Abstract
Objectives:
Anatomical lobectomy has always been the standard operative treatment of early-stage non-small cell lung cancer. However, there have been emerging evidences suggesting that a subanatomical resection, such as segmentectomy, may yield the same treatment results, even in patients with higher-stage non-small cell lung cancer. This study aimed to compare overall 5-year survival rate and disease-free survival between lobectomy and segmentectomy in patients with non-small cell lung cancer.
Methods:
The retrospective study included 380 patients who underwent surgery for non-small cell lung cancer at Ramathibodi Hospital between 1st January 2016 and 31st December 2020. Of 380 patients, 307 patients underwent lobectomy, while the other 73 patients underwent segmentectomy. Operative, admission, and follow-up data were collected from electronic medical records. Missing data were collected by telephone calls to patients or their relatives in deceased cases. Overall and disease-free survival were analyzed.
Results:
Median overall 5-year survival time after lobectomy and segmentectomy seemed to be different but not statistically significant (18.5 months versus 5.8 months, p = 0.127). Median disease-free survival time after lobectomy and segmentectomy was also similar (8.6 months versus 4.5 months, p = 0.511). Two deaths occurred during perioperative period, one from lobectomy group due to acute massive pulmonary embolism (0.3%) and the other from segmentectomy group due to acute exacerbation of chronic obstructive pulmonary disease with respiratory failure (1.4%).
Conclusion:
Lobectomy and segmentectomy result in similar overall 5-year survival rate and disease-free survival between these two comparison groups. Therefore, segmentectomy may be a potential alternative for operative treatment of non-small cell lung cancer. However, a larger and randomized-controlled trial may be needed to further validate these results.
Introduction
Contemporary muscle-sparing treatment for early-stage non-small cell lung cancer (NSCLC) has always been an anatomical lobectomy with mediastinal lymph node staging. According to a trial performed by lung cancer study group (LCSG) in 1995, 1 the only randomized-controlled trial comparing lobectomy and sublobar resection (segmentectomy and wedge resection), lobectomy yielded better disease-free and overall survival. However, there were emerging reports regarding sublobar resection which yielded similar results.2–6 Even that said, some authors still supported lobectomy as the muscle-sparing treatment according to their results.7–10 The reasons behind these conflicting results lied behind the fact that there was a heterogeneity in study population and the general patient characteristics between the two cohorts were significantly different. According to different studies, markers affecting worse outcomes for segmentectomy included low number of mediastinal lymph nodes sampling 3 and positive surgical margin whether from final pathological reports or frozen section.4,5
We performed a retrospective cohort study to compare lobectomy versus segmentectomy in patients with NSCLC. We collected and analyzed general demographic data, smoking status, preoperative forced expiratory volume in 1 second (FEV1) values, operative details, perioperative complications, time to recurrence, and time to death. Our primary outcomes were disease-free survival and overall survival between lobectomy and segmentectomy. We hypothesized that disease recurrence and overall survival rate were similar between the two groups.
Materials and methods
The study protocol and ethical issues were reviewed and approved by Human Research Ethics Committee, Faculty of Medicine, Ramathibodi Hospital, Mahidol University, Bangkok, Thailand. All patients recorded underwent chest computed tomography (CT) imaging before operations. Pulmonary function tests (PFTs) as well as standard investigations for preoperative lung cancer staging, such as positron emission tomography/CT (PET/CT) scan were performed in all patients. FEV1 values were collected and analyzed accordingly.
The final cohort in our study included 380 patients who underwent surgery for NSCLC at Ramathibodi Hospital between 1st January, 2016, and 31st December, 2020. Lung parenchymal, bronchial, and vascular resections were done using endostaples from either Covidien or Ethicon in all patients. Of the 380 patients, 307 patients underwent lobectomy, while the other 73 patients underwent segmentectomy. When deciding which operation to perform, the surgeons gave advice to all the patients eligible for surgery in the study about the less radical nature of segmentectomy as compared to lobectomy, with no difference in 5-year survival according to multiple contemporary studies. The final decision, however, depended on the suitable anatomy for segmentectomy (i.e., tumor totally located within a particular segment, peripherally located tumor confined to the outer third of the lung parenchyma), but not mainly due to the size or extent of tumor. We also stratified the cases into muscle-sparing thoracotomy or video-assisted thoracoscopic surgery (VATS) for our analysis. Operative, admission, and follow-up data were collected from electronic medical records. Medical records data included general demographic data, comorbidities, details of operation (operative time, blood loss, and blood transfusion), complications, length of hospital stay, histopathology, as well as pleural lavage cytology (PLC). For PLC, we routinely performed three lavages: first lavage (PLC 1) upon entering the thoracic cavity by irrigating and collecting it with 500 milliliters (ml) of normal saline; second lavage (PLC 2) after complete lung resection and lymph node dissection, also by irrigating and collecting the thoracic cavity with 500 ml of normal saline; and third lavage (PLC 3) which was done after performing pleural washing with 5000 ml of normal saline to wash out any possible residual malignant cells from lung manipulation and then irrigating and collecting the thoracic cavity with 500 ml of normal saline, as in PLC 1 and PLC 2, before closing the thoracic cavity. Reports were divided into three groups: positive (malignant cells in pleural lavage fluid), negative (no malignant cells found in pleural lavage fluid), and equivocal (atypical cells found in pleural lavage fluid). Missing data were collected through telephone calls to patients or their relatives in deceased cases. Patients were staged postoperatively according to the 8th edition of the TNM staging system of the American Joint Committee on Cancer (AJCC). 11
Statistical analysis
Patient characteristics with continuous variables were compared using the Student’s t-test (Student. The probable error of a mean, Biometrika, 1908; 1–25.), while categorical variables were compared with chi-square or Fisher’s exact tests. A p-value < 0.05 was considered statistically significant. Potential risk factors were analyzed by univariate and multivariate methods using the Cox regression model. Independent risk factors were expressed as hazard ratios (HR) with 95% CIs. Survival analyses were performed using the Kaplan–Meier method and evaluated with the log-rank test.
Results
Demographic data and clinical characteristics
The study included 380 patients, 307 of whom underwent lobectomy and the other 73 underwent segmentectomy. The demographic and pathological characteristics of the patients are shown in Table 1. Most patients from both groups, although different in number, had similar clinical characteristics, smoking status, and preoperative FEV1 values. Patient’s mean age between the two groups, although statistically different (p = 0.009), was numerically similar (mean ± SD 65.3 ± 9.3 years versus 68.5 ± 10.5 years).
Demographic data and clinical characteristics.

BMI: body mass index; COPD: chronic obstructive pulmonary disease; OSA: obstructive sleep apnea; ILD: interstitial lung disease; HT: hypertension; DM: diabetes mellitus; DLP: dyslipidemia; CKD: chronic kidney disease; CVA: cerebrovascular accident; AF: atrial fibrillation; CAD: coronary artery disease; PAD: peripheral arterial disease; FEV1: forced expiratory volume in 1 second; IQR: interquartile range.
Staging and 5-year follow-up
As previously stated, pathological staging was done postoperatively according to the 8th edition of the TNM staging system of the AJCC. As shown in Table 2, there were patients in almost every stage of NSCLC in both groups. Most patients were in early stages (T1BN0M0, T1CN0M0, and T2AN0M0). According to Table 3, there were no differences in lymphovascular invasion (LVI), visceral pleural invasion (VPI), and surgical margin acquired after operation between lobectomy and segmentectomy group (p = 0.123, 0.493, and 0.999, respectively).
Staging and 5-year follow-up: Pathological NSCLC in both groups.

Pathological finding of NSCLC in both groups (LVI, VPI, and margin status).

NSCLC: non-small cell lung cancer; LVI: lymphovascular invasion; VPI: visceral pleural invasion.
Operative details
VATS were performed more than muscle-sparing thoracotomy surgeries in our hospital; VATS/muscle-sparing thoracotomy: 74.3%/25.7% for lobectomy and VATS/muscle-sparing thoracotomy: 87.7%/12.3% for segmentectomy. For 73 patients who underwent segmentectomy, most patients had only one segment resected [49 patients had one segment resected (67.12%), 3 patients had two segments resected (4.11%), and 21 patients had three segments resected (28.77%)]. For PLC status, there were no statistically significant differences of PLC 1, PLC 2, and PLC 3 results between lobectomy and segmentectomy groups (p = 0.864, 0.658, and 0.546, respectively). Tumor size was significantly larger in the lobectomy group than in the segmentectomy group (median 2.5 cm versus 2.0 cm, p = 0.000). Also, number of mediastinal lymph nodes examined was significantly higher in the lobectomy group than in the segmentectomy group (median 8 nodes versus 3 nodes, p = 0.000). We didn’t perform intraoperative frozen section on lymph nodes acquired during surgery. Also, there was no conversion from segmentectomy to lobectomy in any case.
On the whole, blood loss was significantly higher in the lobectomy group than in the segmentectomy group (median 150 ml versus 100 ml, p = 0.001). We also calculated and analyzed blood loss in all four possible combinations of operations separately (muscle-sparing thoracotomy lobectomy, muscle-sparing thoracotomy segmentectomy, VATS lobectomy, and VATS segmentectomy). For patients undergoing muscle-sparing thoracotomy, blood loss in both lobectomy and segmentectomy group was statistically similar (median 200 ml versus 200 ml, p = 0.591). However, for patients undergoing VATS, blood loss was significantly higher in the lobectomy group (median 120 ml versus 100 ml, p = 0.001).
For operative time, the result was similar between the lobectomy and segmentectomy group (mean ± SD, 191 ± 53 min versus 184 ± 55 min, p = 0.291). Again, we also separately analyzed the operative time according to different combinations of operations. For patients undergoing muscle-sparing thoracotomy, operative time was significantly lower in the lobectomy group than the segmentectomy group (mean ± SD, 201 ± 53 versus 244 ± 54, p = 0.024). For patients undergoing VATS, however, operative time was similar between the two groups (mean ± SD, 187 ± 53 versus 175 ± 50, p = 0.096).
Perioperative complications
Complications recorded were postoperative atrial fibrillation (AF), air leak >5 days, postoperative bleeding requiring re-thoracotomy, prolonged ventilation >48 h, postoperative pneumonia, postoperative myocardial infarction (MI), chylothorax, acute renal failure, pulmonary embolism (PE), stroke, and perioperative mortality as shown in Table 4. Definitions of complications were from STS General Thoracic Surgery Database Full Specifications—for Surgeons version: 2.06 and STS SCA Data Specifications v4.20.2 (Table 5).
Operative details.

VATS: video-assisted thoracoscopy surgery; PLC: pleural lavage cytology; IQR: interquartile range.
Perioperative complications.

AF: atrial fibrillation; RML: right middle lobe; MI: myocardial infarction; IQR: interquartile range; PE: Pulmonary embolism.
The most commonly encountered complication in both groups was air leak >5 days, although none needed repeated surgery (9.8% in lobectomy group and 9.6% in segmentectomy group). However, there were 2 cases which required reoperation: one from lobectomy group for right middle lobe (RML) torsion and another one from segmentectomy group for postoperative bleeding. There was one case of perioperative acute massive PE (0.3%) in the lobectomy group and another case of acute exacerbation of chronic obstructive pulmonary disease (COPD) with respiratory failure in the segmentectomy group (1.4%), both resulting in hospital deaths. All other complications recorded were similar in the 2 groups, although less in number. Hospital stay and chest tube duration were not statistically different between lobectomy and segmentectomy group, with the median values (days) being 6 days (p = 0.802) and 4 days (p = 0.775), respectively. No patients were admitted to the intensive care unit (ICU) postoperatively.
Survival analysis
From Table 2, we separately collected and analyzed the data according to each stage. For patients with early-stage NSCLC (Tmi-1CN0M0) both in lobectomy and segmentectomy group, there was no obvious difference in death, despite the less number of patients in each group (lobectomy versus segmentectomy; TmiN0M0: 1 versus 0, T1AN0M0: 0 versus 0, T1BN0M0: 3 versus 1, and T1CN0M0: 4 versus 0). The results were also similar in patients with recurrence (lobectomy versus segmentectomy; TmiN0M0: 0 versus 0, T1AN0M0: 1 versus 0, T1BN0M0: 6 versus 1, and T1CN0M0: 8 versus 4). For patients with higher-stage NSCLC (T2 and above), again also numerically small, there were no large differences in death and recurrence between the two groups.
As shown in Table 6, the overall 5-year survival rate and disease-free survival between lobectomy and segmentectomy group were similar, as stated by the number of patients with recurrent disease (20.2% versus 16.4%, p = 0.466) and who died on follow-up (6.8% versus 2.7%, p = 0.187). The median time to death in lobectomy group seemed to be more than segmentectomy group, but not statistically different (18.5 months versus 5.8 months, p = 0.127). The median time to recurrence was also similar between the two groups (8.6 months versus 4.5 months, p = 0.511). The median follow-up time for our study was 26.5 months in lobectomy group and 29.7 months in segmentectomy group (Figure 1).
Survival analysis.
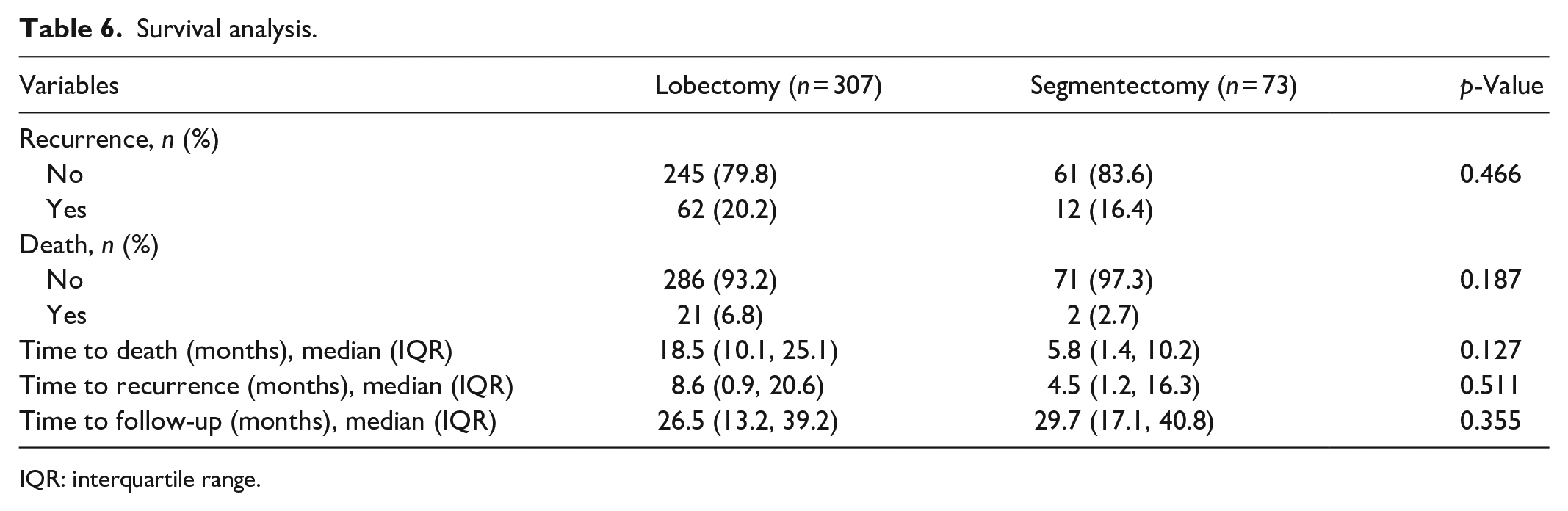
IQR: interquartile range.

Kaplan–Meier curves comparing (a) overall and (b) disease-free survival between lobectomy and segmentectomy group are shown. 12
Discussion
As previously stated, anatomical lobectomy is and has always been the standard treatment for early-stage NSCLC according to the only randomized-controlled trial conducted by LCSG in 1995. 1 However, there are several new reports suggesting that a less radical (sublobar) resection might have similar results regarding recurrence rate and overall survival. Most of the studies were performed only on patients with early-stage (IA) NSCLC. Our study aimed to primarily compare overall 5-year survival rate and disease-free survival in all patients with resectable NSCLC (excluding positive M staging).
There was a study performed by collecting data from the SEER database from 1998 to 2006 comparing outcomes between segmentectomy and wedge resection in patients with stage IA NSCLC. 13 The study population was 3525 patients. The overall and disease-free survival were significantly higher in the segmentectomy group than the wedge resection group. According to the results, our study excluded all patients who underwent wedge resection or any operation even less radical and included only segmentectomy and lobectomy.
A study performed by Subramanian et al. 2 compared patients with stage I NSCLC who underwent lobectomy to all sublobar resections (segmentectomy and wedge resection). The data were collected from National Cancer Database between 2007 and 2012 and included 1687 patients, of which 1354 underwent lobectomy and the other 333 underwent sublobar resection. The study concluded that lobectomy yielded better overall and disease-free survival. In this study, however, the rate of positive surgical margin was significantly higher in the sublobar resection group. We hypothesized that this might result in the worse outcomes with the sublobar resection group in their study. Therefore, we collected the results of surgical margins in all cases in our study. In both lobectomy and segmentectomy group in our study, there was no statistical difference in surgical margin (98.7% versus 100%, p = 0.999). We believed that this homogeneous result would not negatively favor the results of segmentectomy being inferior to lobectomy in our final analysis. Another trial performed by Qiu et al. 5 also emphasized on the importance of surgical margin affecting the outcomes. Their study compared overall and disease-free survival in 245 patients with stage I NSCLC between lobectomy and segmentectomy. The data were collected from Institute of Oncology, Shandong Provincial Hospital Affiliated to Shandong University from 2006 to 2012. They utilized frozen section as a tool to confirm a negative surgical margin during surgery. Therefore, there were no cases with positive surgical margin in the final pathological report in their study, causing no heterogeneity in their patient population. A trial done by Tsutani et al. 6 gave results similar to Qiu et al. and also emphasized the importance of surgical margin, further supporting our trial. Sato et al. 10 initially supported lobectomy, the standard treatment, as being superior to segmentectomy for treatment of stage I NSCLC. However, their segmentectomy group had significantly more cases with positive surgical margin compared to lobectomy group. When analyzing only cases with negative surgical margin, their final results were similar to the previously mentioned trials and also ours.
Another study mentioned another important factor contributing to a favorable outcome for segmentectomy group. Deng et al. 3 collected data from Mayo Clinic from 1997 to 2012 comparing overall survival and disease-free survival between lobectomy and segmentectomy in patients with early-stage NSCLC. The study results favored lobectomy to segmentectomy. The author also mentioned the use of mediastinal lymphadenectomy techniques in all cases, which was the same as our study, instead of more commonly performed mediastinal lymph node sampling. The number of lymph nodes acquired, however, was significantly lower in the segmentectomy group than the lobectomy group in this study. Acquiring lower number of mediastinal lymph nodes meant lower chance of identifying positive nodes and subsequent understaging of disease, which would ultimately lead to undertreatment. This might be the one reason that contributed to the poorer results in the segmentectomy group. We initially hypothesized that, from the results of this study, the number of mediastinal lymph nodes acquired was an important factor affecting the overall and disease-free survival. Therefore, we also collected the data regarding number of mediastinal lymph nodes acquired during surgery. As predicted, the number of mediastinal lymph nodes acquired during surgery in our study were similar to the study by Deng et al., with the lobectomy group being more than the segmentectomy group (median 8 nodes versus 3 nodes, p = 0.000). However, our results proved the contrary, with segmentectomy being statistically similar to lobectomy in overall and disease-free survival, suggesting that the number of acquired mediastinal lymph nodes might not be the only factor affecting the outcomes. From these results, we suggested that our operations might be more adequate than theirs in terms of surgical resection and lymph node dissection. A study performed by Landreneau et al. 4 supported our assumptions. The authors’ study focused on comparing overall and disease-free survival between lobectomy and segmentectomy in patients with stage I NSCLC collected from Lung Cancer Database of the University of Pittsburgh. Their results were similar to ours. They also emphasized the importance of surgical margin. Importantly, they mentioned their literature review from American College of Surgeons Oncology group Z0030 investigation of mediastinal node dissection versus systematic sampling. The final results of the study suggested that mediastinal lymph node dissection or sampling would result in similar overall and disease-free survival, which further supported the results of our study.
Khullar et al. 7 initially concluded that lobectomy resulted in better overall survival than segmentectomy in their study. However, the number of mediastinal lymph nodes acquired in segmentectomy group was equal to or less than 3, significantly lower than lobectomy group. Their initial results were different than ours, with segmentectomy being inferior to lobectomy. However, when analyzing only the cases with similar number of mediastinal lymph nodes acquired, the final results proved the contrary, with segmentectomy being similar to lobectomy. This study suggested the importance of homogeneity in the number of mediastinal lymph nodes acquired between the two groups to compare the results precisely and correctly. Zhang et al. 9 also conducted a similar trial and ended up with the same conclusion as Khullar et al.’s. Because of the contradictory results in comparison of the above trials to ours, the number of lymph nodes acquired during surgery that affects the outcomes should be further studied.
Our results paralleled with the previously mentioned trials. Overall 5-year survival rate and disease-free survival were similar between lobectomy and segmentectomy group as expected. Moreover, not only we collected and analyzed patients with early-stage lung cancer, we included patients with all stages to further evaluate the results of different operations. Again, the results of segmentectomy were still satisfying, being similar to lobectomy even with the inclusion of higher-stages NSCLC.
We initially believed that segmentectomy might cause the remaining lung tissue in the same lobe to consolidate postoperatively, which would precipitate pneumonia. Also, being more technically demanding and requiring adequate learning curve, we suspected that segmentectomy could result in longer operative time and more perioperative complications. We collected the most common perioperative complications reviewed from different literatures. There were 57 cases with complications from both groups. The difference of complications between lobectomy and segmentectomy was not statistically significant. This could imply that performing segmentectomy might not be as difficult as it was thought to be and should strongly be encouraged.
As stated by previous studies,11,14,15 the authors found that positive PLC had a negative impact on survival and was also associated with high recurrence rate. Therefore, we also collected and analyzed the results of PLC, which was performed in 211 cases. For PLC, we routinely performed three lavages: PLC 1 upon entering the thoracic cavity by irrigating and collecting it with 500 ml of normal saline, PLC 2 after complete lung resection and lymph node dissection, also by irrigating and collecting the thoracic cavity with 500 ml of normal saline, and PLC 3 which was done after performing pleural washing with 5000 ml of normal saline to wash out any possible residual malignant cells from lung manipulation and then irrigating and collecting the thoracic cavity with 500 ml of normal saline, as in PLC 1 and PLC 2, before closing the thoracic cavity, as previously explained in Section “Materials and method.” The results of PLC were similar between the two groups (p = 0.864, 0.658, and 0.546, respectively), suggesting that tumor spillage was actually not a concern when performing segmentectomy, which usually required more parenchymal manipulation than lobectomy.
As shown in Section “Result” on operative details, the number of lymph nodes acquired was significantly lower in segmentectomy than lobectomy group (3 versus 8). The reason for this was technical issues in performing segmentectomy; hilar lymph nodes would sometimes be difficult to dissect with the remaining lung parenchyma still intact, as opposed to lobectomy.
We performed both VATS and muscle-sparing thoracotomy in our institution. The decision for which operation to perform was mainly made after analyzing the preoperative imaging (CT chest) on tumor location. For example, if the tumor was deep within the parenchyma, muscle-sparing thoracotomy would be preferred as palpation of the tumor would be possible with this approach. The tumor size was less of a concern.
When comparing surgical approach, we found that VATS was performed more than muscle-sparing thoracotomy in both groups (74.3% in lobectomy group and 87.7% in segmentectomy group), suggesting that there might be added benefits to this approach. Although this study didn’t aim to directly compare the results of VATS and muscle-sparing thoracotomy, the results suggested that a further study on this topic should be encouraged.
As stated, for patients undergoing muscle-sparing thoracotomy, blood loss in both lobectomy and segmentectomy group was statistically similar (median 200 ml versus 200 ml, p = 0.591). However, operative time was significantly lower in the lobectomy group than the segmentectomy group (mean ± SD, 201 ± 53 versus 244 ± 54, p = 0.024). The explanation behind these findings was that judging from the same operative approach, segmentectomy didn’t result in more blood loss even though the technique required more parenchymal manipulation. The operative time for lobectomy was, however, shorter, suggesting that surgeons might be more familiar with the muscle-sparing thoracotomy lobectomy than segmentectomy.
For patients undergoing VATS, blood loss was significantly higher in the lobectomy group (median 120 ml versus 100 ml, p = 0.001), further strengthening our point that segmentectomy alone would not result in more blood loss. Operative time was similar between the two groups (mean ± SD, 187 ± 53 versus 175 ± 50, p = 0.096), suggesting that the thought-to-be-complicated segmentectomy, although performed through a smaller-incision VATS approach, was not actually more technically demanding than lobectomy.
We acknowledged that there were limitations of some aspects of our study. First, not being a randomized-controlled trial, bias couldn’t be fully eliminated as retrospective studies rely on collecting previously recorded medical records by multiple different physicians at different times, resulting in heterogeneity and inaccuracy of the data collected, such as staging and perioperative complications. There was also a quite significant difference in the number of patients in each cohort (307 in lobectomy and 73 in segmentectomy). We initially considered using propensity score matching but after analyzing the general characteristics of patients in both cohorts, we found that there was no heterogeneity between the two groups and using propensity score matching would result in unnecessary loss of too many patients for analysis. Also, neither sample size nor power analysis was used in this study. At Ramathibodi Hospital, we performed segmentectomy if the anatomy was suitable, as stated earlier, not because the particular patient was unsuitable for the more radical lobectomy, thereby resulting in the patient characteristics and preoperative FEV1 results being statistically similar in both groups (1.9 liters versus 1.9 liters, p = 0.502). The mean follow-up time in our study was 26.5 months in lobectomy group and 29.7 months in segmentectomy group. Further follow-ups and studies on these patients may be required to assess the true long-term results of overall and disease-free survival. Lastly, preoperative clinical stages as well as the postoperative pathological diseases of the enrolled patients were not assessed for this study. The reason for this was the limited number of patients in each clinical and pathological stage, so we decided to proceed to analyze the patients in lobectomy and segmentectomy group as two whole separate groups, not individually in each stage. Also, we intended just to analyze the pathological stages because we thought this would be more relevant to our final clinical outcomes than the preoperative clinical stages.
Conclusion
Lobectomy and segmentectomy result in similar overall 5-year survival rate and disease-free survival between these two comparison groups. Therefore, segmentectomy may be a potential alternative for operative treatment of NSCLC. However, a larger and randomized-controlled trial may be needed to further validate these results.
Supplemental Material
sj-docx-1-smo-10.1177_20503121221142171 – Supplemental material for Overall 5-year survival rate and disease-free survival after segmentectomy versus lobectomy in patients with non-small cell lung cancer
Supplemental material, sj-docx-1-smo-10.1177_20503121221142171 for Overall 5-year survival rate and disease-free survival after segmentectomy versus lobectomy in patients with non-small cell lung cancer by Khunthorn Kadeetham, Montien Ngodngamthaweesuk, Narongrit Kantathut, Piya Samankatiwat, Piya Cherntanomwong, Parinya Leelayana and Siam Khajarern in SAGE Open Medicine
Footnotes
Acknowledgements
I would like to acknowledge and give my warmest thanks to my corresponding author and advisor, Montien Ngodngamthaweesuk, M.D., for his expertise and assistance throughout all aspects of our study and for his help in writing the article. I would also like to give my gratitude to my co-authors, Narongrit Kantathut, M.D., Piya Samankatiwat, M.D., Piya Cherntanomwong, M.D., Parinya Leelayana, M.D., and Siam Khajarern, M.D., for kindly providing the information needed to make all this possible.
Author contributions
Khunthorn Kadeetham: concept and design, data correction, review the data, analysis, and interpretation of data, drafting and revising the article, and final approval. Montien Ngodngamthaweesuk: correcting and reviewing the data, revising the article, and final approval. Narongrit Kantathut: correcting, reviewing, and interpreting the data. Piya Samankatiwat: correcting, reviewing, and interpreting the data. Piya Cherntanomwong: correcting, reviewing, and interpreting the data. Parinya Leelayana: correcting, reviewing, and interpreting the data. Siam Khajarern: correcting, reviewing, and interpreting the data.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethics committee, Faculty of Medicine Ramathibodi Hospital, Mahidol University, approved the present study (No. MURA2020/1524).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Informed consent for patient information to be published in this article was not obtained because this is a retrospective study. Requirement was waived by the ethics committee.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
