common femoral vein; FV: femoral vein; GSV: great saphenous vein; SSV: small saphenous vein; L: left; AP: antero-posterior diameter.
Venous Doppler ultrasound results from measurement on lower left limb.

CIV: common iliac vein; EIV: external iliac vein; CFV: common femoral vein; SFJ: sapheno-femoral junction; L: left; PSV: peak systolic blood flow velocity.
Cell-free and sterile-filtered AF, Dermacyte® Amniotic Wound Care Liquid (Dermacyte® Liquid) and dehydrated AM (Dermacyte® Matrix) (both from Merakris Therapeutics Inc., Research Triangle, NC, USA) were used for biologic therapy. The treatment course lasted for 10 weeks, with a follow-up at 17 weeks (Figure 1(a)). Both medial and lateral wounds were assessed qualitatively and quantitatively 4 weeks prior to initial treatment (Figure 1(b)–(e)). Wound measurement was performed in-office by measuring the maximum wound dimensions for length, width, and depth and further analyzed via ImageJ (available at https://imagej.nih.gov).

Treatment approach/follow-up timeline and results for CVI class C6 ((CEAP) classification) patient using AF and AM. (a) Treatment approach and timeline (FU = follow-up). (b) Qualitative analysis and images for medial wound. (c) Quantitative analysis of medial wound dimensions over treatment time course showing maximum measured values of length, width, and depth of wound in centimeters (note: medial wound closure observed at 4 weeks; no data (ND) recorded for two last follow-ups). (d) Qualitative analysis and images for lateral wound. (e) Quantitative analysis of lateral wound dimensions over treatment time course showing maximum measured values of length, width, and depth of wound in centimeters. (f) Qualitative image of lateral wound at week 17 follow-up visit.
A total of three treatment sessions were performed over 10 weeks and a final observation was conducted at week 17 (Figure 1(a)). When the patient arrived for the first treatment, both the medial (Figure 1(b) and (c)) and lateral (Figure 1(d)) wounds were non-healed despite successful RFA. During the first treatment (Figure 1(b) and (d) “week 0”), two 2 mL vials of Dermacyte® Liquid were diluted 1:1 (8 mL total volume) and injected around the wound bed periphery of the lateral VLU and one 2 mL vial (4 mL total diluted solution) was used for the medial VLU. Peripheral areas that showed signs of healing were avoided during injection to mitigate the risk of tissue damage. Dermacyte® Matrix was then applied topically to each VLU and secured with compression. After completion of therapy, sterile and compression dressings were applied from heel to calf. Interestingly, closure of the medial wound was observed after 4 weeks and its treatment was discontinued (Figure 1(b) and (c)). The lateral wound also showed qualitative (Figure 1(d)) and quantitative improvement during the 4-week time (Figure 1(e)).
During the second and third treatment sessions, the lateral wound was treated with a 4 mL Dermacyte® dilution as described above and then covered with Dermacyte® Matrix. Additional reduction in wound size was observed at weeks 8 and 10 (Figure 1(d) and (e)). At the 17-week follow-up, the patient demonstrated almost complete wound closure with significant areas of re-keratinization (Figure 1(f)). There were no adverse events or side effects reported. A 6-month follow-up visit showed no evidence of ulcer recurrence of the medial or lateral wound, and again no side effects were reported. Based on this preliminary observation, Dermacyte® Liquid and Matrix used in combination were a safe and effective treatment for chronic wounds caused by CVI in this patient.
Discussion
Chronic, non-healing VLUs of the lower extremity are frequently limb-threatening conditions.8,11,13,14,16 Their management is often characterized by a lengthy and often frustrating course for the patient and physician, with primary goals for treatment being rapid healing and the prevention of recurrence.17,18 In this patient’s case, clinical exclusion of PAD, DM, hypercoagulability, trauma, and malignancy combined with ultrasonographic and clinical evidence of long standing CVI indicated that venous reflux disease was the underlying etiology of ulcerations. While CVI can often be corrected surgically, many patients develop chronic wounds resistant to standard-of-care treatment. In this patient, placental biologics effectively initiated and accelerated wound closure after standard-of-care treatments failed.
Analysis of AF and amnion components indicates the presence of immune-modulating, anti-inflammatory, and pro-growth molecules. 19 These molecules are derived, at least in part, via secretions from fetal and placental mesenchymal stem/stromal cells (MSCs) and amniotic epithelial cells (AECs). 20 Their presence and supporting clinical evidence indicate amnion components can support the treatment of chronic wounds. 20 We postulate that AF and AM efficiently induced rapid progression through three phases of wound healing in chronic wounds that do not otherwise heal. These consist of (1) an “activation” state in which immune cells are recruited to the site and epithelial-to-mesenchymal transition (EMT) is induced, (2) a “transition” state in which immune activation/inflammation and EMT are reduced and tissue remodeling is initiated, and, finally, (3) a “late healing” state in which a mesenchymal-to-epithelial transition (MET) occurs, concomitant with re-epithelialization and re-keratinization to induce final closure of the wound.21,22 Further research is required to elucidate the precise mechanisms through which amnion components can modulate or enhance the progression through these stages.
Conclusion
This case report suggests that AF and AM are effective tools in the treatment of non-healing wounds that persist despite successful ETA of superficial venous reflux disease, especially in CVI-C6 patients without co-existing PAD, DM, hypercoagulability, tumors, and/or trauma. In this patient, correction of underlying venous reflux with RFA and standard-of-care wound treatment was insufficient to drive effective wound healing as evidenced by 3-year persistent wound status. Despite persistent status, subcutaneous AF injections combined with topical AM safely and effectively promoted wound closure without side effects. Although these data are promising, further clinical studies are needed to fully assess this treatment modality to properly inform clinicians on expectations for patients with CVI-induced chronic wounds.
Footnotes
Acknowledgements
We thank Dr. Jovan Markovic (Duke University) for assistance with drafting the case report.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: T.C.B. and W.S.F. are co-founders of and shareholders in Merakris Therapeutics. The products used to treat the patient in this case report were Dermacyte® Liquid and Dermacyte® Matrix, and were obtained from Merakris Therapeutics.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series. Pursuant to the Federal Regulations and UTMB IRB Policies and Procedures, case reports as defined below do not require IRB approval. Use medical information collected from a clinical activity rather than a research activity and presented on no more than three patients. Case reports are generally done by retrospective review of the medical record and highlights a unique treatment, case or outcome. The examination of the case is usually not systematic and there is usually no data analysis or testing of a hypothesis. Investigators must ensure that the HIPAA privacy rules are followed with respect to using or accessing PHI (a HIPAA authorization or waiver may be required). Study of a single patient/subject would not yield generalizable knowledge and therefore not meet the definition of research outlined at 45 CFR 46.102(l).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We thank our funding sources including the Sealy-Smith Endowment and NIH KL2TR001441-07 to W.S.F.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
