Abstract
Objective:
Intravenous solutions such as dextrose 5% in water and 0.9% sodium chloride (normal saline) differ in electrolyte composition from human plasma and may contribute to serum chloride derangements (dyschloremia). This retrospective study aimed to explore the relationship between fluid composition, particularly medication diluents, and dyschloremia development in the intensive care unit.
Methods:
This was a retrospective chart review of adult intensive care unit patients with normal serum chloride on admission who developed dyschloremia after 48 h of intensive care unit admission. Data were collected on fluid types and volumes administered in the intensive care unit in the 7 days prior to dyschloremia onset. Descriptive statistics and chi-square tests were used to compare characteristics between patients who developed hyperchloremia and hypochloremia.
Results:
Of 884 screened patients, 85 developed dyschloremia after 48 h (41 hypochloremia, 44 hyperchloremia). There was no significant association between the proportion of normal saline or dextrose 5% in water-containing fluids and the type of dyschloremia. However, dexmedetomidine, typically diluted in normal saline, was associated with hyperchloremia.
Conclusion:
While total fluid composition was not associated with dyschloremia type, high-volume use of specific diluent-medication combinations may contribute. Strategies to minimize diluent volume could help reduce dyschloremia risk.
Keywords
Background
Critically ill patients frequently require the administration of intravenous (IV) fluids to meet their pharmacotherapeutic goals. There are four main categories of fluids provided to critically ill patients: resuscitation fluids (for intravascular volume expansion), maintenance fluids (to replace ongoing fluid losses), nutritional fluids, and inadvertent fluids (predominantly as a diluent for intravenously administered medications or fluids administered to keep an intravenous or intra-arterial catheter patent). Volume overload in critically ill patients is associated with poor outcomes, including multi-organ failure, electrolyte imbalances, increased hospital stay, and mortality.1,2 IV medications, specifically antibiotics, have been previously shown to be significant contributors (up to 40%) to the daily fluid intake of intensive care unit (ICU) patients.2,3
Dextrose 5% in water (D5W) and 0.9% sodium chloride or normal saline (NS) are the most common IV solutions used in the ICU setting. These solutions have different concentrations of electrolytes compared to human serum plasma. Balanced salt solutions (BSS), such as Plasmalyte® or Ringer’s lactate (RL), provide electrolyte replacement in concentrations like normal serum concentrations (Table 1).
Na: sodium; Cl: chloride; K: potassium; pH: potential of hydrogen; NS: normal saline; D5W: dextrose 5% in water; RL: Ringer’s lactate; IV: intravenous.
Variability in different labs’ references.
Because of the difference in concentration of the electrolytes of the NS and D5W with plasma, large-volume administration of such fluids can lead to electrolyte disturbances. In one study, administration of NS as the primary IV medication diluent in ICU patients was associated with a significantly increased risk of hyperchloremia compared to when D5W was used as the primary diluent (17.9% versus 10.5%, p = 0.037). 7 Several randomized controlled trials have compared BSS and NS in adult ICU and emergency department patients. The SALT-ED and SMART trials demonstrated that use of BSS was associated with lower rates of major adverse kidney events compared to NS in both critically ill and non-critically ill populations.8,9 The SMART trial highlighted that the use of 0.9% saline in critically ill adults is associated with higher serum chloride levels compared to balanced crystalloids, which can increase the risk of hyperchloremia. 9
Excessive salt intake or prolonged diarrhea are risk factors for developing hyperchloremia.10,11 Reduced intake, vomiting, diarrhea, high-dose diuresis, and congestive heart failure can lead to hypochloremia development.10,11 Studies have previously shown an association between dyschloremia and an increased mortality risk.12–19 Specifically, abnormalities in serum chloride on days 1 and 3 are independently associated with extended hospital stay. 20 Although association does not always suggest causation, it is possible that hyperchloremia-induced metabolic acidosis or hypochloremia-induced metabolic alkalosis could have contributed to the observed effects.10,21 Dyschloremia has been associated with acute kidney injuries (AKIs) in the literature.10,19,20–25
Given the contribution of IV medications to fluid intake of critically ill patients and the association of NS and D5W solution administration with the development of hyperchloremia and hypochloremia, respectively, mixing the IV medications in BSS as diluents may minimize the risk of these electrolyte disturbances. This practice could reduce the incidence of AKIs and improve patient outcomes. Very few medications are routinely mixed with BSS in critical care settings. Reasons for this include slightly higher procurement costs, limited stability and compatibility data available for use as IV medication diluents, and a need for more extensive clinical experience.
The purpose of this study was to determine the contribution of different fluid types (purpose, volume, chloride concentration) to dyschloremia development in ICU patients. It aims to identify specific drugs (and their associated diluents) that are the most significant contributors to dyschloremia development. This will identify potential targets for alteration in the medication diluent to minimize the contribution to dyschloremia.
Methods
This was a retrospective chart review of adult patients admitted to the medical ICU from January 1, 2023, to December 31, 2023, with normal serum chloride who subsequently developed at least one abnormal serum chloride concentration measurement on day 3 or later during their ICU stay. Exclusion criteria included ICU stay less than 3 days, development of dyschloremia within the first 48 h of admission, no dyschloremia development during ICU stay, patients with incomplete chart data, patients requiring dialysis, patients with glomerular filtration rate <15 mL/min, and patients previously enrolled in the study during the chart review process. The prevalence of dyschloremia developing in the medical ICU was anticipated to be about 30% based on previous studies (note studies’ findings have been inconsistent). 15 All patients admitted to the ICU in the calendar year 2023 were eligible for inclusion.
Dyschloremia was defined as any serum chloride concentration <98 mmol/L for hypochloremia or >108 mmol/L for hyperchloremia, based on institutional lab reference ranges. 10 Only results from central lab analyzers were included; point-of-care results were excluded. Patients needed at least one abnormal value after 48 h of ICU admission to qualify.
Fluid intake data was collected for the 7 days preceding the patients’ first dyschloremia in the ICU. Volumes and types of fluid received were recorded for only the time within the ICU and not from any other areas the patient may have been prior to ICU admission. Data was only collected on the patients’ first, and not any subsequent, dyschloremia event, to avoid any potential bias from the first dyschloremia increasing the risk of future events.
Patients were excluded if they had incomplete data in the electronic medical records, had less than 3 days of ICU stay, had dyschloremia in the first 48 h or no dyschloremia at all during their ICU admission, were pregnant, or if they had end-stage renal disease at some point up to 7 days before the development of the first dyschloremia. The specific volume of undiluted liquid medications, intravenous or oral contrast solutions, or blood products (except intravenous albumin) was not collected.
Statistical analysis
The majority of the data analysis was descriptive in nature. Comparative analyses between patients who developed hypochloremia versus hyperchloremia were performed using chi-square test of independence with a significance threshold of 5% (α = 0.05). 26 Given the exploratory nature and limited sample size, multivariable regression was not conducted. The study was not powered to detect relative risks, and findings should be interpreted as hypothesis-generating. Sensitivity analyses were not performed as part of this study due to the limited availability of additional variables for subgroup evaluation.
The targeted outcomes included the following: percentage of patients developing hyperchloremia and hypochloremia post 48 h of ICU stay, proportion of NS contribution to total daily fluid intake in patients with hyperchloremia, proportion of D5W contribution to total daily fluid intake in patients with hypochloremia, proportion of NS or D5W fluid intake as each of the fluid types (resuscitation versus nutrition versus inadvertent versus maintenance), and most significant high-volume medications that could be potential contributors to dyschloremia. Secondary outcomes evaluated included the rate of AKI development, metabolic acidosis, and metabolic alkalosis in the 7 days following the first dyschloremia.
Results
Eight hundred eighty-four patients were admitted to the ICU in calendar year 2023. Reasons for exclusion from the evaluation are listed in Figure 1. Eighty-five patients (9.6%) developed dyschloremia following 48 h of admission to the ICU and met the inclusion criteria. Forty-one patients developed hypochloremia, and 45 patients developed hyperchloremia (Figure 1).
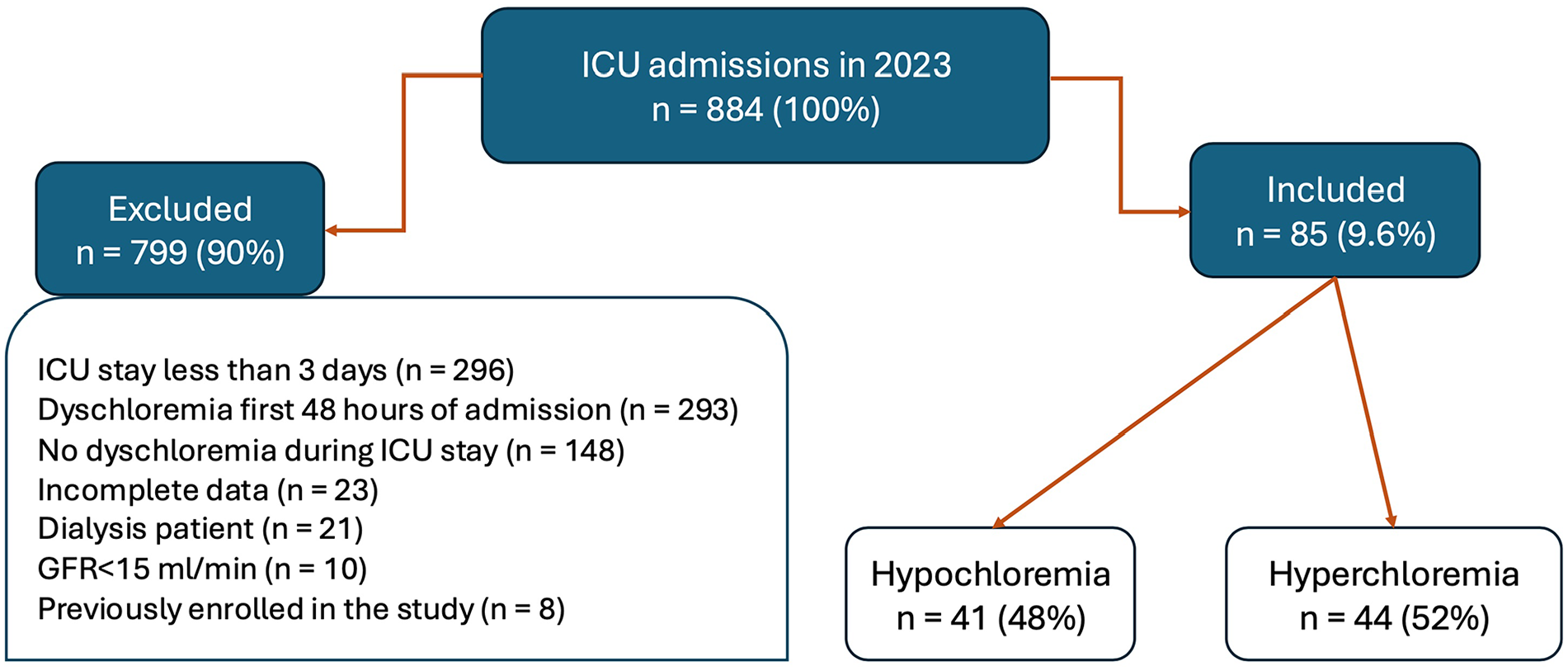
Overview of patient screening and selection process.
Baseline characteristics were not significantly different between the two groups (Table 2). Twenty-five percent of patients had chronic lung disease (e.g., asthma or chronic obstructive pulmonary disease) at baseline.
Composition of baseline characteristics.

In patients who developed hyperchloremia, 23% of their total daily fluid contribution consisted of NS-containing solutions, and in patients with hypochloremia, 31% of their total daily fluid contribution consisted of D5W-containing solutions. The mean duration of ICU admission before development of the first dyschloremia was 4.12 days. The majority of the volume of fluid was administered on the first day of admission, which corresponded to 2–4 days before the development of dyschloremia (Figure 2). Patients received a mean of 2.9 L of fluid daily, about 1 L of which consisted of IV medications followed by 939 mL of nutritional fluid (Figure 3) over a maximum of 7 days before the development of dyschloremia.

Daily breakdown of fluid composition.

Mean daily volume of fluid per patient.
IV medications mixed in D5W as diluent contributed more to total daily fluid intake (~27%) than medications mixed in NS (~10.5%) in patients with dyschloremia (Figure 4). The volume contribution of NS and D5W containing IV medications was similar between patients who had developed hypochloremia and those who had developed hyperchloremia.

Mean daily volume of fluid type per patient.
Medications contributing the greatest volume to the patients’ daily fluid intake for patients receiving any of dexmedetomidine (mixed in NS), azithromycin (mixed in D5W), and multivitamin (mixed in D5W) are demonstrated in Table 3. The only medication significantly associated with an increased risk of dyschloremia (hyperchloremia) was dexmedetomidine (Table 4).
Top large-volume medications.

D5W: dextrose 5% in water; NS: normal saline.
Association of high-volume medication administration with dyschloremia.

The only statistically significant association of dyschloremia with an adverse event was found to be the association of hypochloremia with more metabolic alkalosis development (Table 5).
Association of dyschloremia with adverse events.

Discussion
The results showed only a 10% incidence rate of dyschloremia development in patients admitted to St. Paul’s Hospital ICU, lower than the 30% previously described. 15 BSS were the predominant fluid used for resuscitation (Figure 4). Studies have previously suggested using BSS instead of chloride-rich solutions for resuscitation in the ICU could prevent the development of hyperchloremia and its sequalae.8,9 This study found the largest daily volume contribution to be from IV medication diluents rather than resuscitation fluids. It is worth noting that the ICU pharmacist frequently intervenes by changing the diluent of IV medications if a trend in the serum chloride concentration suggests an imminent risk of developing dyschloremia. Such efforts are limited by the available literature describing the safety of utilizing alternative diluents and concentrations for many medications for intravenous administration. The pharmacists’ interventions may have led to a lower rate of development of dyschloremia in our patients than expected based on previous studies. 17
We acknowledge that by including only patients who developed dyschloremia, our study does not allow for the estimation of relative risk compared to normochloremic patients. Additionally, while chi-square analysis was used to explore associations between exposures and type of dyschloremia, multivariable regression would provide a more robust method of assessing independent predictors. These approaches were not feasible within the scope of our data set, but future studies could adopt these methods to confirm our findings.
We excluded patients with dyschloremia within the first 48 h of ICU admission to isolate the effect of ICU-administered fluids and disregarded the fluids administered prior to ICU admission (e.g., in the emergency department), similar to previous studies.15,17
Contrary to our initial hypothesis, we did not observe a direct association between the proportion of NS- or D5W-containing fluids and the development of hyperchloremia or hypochloremia. However, all patients who developed dyschloremia had received substantial daily fluid volumes, with IV medications contributing the majority. Interestingly, D5W-based IV medications made up a larger portion of fluid intake across both dyschloremia types, even in patients who developed hyperchloremia. This paradox may reflect the complex interplay of disease processes such as diuresis, diarrhea, and insensible losses that affect serum chloride independently of fluid input. Although we did not find a direct association between the overall proportion of NS- or D5W-containing fluids and the development of dyschloremia, this did not rule out the potential impact of specific high-volume exposures. Dyschloremia, in our study, functioned as a clinically meaningful signal for identifying patients who had received significant fluid volumes. Since most of this volume came from IV medications, we analyzed individual agents to determine whether any single large volume medication-diluent combination disproportionately contributed to fluid load and, in turn, altered serum chloride. This more granular approach revealed that dexmedetomidine, typically diluted in NS, was associated with hyperchloremia despite the lack of a broader correlation with NS volume.
Dexmedetomidine, an alpha-2 agonist commonly used as a sedative in the ICU, contributed the largest volume to patients’ daily volume intake (14%) and was associated with the development of hyperchloremia in patients who received it. This can be expected as dexmedetomidine is routinely mixed in chloride-rich NS.
Various concentrations of dexmedetomidine solution (4, 8, 12, and 20 mcg/mL) have demonstrated physical and chemical stability in NS as a diluent.27–29 Limited stability data (up to 24 h at room temperature) exists for mixing dexmedetomidine in D5W and only at the standard concentration of 4 mcg/mL. 30 In terms of dexmedetomidine stability in BSS, 100 mcg/mL has demonstrated only physical stability in RL. 31 Compatibility data only exists for the standard concentration of dexmedetomidine (4 mcg/mL), which suggests compatibility with NS, D5W, and RL.27–31
Limitations
This study has several limitations. There was no sample size calculation, and it was empirically decided that all patients admitted to St. Paul’s Hospital in the year 2023 be screened for inclusion. By including only patients who developed dyschloremia, we were unable to calculate relative risk compared to patients who maintained normal chloride levels. Our retrospective design limited the ability to control for confounding variables such as the use of diuretics, gastrointestinal losses, and diaphoresis, all of which may influence serum chloride levels. The sample size of patients with dyschloremia, while sufficient for exploratory comparisons, restricts the power of inferential statistics. Nonetheless, this study contributes valuable insights by highlighting the often-overlooked role of medication diluents in ICU fluid and electrolyte management, and it provides a foundation for future prospective studies to examine this relationship more robustly.
Regardless of dyschloremia development, increased volume intake in the intensive care setting is associated with increased length of stay, mortality, and increased mechanical ventilation days. 2 Other adverse effects associated with fluid overload include cardiac dysfunction, AKI, abdominal compartment syndrome, and impaired wound healing. 32 Recent reviews on fluid accumulation syndrome in sepsis and septic shock emphasize the importance of a multi-modal approach to de-resuscitation, combining a restrictive fluid management regime and limiting IV medications.33,34
To limit the daily fluid intake and potential adverse events described above, ICU clinicians should explore the potential for minimizing the provision of high-volume IV medications. As soon as the ICU patient has gained tissue perfusion and can use the gastrointestinal tract with adequate absorption, IV medications can be switched to formulations (tablet, capsule, liquid) for enteral administration, wherever possible. For example, dexmedetomidine could be switched to oral clonidine; azithromycin could be switched to an oral tablet with similar dosing, and enteral multivitamin formulations could be utilized.
If adequate oral absorption cannot be achieved in a patient at high risk of developing hyperchloremia, one can consider concentrating the dexmedetomidine solution to limit the diluent volume. Preslaski et al. have demonstrated the stability of 20 mcg/mL concentration in NS, but compatibility data with other solutions and medications is not available at this time. 29 Hence, while a 20 mcg/mL bag may be used, a dedicated IV line for the administration of dexmedetomidine would be necessary. This requirement may be complex for patients in critical condition needing multiple IV access or those who have limited IV access. The physical compatibility of dexmedetomidine 100 mcg/mL in RL has also been established, but the chemical compatibility and stability remain unknown. 31 Future research to determine the compatibilities of more concentrated dexmedetomidine solutions with NS and BSS would help guide clinicians.
Conclusions
This study found a 10% rate of dyschloremia in ICU patients developing after 48 h. While the overall fluid composition (NS or D5W) was not associated with the type of dyschloremia, specific high-volume medications such as dexmedetomidine mixed in NS were associated with hyperchloremia. Future studies should explore the compatibility of concentrated or alternative diluent formulations to minimize fluid burden and electrolyte disturbances in critically ill patients.
Footnotes
Ethical Considerations
Ethics approval was granted on November 15, 2023, by the University of British Columbia-Providence Health Care Research Ethics Board (UBC-PHC REB). Approval number/ID: H23-03431.
Consent to Participate
Written informed consent was waived by the Institutional Review Board (University of British Columbia-Providence Health Care Research Ethics Board (UBC-PHC REB)) due to the retrospective nature of the study and the absence of identifiable patient data.
Consent for Publication
Written informed consent was waived by the Institutional Review Board (University of British Columbia-Providence Health Care Research Ethics Board (UBC-PHC REB)) due to the retrospective nature of the study and the absence of identifiable patient data.
The authors declare that they have no competing interests.
The authors declare that they received no funding for this project. This lack of external funding helps to ensure the independence of the research and eliminates the potential for conflicts of interest related to sponsor influence on study design, analysis, or interpretation.
Author Contributions
GB and MM designed the research question and project. GB retrieved the patient lists from electronic medical records. MM screened patients and collected data. GB contributed to a data analysis plan, which MM performed. All the authors have interpreted the data and contributed to the article’s writing. All the authors have read and approved the final article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data set utilized for this analysis is not available for other researchers based on limitations imposed by the Ethics Board.
Informed Consent
No patient identifiers were included in this retrospective study, and no consent was required to obtain ethics approval.

