Abstract
Introduction:
The World Health Organization recommends the viral load test as the preferred method for monitoring responses to antiretroviral treatment; however, this test is not widely available in developing countries. Timely monitoring is crucial; early detection of virological failure can significantly improve health outcomes for these children and help prevent the development of drug resistance. Therefore, this study aims to contribute valuable insights into time to virological failure and its predictors among children with human immunodeficiency virus.
Methods:
An institution-based retrospective follow-up study was conducted among 497 children who were enrolled for first-line antiretroviral treatment from January 1, 2017 to December 31, 2021 in Eastern Ethiopia public Hospitals. A standard pretested checklist was used to extract data. Data were entered using Epi-Data 4.6 and analyzed using STATA 16. Kaplan–Meier was used to estimate the cumulative probability of virological failure, and the log-rank test was used to compare failure curves. The Cox proportional hazard regression model was used to analyze the relationship between independent and outcome variables.
Results:
The overall incidence rate of virological failure was 3.70/1000 person-month observations (95% confidence interval: 2.88–4.76). World Health Organization clinical stages 3 and 4 at the time of antiretroviral treatment beginning (adjusted hazard ratio: 2.38 (95% confidence interval: 1.34–4.26)), baseline cluster of differentiation 4 cells, type of T lymphocytes count below the threshold (adjusted hazard ratio: 3.65 (95% confidence interval: 2.12–6.30)), tuberculosis positive during the follow-up period (adjusted hazard ratio: 4.81 (95% confidence interval: 2.51–9.23)), and poor adherence to the antiretroviral treatment regimen (adjusted hazard ratio: 3.87 (95% confidence interval: 2.08–7.22)) were independent predictors of virological failure.
Conclusion:
The incidence of virological failure in this area was significantly higher compared to nationally conducted studies in Ethiopia. Baseline World Health Organization clinical stages 3 and 4, baseline cluster of differentiation 4 cells, type of T lymphocytes level below the threshold, being tuberculosis infected during the follow-up, and poor adherence were identified as independent predictors of time to virological failure.
Introduction
Virological failure (VF) defined as a persistent viral load >1000 copies/ml after at least 6 months of using antiretroviral treatment (ART) and two consecutive viral load measurements after 3 months of adherence support.1,2 Achieving viral load suppression (VLS) is crucial for successful treatment to enhance the quality of life, reduce the risk of transmitting multidrug-resistant human immunodeficiency virus (HIV), and lower the incidence of HIV/acquired immune deficiency syndrome (AIDS)-related illnesses and deaths in children. 3
According to United States Agency for International Development (USAID) in 2021 report globally, over half (60%) of children receiving ART had not suppressed viral loads, in contrast to 33% of adults. 4 Of the total HIV-infected children and adolescents worldwide, ~90% reside in sub-Saharan Africa. 4 The prevalence is particularly high in eastern and southern Africa, where around 1.2 million (70.6%) of the world’s HIV-positive children reside. Furthermore, children experience lower rates of VLS compared to their adult counterparts. Specifically, based on the study conducted in Malawi, Uganda, and Zimbabwe, the percentages of children aged 1–4 years living with HIV who do not have suppressed viral loads are 62% in 2016, 58% in 2017, and 54% in 2018. 5 Additionally, data from the Ethiopian Demographic Health Survey conducted in 2016 indicates that among HIV-positive children aged 0–14 on ART, 47% experienced failure to achieve VLS. 6
Achieving long-term viral suppression in children receiving ART often poses significant challenges. The progression of the disease in this population tends to be rapid, often leading to adverse health consequences. Failure to identify VF early, combined with inadequate treatment, can result in the emergence of drug-resistant strains of the virus. This situation may ultimately contribute to a decline in clinical status and an increase in mortality rates.7–9
Various studies have shown different factors related to VF such as male gender, 10 poor/fair ART adherence, 11 orphan, 12 history of treatment interruption, 13 World Health Organization (WHO) clinical staging (advanced), 14 opportunistic infections (OI), and younger age.8,12
Over the past 15 years, the Ethiopian government has made significant progress in expanding ART care and treatment across various administrative levels, including regional, zonal, Woreda, and Kebele levels. However, despite these improvements, Ethiopia did not achieve the target of 90% VLS coverage by 2020. The country remains significantly behind in meeting the ambitious 95–95–95 fast-track global targets aimed at ending AIDS among children by 2030. This deficit indicates that VF continues to be a critical issue that requires further investigation into its underlying predictors.7–9,12–16
Currently, achieving VLS is a global priority, as well as a key goal for Ethiopia, in efforts to halt HIV transmission and ultimately end AIDS. In Ethiopia, various studies have been conducted to examine the incidence and predictors of VF in children undergoing ART. However, there has been a lack of research focusing specifically on the timing of VF and its associated predictors among children. Therefore, this study is designed to assess both the timing of VF and its predictors among children receiving first-line ART at selected public hospitals in Eastern Ethiopia.
Method and materials
Study design, period, and area
An institution-based retrospective follow-up study was conducted from July to August, 2022. The study was conducted in Dire Dawa, Harar, and Jigjiga cities’ Public Hospitals. Dawa city is one of the Federal city administrations in Ethiopia, located approximately 515 km east of Addis Ababa (the capital city). Its total population is 341,834, of whom 171,461 are male and 140,461 are female. It has two Public Hospitals, namely Dilchora Hospital (DH) and Sabiyan Hospital (SH). There was a total of 123 children-initiated ART from 2017 to 2021 in these Hospitals.
Harar city is the major city of the Harari Region located 525 km away from Addis Ababa with 215,000 total population residents. It has two Public Hospitals called Hiwot Fana Specialized University Hospital (HFSUH) and Jugel Hospital (JH). HFSUH is the largest Referral Hospital in Eastern Ethiopia, serving for around seven million population. There was a total of 239 children-initiated ART from 2017 to 2021 in these Hospitals.
Jigjiga city is the major city of the Somali Region, located approximately 616 kmfrom Addis Ababa. It has an estimated total population of 304,000 of whom 153,578 are women and 151,422 are men. Jigjiga Suldan Shiek Hassan Yabare University Referral Hospital (JSSHYURH) and Karamara General Hospital (KGH) are the two public Hospitals in Jigjiga city, and there was a total of 135 children-initiated ART from 2017 to 2021 in these Hospitals.
Population, sample size determination, technique, and sampling procedure
The source population was all children treated with first-line ART in Dire Dawa, Harar, and Jigjiga cities’ Public Hospitals, Eastern Ethiopia. The study population all children treated with first-line ART in Dire Dawa, Harar, and Jigjiga cities’ Public Hospitals from the first of January 2017 to the end of December 2021 and children who fulfilled the inclusion criteria. All registered children who received first-line ART during the study period (January 1, 2017–December 31, 2021) in the selected study area were included. Whereas, Incomplete records of registered children, lost records of registered children on first-line ART, and children who initiated ART <9 months during the study period were excluded from the study.
The sample size was determined by using a single population proportion formula with considerations of the following statistical assumption: confidence level 95%, P = the proportion of VF among HIV seropositive children on first-line ART, 28.2% from a study done in Hawasa, Ethiopia, 17 and 5% margin of error (D). Therefore, the minimum sample size was 311. By considering incomplete patient records, 10% of the initial sample size was added, making the final sample size 343.
For predictors of VF, the double population proportion formula was used to estimate the sample size by using adherence status, WHO clinical stage, and treatment regimen as the major predictor variables. Then, a variable that gives the largest sample is used as the independent predictor, which is the WHO clinical stage (to decrease the role of chance), 480 (Table 1). The sample size is calculated by using Epi info version 7.2.3.1 statistical package.
Sample size calculation to assess the time to virological failure and its predictors among children receiving first-line ART in Eastern Ethiopia’s major cities’ Public Hospital, 2022.
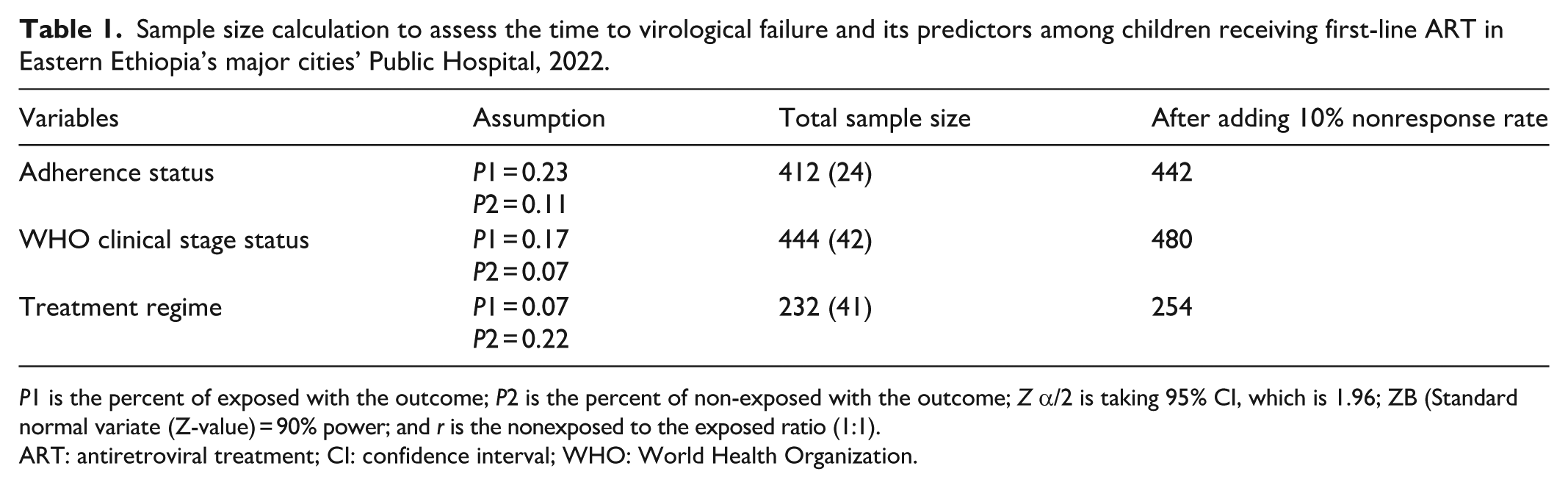
P1 is the percent of exposed with the outcome; P2 is the percent of non-exposed with the outcome; Z α/2 is taking 95% CI, which is 1.96; ZB (Standard normal variate (Z-value) = 90% power; and r is the nonexposed to the exposed ratio (1:1).
ART: antiretroviral treatment; CI: confidence interval; WHO: World Health Organization.
Finally, a census method was used to determine the sample size due to the limited number of cases in the study area. There was a total of 497 children-initiated ART in the study area from 2017–2021, and this is almost the same with the statistically obtained sample size. Therefore, the sample size selected for this study was 497.
The six Hospitals in Dire Dawa, Harar, and Jigjiga cities were purposively selected for this study. All children receiving first-line ART in these Hospitals were selected as study participants. The medical record numbers of the children were used as a sampling frame (Figure 1).
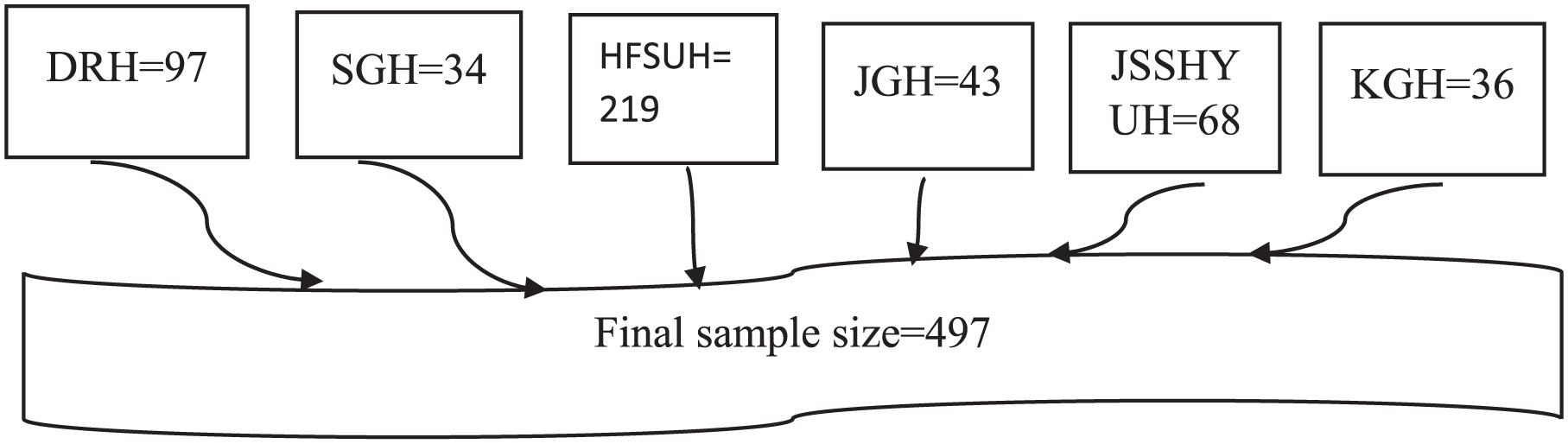
Schematic presentation of sampling procedure to assess the time to virological failure and its predictors among children receiving first-line antiretroviral treatment in selected Public Hospitals, Eastern Ethiopia, 2022.
Variables of the study
The dependent variable was time to VF (event (1) and censor (0)), whereas the independent variables were sociodemographic factors: age of the child, sex of the child, residence, age of caregiver, HIV status of caregiver, child caregiver relationship, parental status alive/dead. Baseline clinical and laboratory predictors: WHO clinical stage at baseline, disclosure status, cluster of differentiation 4 cells, type of T lymphocytes (CD4) absolute count at base line, hemoglobin level at baseline, OI during the follow-up, tuberculosis (TB) diagnosed as positive previously/currently. Highly active ART (HAART) and other medications predictors: types of ART regime, ART adherence level, drug side effect, drug substitution, OI prophylaxis, cotrimoxazole preventive therapy.
Definition of variables
Anemia: For age 6–59 months, <11 g/dl; for 5–12 years, <11.5 g/dl; and for 12–15 years, <12 g/dl (43).
Censored: The children’s outcomes during the study period included those with suppressed viral load, those lost to follow-up, those who died without registered VF, or those transferred out to other health institutions with an unknown outcome.
Follow-up time: The time from ART initiation until either VF or censorship occurs.
Survival time: Measures the follow-up time from ART initiation to the occurrence of the outcome (VF).
Times to event: Time to occurrence of VF measured from the initiation time of ART to the date the event occurred.
Time scale: The time to occurrence of VF or censored cases measured in months.
A loss to follow-up: Children who did not return to the facility for 90 days or more after their last scheduled appointment. 18
VF: Defined as a plasma Viral Load (VL) >1000 copies/ml based on two consecutive viral load measurements at least 3 months apart with intensive adherence support. 19
Adherence to ART: It is the level of compliance to taking ART drugs by the patients during ART follow-up and is classified into good, fair, or poor by the percentage of drug dosage calculated from the total monthly doses of ART drugs. 20
Good adherence level: Drug adherence level ⩾95% or ⩽2 missed drug doses of 30 doses or <3 missed drug doses of 60 doses. 20
Fair adherence level: Drug adherence level of 85%–94% or three to five missed drug doses of 30 doses or four to nine missed drug doses of 60 doses. 20
Poor adherence level: Drug adherence level of <85% or ⩾6 doses of ART missed from 30 doses or >9 doses of ART missed from 60 doses. 20
CD4 count below the threshold for severe immunodeficiency: Is classified according to the child’s age (for infants CD4 <1500/mm3, 12–35 months <750/mm3, 36–59 months <350/mm3, and ⩾5 years <200/mm3). 21
Data collection tool and procedure
The tools were adapted from different literature and modified according to the Ethiopian context. The questionnaire was prepared as a checklist in the English language. The tool consists of sociodemographic characteristics of children and caregivers, base line clinical and laboratory related characteristics, HAART and other medications characteristics, date of ART initiation, and date of outcome. VF time was obtained by considering the time between the date of ART initiation and the date of outcome (the date of VF).
Data quality control, data processing, and analysis
The checklist was pretested among 5% of the study participants in the study area (Dire Dawa, Harar, and Jigjiga cities’ Public Hospitals) to assure the data quality. The necessary modifications and corrections were made to standardize and ensure its validity. The data collectors were senior experienced BSc nurses who are working in ART. One day, training was given on the purpose of the study, the data collection tool, data collection methods, and ethical concerns during data collection. The principal investigator and co-investigator had monitored and checked the data collection process of the data collectors. Data were entered using Epi-Data version 4.6.0 and analyzed by STATA 16 statistical software. Data were cleaned and edited before analysis. Childrens’ cohort characteristics of continuous data were described by mean and standard deviation (SD). Categorical data were calculated using frequency distribution. Then, the outcome of each participant children was dichotomized into censored or VF. Incidence density rate was estimated for the entire study period and specified month intervals of ART follow-up. Consequently, the incidence rate of VF within the study period was divided by the total person month at hazard on follow-up and reported per 1000 person-months. Kaplan–Meier was used to calculating cumulative probability of VF. The log-rank test was used to compare VF curves after initiation of ART. Life table was used to estimating cumulative survival probability at certain time intervals. The Cox proportional hazard regression model was used to analyze the relationship between independent and outcome variables. Bivariate analysis was done for all variables, and variables with a p ⩽ 0.25 were transferred to multivariate analysis. Before multivariate analysis, multicollinearity was tested (mean variance inflation factor = 1.12). Then, proportional hazard assumptions were checked using both the Schoenfeld residual test (global test) with a value of p > chi 2 = 0.6571 and the concordance probability test (Harrell’s c = 0.8317). Also, Cox proportional hazard model fitness to the data was checked using Cox Snell residuals. In multivariate analysis, any statistical test with a p < 0.05 was considered statistically significant. Then, associations were summarized using an adjusted hazard ratio (AHR) and statistical significances were tested at a 95% confidence interval (CI). Lastly, the results were presented using texts, tables, and graphs.
Ethical considerations
Ethical clearance was obtained from Dire Dawa University institutional review board (IRB) with ethical review letter, DDU/IRB/2022/099, and a permission letter was obtained from the management of HFSUH, JH, DH, SH, JSSHYURH, and KGH, along with their HIV care clinics focal person to utilize secondary data for this study. No names or identifying information were recorded on the questionnaires, ensuring that all data extracted from patient charts remained strictly confidential and securely stored. Since this study utilizes secondary data, the IRB granted a waiver/exemption for written informed consent.
Results
Description of study participants
Sociodemographic characteristics of the study participants
After reviewing 497 medical cards of children with HIV/AIDS and initiating ART, 476 (95.77%) of children were found to meet the inclusion criteria and enrolled in the analysis. More than half (244 (51.26%)) were men. The majority of the children (83.61%) were from urban areas. The average age of the cohort’s children at follow-up was 8.73 ± 0.24 SD years (95% CI: 8.25–9.20). Over half of the caregivers (79.20%) were HIV positive (see Table 2).
Sociodemographic characteristics of children and their caregivers in selected Public Hospitals, Eastern Ethiopia, January 1, 2017–December 31, 2021.

HIV: human immunodeficiency virus.
Clinical and laboratory-related characteristics of the study participant
Of the research participants in this study, 18.49% were in WHO clinical stages 3 and 4, and ~35.92% of the children had baseline CD4 levels below the threshold at the start of first-line ART (see Table 3).
Clinical and laboratory-related characteristics of children in selected Public Hospitals, Eastern Ethiopia, January 1, 2017–December 31, 2021.

CD4: cluster of differentiation 4 cells, type of T lymphocytes; OI: opportunistic infection; TB: tuberculosis; WHO: World Health Organization.
HAART and other medications related characteristics of the study participants
From a total of 476 study participant children 65 (13.66%) of them had poor ART adherence, which accounts for 32.79 of the cases of VF (see Table 4).
HAART and other medication-related characteristics of children in selected Public Hospitals, Eastern Ethiopia, January 1, 2017–December 31, 2021.
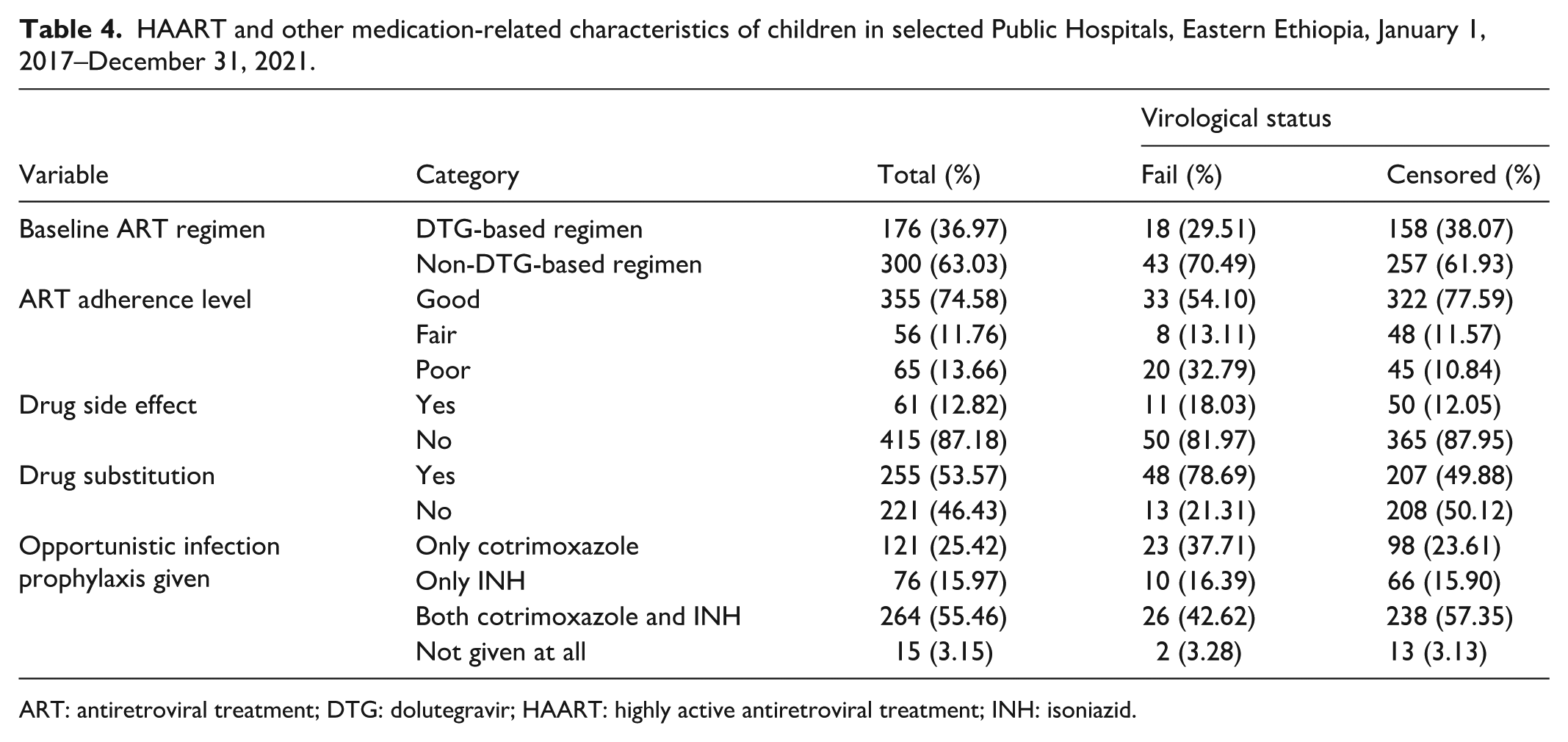
ART: antiretroviral treatment; DTG: dolutegravir; HAART: highly active antiretroviral treatment; INH: isoniazid.
Outcome status of children
A total of 476 children enrolled in the ART clinic were followed for 60 months after their admission. The shortest and maximum follow-up times were 9 and 60 months, respectively, and the total mean follow-up time was 37.50 (95% CI: 36.02–38.98). In this finding, during the follow-up period, 61 (12.82% (95% CI: 10.09–16.14)) of the study participants experienced VF, and 415 (87.18%) were censored (death without registered VF, lost to follow-up, referred to other health facilities, and end of follow-up time; Figure 2).

Outcome of children receiving first-line antiretroviral treatment in selected public hospitals, Eastern Ethiopia, January 1, 2017–December 31, 2021.
VF of children and overall Kaplan–Meier failure function
In this study, the total follow-up time was 16,483.33 person-months, with an incidence rate of 3.70/1000 person-month observations (95% CI: 2.88–4.76). Moreover, the cumulative survival probability of VF in children at 12 months was 98.95%. In addition, the survival probability of children at 24, 36, 48, and 60 months was 95.64%, 90.40%, 84.65%, and 77.66%, respectively. This indicates that as the duration of ART increases, the hazard probability of VF increases (see Table 5 and Figure 3).
Log-rank test for equality of survivor functions of children receiving first-line ART in selected public Hospitals, Eastern Ethiopia, January 1, 2017–December 31, 2021.

ART: antiretroviral treatment; CD4: cluster of differentiation 4 cells, type of T lymphocytes; HIV: human immunodeficiency virus; TB: tuberculosis; WHO: World Health Organization.
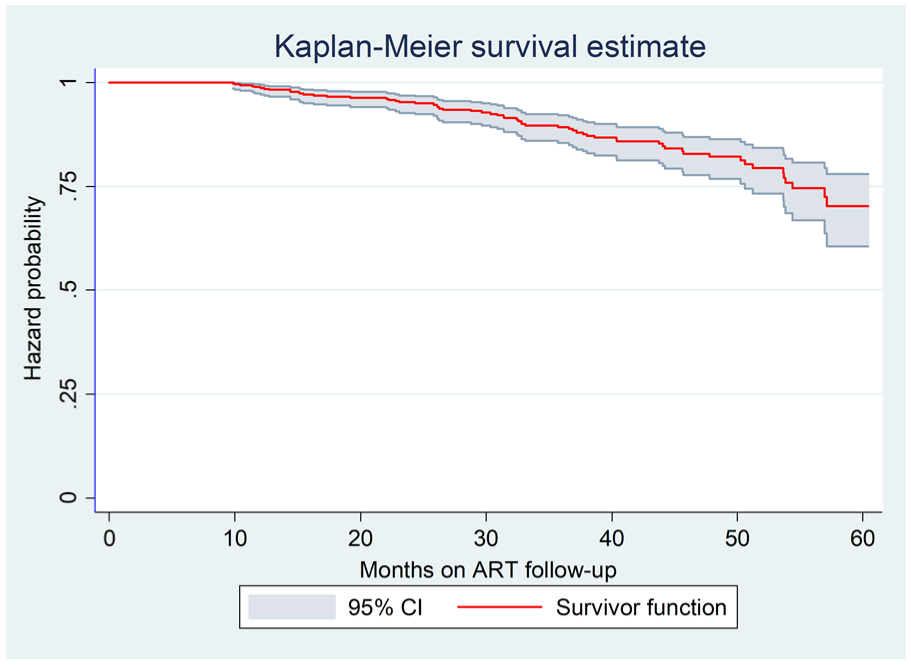
Overall probability of survival among children receiving first-line antiretroviral treatment in selected public Hospitals, Eastern Ethiopia, January 1, 2017–December 31, 2021.
Comparison of failure time for different groups of variables
Children who initiated HAART with baseline WHO clinical stages 3 and 4 had a lower survival time compared to stages 1 and 2. In terms of log rank and p value, this survival time difference was statistically significant (p = 0.0106; see Table 6 and Figure 4).
Bivariate and multivariate analyses for predictors of time to virological failure among children receiving first-line ART in selected public Hospitals, Eastern Ethiopia, January 1, 2017–December 31, 2021.

Those bold numbers above showed significantly associated predictors of time to death.
AHR: adjusted hazard ratio; ART: antiretroviral treatment; CD4: cluster of differentiation 4 cells, type of T lymphocytes; CHR: crude hazard ratio; CI: confidence interval; DTG: dolutegravir; TB: tuberculosis; WHO: World Health Organization.
p value: level of significance at <0.05.

The Kaplan–Meier failure curves for the cohort of children on ART follow-up according to their baseline WHO clinical stage.
Children diagnosed with TB during their follow-up had a higher hazard probability of VF than their counterparts. This difference was statistically significant at log-rank, p = 0.000 (Figure 5).

The Kaplan–Meier failure curves for the cohort of children on ART follow-up according to tuberculosis infection during the follow-up.
The survival rate of children with poor ART adherence was considerably lower than that of children with fair and good adherence (Figure 6). Log-rank and p value of 0.000 indicated that the difference was statistically significant (see Table 6).

The Kaplan–Meier survival curves for the cohort of children on ART follow-up according to their ART adherence level.
On the contrary, there is no survival difference among children who initiate ART with dolutegravir (DTG)-based regimen and non-DTG-based regimen (log-rank p = 0.1659; Figure 7).
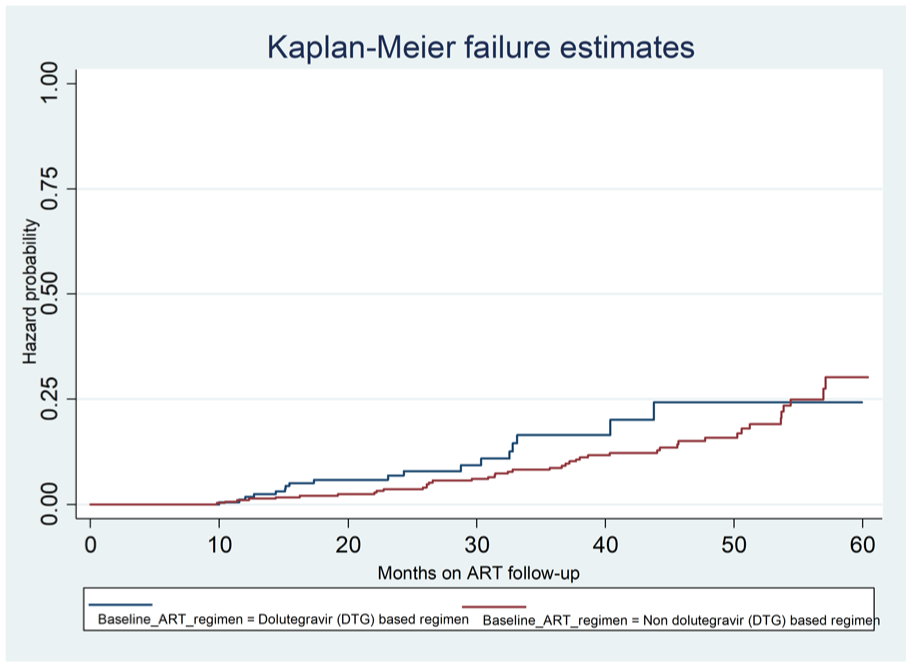
The Kaplan–Meier survival curves for the cohort of children on ART follow-up according to their ART initiation regimen.
Predictors of time to VF among children on first-line ART
The bivariate Cox proportional regression analysis revealed that the following factors were statistically significant predictors of time to VF: being female, WHO clinical stages 3 and 4, baseline CD4 count below the threshold, baseline anemia, baseline TB-positive, TB-positivity during follow-up, and poor adherence (p < 0.05).
According to multivariate analysis, children in WHO clinical stages 3 and 4 at the time of ART beginning had a roughly twofold higher risk of VF (AHR: 2.38 (95% CI: 1.34–4.26)) than children in WHO clinical stages 1 and 2. The hazard probability for VF was 3.65 times higher among children with baseline CD4 count below the threshold as compared to those with normal CD4 count (AHR: 3.65 (95% CI: 2.12–6.30)). Compared to their counterparts, children who were identified as TB-positive during their follow-up period had approximately a fivefold greater hazard ratio for VF (AHR: 4.81 (95% CI: 2.51–9.23)). Similarly, children who had poor adherence to their ART regimen during the follow-up period were 3.87 times more likely to experience VF than children who had good adherence (AHR: 3.87 (95% CI: 2.08–7.22); see Table 6).
Discussion
This retrospective follow-up study aimed to determine the time to VF and its predictors among children receiving first-line ART in Eastern Ethiopia selected public Hospitals. This study shows that the overall VF in Eastern Ethiopia selected public Hospitals in the study period was 61 (12.19%). This finding was approximately two times higher than the study done in Shashemene Town Health Facilities, Oromia Region, Ethiopia (6.5%) 22 and slightly in line with a study conducted in General Hospitals of Two Zones, Tigray, Ethiopia (14%) 14 and Cameroon (17%). 12 However, this finding was lower than studies conducted in Hawasa, Ethiopia (28.2%), 17 Malawi (66%), 23 and Senegal (64%). 24 This apparent difference could be related to difference in study period and setting. The other disagreement might result from variations in the inclusion criteria as well as variations in the definition of VF and its cutoff point.
At the end of the follow-up period, the overall incidence rate of VF was 3.70/1000 person-month observations. This result was in line with studies conducted in Wolaita zone (3.2/1000 person-months of observation) 18 and Tigray Region (5.1/1000 person-months). 14 This similarity may be due to the Ethiopian government’s expansion of HIV diagnosis, ART, and care activities, including the same basic minimum packages of HIV care services with trained health professionals and community health agents at the regional, zonal, Woreda, and Kebele levels. 25
In this study, the cumulative survival probability of VF in children at 12 months was 98.95%. In addition, the survival probability of children at 24, 36, 48, and 60 months was 95.64%, 90.40%, 84.65%, and 77.66%, respectively. This study was in line with a study done in Wolaita zone (99%, 94%, 90%, 85%, and 77%, respectively). 18
After adjusting the multivariable Cox proportional regression model, the significant independent predictors of time to VF were baseline WHO clinical stages 3 and 4, baseline CD4 count below the threshold, TB-positive during their follow-up and poor adherence.
WHO clinical stages 3 and 4 at the time of ART initiation was an important independent predictor of time to VF in which children with baseline WHO clinical stages 3 and 4 at the time of ART initiation had 2.38 times higher hazard of VF compared to children with WHO clinical stages 1 and 2. This finding is comparable with other studies conducted in Amhara Region, Tigray Region, Bahirdar town, Shashemene town, and Jimma University Hospital ART clinic.13–15,22,26 The possible scientific justification is that HIV patient in advanced WHO clinical stage is vulnerable to many OI like pulmonary TB, and severe systemic bacterial infections and AIDS-defining diseases like pneumocystis pneumonia, Kaposi’s sarcoma, esophageal candidiasis, and other many sever illnesses. 27 These OI and AIDS-defining diseases affect the body’s immune system to fight against the HIV replication.
CD4 count below the threshold at the beginning of ART was another significant predictor of time to VF. CD4 count below the threshold increased the hazard of time VF by 3.65 times than normal CD4 count. This result was in agreement with other studies conducted in Southern Ghana 22 and the Wolaita zone.18,28 This could be because of the scientific theory of an inverse association between CD4 cell count and viral replication. Low CD4 cell counts are correlated with reduced immune function, a high risk of OI, and increased viral replication. 29
The hazard of VF for children diagnosed as TB-positive during their follow-up was 4.81 times higher compared to those children who were diagnosed with TB. This is due to the fact that TB/HIV co-infection reduces the CD4 cell count and weakens the immune system, which speeds up the advancement of HIV infection to AIDS, resulting in an increased probability of VF. 30 The other justification may be that a high pill burden and side-effect profile during TB treatment may lead to decreased adherence to ART, which could promote viral replication. 31
The last significant variable that predicted the time to VF of children in this study was poor ART adherence. Children who had poor adherence to their ART regimen during the follow-up period were 3.87 times more likely to experience VF than children who had good adherence. This study is in line with other studies conducted in North West Ethiopia, Amhara Region Referral Hospitals, West Gojjam zone, Amhara Region, Addis Ababa, Tigray Region, and Bahir Dar town public health facilities.13–15,32–34 This is because poor adherence to the ART regimen allows HIV to replicate, which increases the possibility of drug resistance and virological suppression failure. 35
Limitation of the study
Since the study was conducted using secondary sources, some critical covariates obtain from the caregiver and children were missing. These might be significant predictors of time to VF in children such as educational status of caregiver, occupational status of caregiver, nutritional status of children, marital status of care giver, and economic status.
Conclusion
In this study, there was a significant incidence rate of VF in children, and the hazard of VF increases with the length of ART. Baseline WHO clinical stages 3 and 4, baseline CD4 level below the threshold, TB-positive during follow-up, and poor adherence to the ART regimen were identified as independent predictors of time to VF.
Recommendations
Based on the findings, the following recommendations were forwarded:
To the federal minister of health
Research, including this study, showed that the incidence of VF in children had no reduction progress and therefore, federal health policymakers should revise health policies regarding HIV diagnosis, treatment, and care in children based on up-to-date study findings to address interventions for predictors of time to VF.
To hospitals
Hospitals should give an early HIV diagnosis and treatment for children. Another emphasis should be given to TB infection during ART follow-up, and hospitals should prevent TB infection by strengthening TB prophylaxis provision and other possible measures. Also, hospitals should closely follow and monitor for ART regimen adherence since poor adherence was an independent predictor of time to VF in this study.
To future researchers
Upcoming researchers shall conduct a longitudinal prospective cohort study to incorporate impossible variables in a retrospective study, like educational status of caregiver, occupational status of caregiver, nutritional status of children, marital status of caregiver, and economic status.
Footnotes
Acknowledgements
We would like to thank the Dire Dawa administrative health bureau, Hiwot Fana Hospital, Jugel Hospital, Dilchora Hospital, Sabiyan Hospital, Jigjiga Suldan Shiek Hassan Yabare University Referral Hospital, and Karamara General Hospital for their cooperation and permission for the data access. Our great appreciation also extends to data collectors for their great collaboration and tolerance during data collection.
Author contributions
Data curation, formal analysis, resources, software, validation, visualization: M.K.I., N.M.A., S.S.M., A.T.H. The investigation, methodology, supervision, conceptualization, analysis, and interpretation, writing a detailed review, editing, and preparing article: M.K.I., N.M.A., S.S.M., A.T.H. Finally, all the authors have approved the article for submission.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All relevant data are within the article and its supporting information files.
