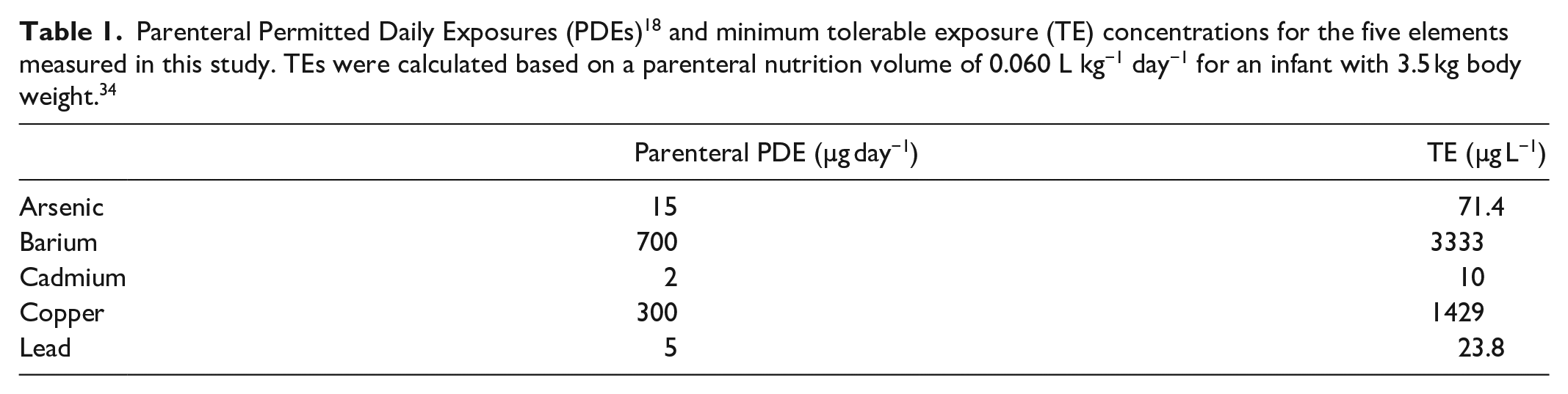
Abstract
Objective:
In the delivery of intravenous fluids, in-line warming devices frequently transfer heat using a metal heating plate, which if uncoated can risk elution. This bench study examined extractable elements detected following long-term use of the parylene-coated enFlow® Disposable IV/Blood Warmer.
Methods:
We tested 16 clinically relevant challenge fluids typical of the surgical setting, including commercially available single donor blood and blood products as well as intravenous saline and electrolyte solutions. After 72 h of warming at 40°C (104°F) via the enFlow, analytical chemistry identified and quantified the most clinically significant extractable elements (arsenic, barium, cadmium, copper, and lead) to estimate chemical exposure. We also measured the extracted concentrations of these five elements following simulated use of the device with three solutions (Sterofundin ISO, Plasma-Lyte 148, and whole blood) that were pumped through the warmed device at two different flow rates (0.2 and 5.5 mL min−1).
Results:
Across all scenarios of acute and long-term exposures for different populations, the enFlow demonstrated low toxicological risks as measured by the calculation of tolerable exposure for extracted arsenic, barium, cadmium, copper, and lead.
Conclusion:
The results suggest biological safety for the use of parylene-coated enFlow with a variety of intravenous solutions and in different therapeutic scenarios.
Keywords
Introduction
Hypothermia is a common complication of surgery occurring during general anesthesia or recovery. 1 Avoidance of hypothermia helps reduce mortality and morbidity and reduces hospital length of stay. 2 Treatment guidelines recommend the use of intravenous (IV) fluid warmers as part of an overall hypothermia prevention strategy. 2 IV fluid warmers usually operate through the use of in-line microwaves, countercurrent cartridges, or direct-contact block heaters. 3 Of these, the direct-contact heaters are most commonly used because of their higher effectiveness and safety profiles, even at high flow rates.
Medical devices in direct contact with blood and IV fluids have the potential to introduce chemicals that may prove toxic, and manufacturers must document contaminants introduced by their devices to ensure patient safety. 4 Recently, aluminum block-based fluid warmers were found to leach high levels of aluminum into the infusate, leading to the recall of several devices. 5 For example, the disposable cartridge for the enFlow™ IV/blood warmer (Vyaire Medical, Mettawa, IL, USA) was voluntarily withdrawn and replaced with a similar cartridge that included a thin coating of parylene over the aluminum heating blocks. 6 Recalls have also been announced by Eight Medical for the Recirculator 8.0 Disposable Lavage kit; Smisson-Cartledge Biomedical for the ThermaCor 1200 Disposable Sets; and Smiths Medical for the Level 1 Fluid Warming Systems.5,7
Parylene is a polymer used to resist the corrosive action of organic solvents and fluids. 8 Parylene is applied as a gas and deposited on the target surface, producing a layer ranging in thickness from angstroms to mils in thickness. The process develops an ultra-thin conformal layer that is pinhole-free and is chemically inert. This layer is waterproof and electrically isolated. 8 The addition of parylene into the enFlow cartridges dramatically reduced aluminum leaching below tolerable exposure (TE) limits set by United States Food and Drug Administration (FDA) 9 without affecting warming capacity. 10 A pilot in vivo animal study showed no evidence of toxicity after mice were injected with fluids warmed for 72 h by the enFlow cartridge. 9 Numerous previous studies have documented concerns over aluminum leaching from fluid warmers.5,9,11 –17
In addition to aluminum, the FDA has established Permitted Daily Exposures (PDEs) for all elements of toxicological concern within finished drug products. 18 The FDA considers daily exposure below these PDE levels to be protective of public health for all patient populations and is established for three different routes of administration: oral, parenteral, and inhalational. In this study, we used the parenteral PDE values since IV administration is the intended use of the enFlow cartridge.
In the same guidance document, the FDA classifies elements into three classes based on their toxicity level and their likelihood to occur in a drug product, with class 1 being the highest risk category and class 3 the lowest. 18 We measured elemental concentrations for five elements: arsenic, barium, cadmium, copper, and lead.
Arsenic, cadmium, and lead are classified in the highest risk category (class 1) based on their high toxicity and likelihood of occurrence. Arsenic is a carcinogen that is present in air, soil, and groundwater. Arsenic exposure has been linked to several types of cancer as well as a range of non-cancerous adverse effects including skin, gastrointestinal tract, neurological, reproductive, and vascular disease. 19 Cadmium is used in electroplating and electronics and can be found in medical devices. Cadmium and cadmium chloride cause lung cancer and are associated with kidney and prostate cancer.20,21 Due to its toxicity, cadmium is one of six substances banned by the European Union’s Restriction of Hazardous Substances (RoHS) directive. 22 Lead is an extremely common element that has been widely used for centuries. It has only been recently found to be a neurotoxin, prompting its removal from non-battery applications such as paint and gasoline. Major regulatory interventions were designed to reduce lead levels in the pediatric age group as it been associated with delayed mental development in children. 23 The US Centers for Disease Control and Prevention (CDC) recently decreased the upper value of the reference range of blood lead level from 50 to 35 µg L−1 for children. 24
Barium and copper are class 3 elements that are common byproducts in manufacturing and medical applications and have clearly defined toxicities. Barium is considered highly toxic as a potassium channel blocker, resulting in neurological and cardiovascular toxicity. 25 Furthermore, barium exposure is associated with renal toxicity. 26 Copper is an essential element in human metabolism, involved in aerobic respiration of eukaryotic cells. Accumulation of high levels in the liver leads to cirrhosis in the genetic disorder Wilson’s disease, and high blood levels have been associated with Alzheimer’s disease.27,28 Furthermore, copper exposure results in gastrointestinal, kidney, and liver adverse effects. 29 Copper is widely used in the electronics industry and can be found in medical devices.
This study was designed to evaluate leaching of arsenic, barium, cadmium, copper, and lead into IV fluids after warming with the parylene-coated aluminum contact heater. We evaluated the levels of these industrial elements in 16 clinically relevant challenge fluids typical of the surgical setting, including commercially available blood and blood products as well as IV saline and electrolyte solutions after prolonged exposure (5 or 72 h) to a parylene-coated aluminum contact heater. We hypothesized that exposure to the enFlow cartridge would not result in leaching of the five elements in levels above the FDA’s exposure limits.
Methods
We performed two different bench experiments over the course of 2 weeks for this study: acute dynamic flow fluid and long-term quasi-static fluid flow.
Experimental setups
The experimental setups for dynamic and quasi-static testing were previously described.9,17,30 Briefly, for the dynamic testing, the challenge fluid was pumped through the enFlow cartridge at a fixed flow rate for 5 h and the outputted fluids were collected. The enFlow warmer was activated for the duration of the testing at a set temperature of 40°C. Each solution was tested at two flow rates: 0.2 mL min−1 to simulate neonatal use and 5.5 mL min−1 to simulate typical use in adults. At the conclusion of each test, element concentrations (µg L−1) within the outputted fluids were measured. The enFlow device underwent a series of extractable studies with a special attention to the amount of aluminum that could be complexed by worst-case extraction matrixes. The fluid path of the cartridge was extracted using a variety of infusates at 40°C for 72 h. The extracts were analyzed by inductively coupled mass spectroscopy (ICP/MS) for metals, headspace gas chromatography-mass spectrometry (HS-GC/MS) for volatile organic compounds (VOCs), and direct injection GC/MS for semi-volatile compounds (SVOCs). Using these analytical chemistry techniques, the extractable compounds and leachable elements were identified and quantified to determine the chemical dose to the user.
For quasi-static testing, the experimental setup and methods followed the principles described in ISO 10993-1:2016 and ISO 10993-18:2005.31,32 Specifically, an enFlow cartridge, which has an internal volume of approximately 5 mL, was filled with a challenge solution and closed with a cap to create a closed system. The filled cartridges were then placed inside a temperature chamber at 40°C and gently rocked continuously for 72 h. Following 72 h, the total content of each mineral within the cartridge was quantified. Since there was no flow during the quasi-static tests, the final content within the closed system was expressed as μg device−1.
Dynamic and quasi-static bench tests were performed by American West Analytical Laboratories (AWAL, South Salt Lake City, UT, USA) and managed by Nelson Laboratories, LLC (Salt Lake City, UT, USA). AWAL is accredited by the National Environmental Laboratory Accreditation Program.
Challenge solutions
For dynamic testing, three challenge solutions were examined: Sterofundin ISO (B. Braun Melsungen AG, Melsungen, Germany), Plasma-Lyte 148 (Baxter International, Deerfield, IL, USA), and whole blood (StemExpress, Folsom, CA, USA).
For quasi-static testing, 16 challenge solutions were tested: Sterofundin ISO (B. Braun); Plasma-Lyte 148 (Baxter International); single donor human whole blood (StemExpress); human packed cells (StemExpress); Ringer’s lactate in 5% dextrose (Baxter International); human platelet lysate (StemExpress); human buffy coat (StemExpress); human plasma diabetic type 2 (StemExpress); 5% dextrose solution (Pfizer Inc., New York, NY, USA); 3% sodium chloride injection (B. Braun); human serum albumin 25% (StemExpress); normal human serum off-the-clot charcoal-dextran 1 (StemExpress); human cord blood (StemExpress); leukocytes (StemExpress); potassium chloride in 5% dextrose and 0.9% sodium chloride (Pfizer Inc.); and 10% dextrose and 0.45% sodium chloride (B. Braun).
For dynamic testing, a single run was performed for each of the three challenge solutions and two flow rates tested for a total of six runs. For quasi-static testing, a single run was done for each of the 16 challenge solutions.
Analytical chemistry
In both dynamic and quasi-static bench testing, the element concentration post-warming within the challenge fluids was determined using ICPMS as described previously. 9 Measured concentrations were not blank corrected.
Each element and concentration measurement has an associated method detection limit (MDL) which corresponds to the minimum concentration a substance can be measured with 99% confidence that the concentration is greater than zero. 33 MDL is dependent on the laboratory, technique, element, and dilutions. For measurements below the MDL, results are presented in this article as “<MDL” or “–.”
Bench testing acceptance criteria
To quantify the toxicological hazard of clinical use of the enFlow cartridge, we compared the measured extracted metal concentrations to the parenteral PDEs established by the FDA. Specifically, we quantified a Margin of Safety for each element which is a unitless index of the ratio between the acceptable level and measured exposure level. A Margin of Safety greater than 1.0 indicates low toxicological risk. 34 For the dynamic testing, we converted the daily parenteral PDEs (µg day−1) to a conservative TE (µg L−1) concentration by assuming the lowest parenteral nutrition volume (0.060 L kg−1 day−1) for the standard infant weight specified in ISO 10993-17:2002 (3.5 kg): 34
For the quasi-static testing, the total element content that accumulated within the enFlow cartridge over the 72-h period was compared to the 24-h PDE:
Table 1 lists the PDEs and corresponding TEs for the five elements measured in this study.
Results
Dynamic bench test
The concentrations of the five elements of the fluids that were pumped through a heated enFlow cartridge were quantified and compared to TE levels of each element based on the FDA’s parenteral PDE values (Table 2). The tests were run for three different challenge solutions at two different flow rates for a total of six test conditions. For runs using the Sterofundin ISO and Plasma-Lyte 148 challenge solutions, concentrations of arsenic, cadmium, copper, and lead were not detected above the MDL. Margin of Safety values for these tests ranged between at least 33 and 882 corresponding to very low toxicological risk. Barium levels were detected above the MDL but were still well below the TE level of 3333 µg L−1. The highest level of barium detected was 48.7 µg L−1with the Sterofundin ISO at a flow rate of 0.2 mL min−1, corresponding to a Margin of Safety of 68.
Dynamic testing analytical results for the concentrations of five elements with three challenge solutions (Sterofundin ISO, Plasma-Lyte 148, and whole blood) at two different flow rates (0.2 and 5.5 mL min−1). If the compound was not detected above the minimum detection limit (MDL), the MDL value for that test is listed. All concentrations are estimates and not blank corrected.
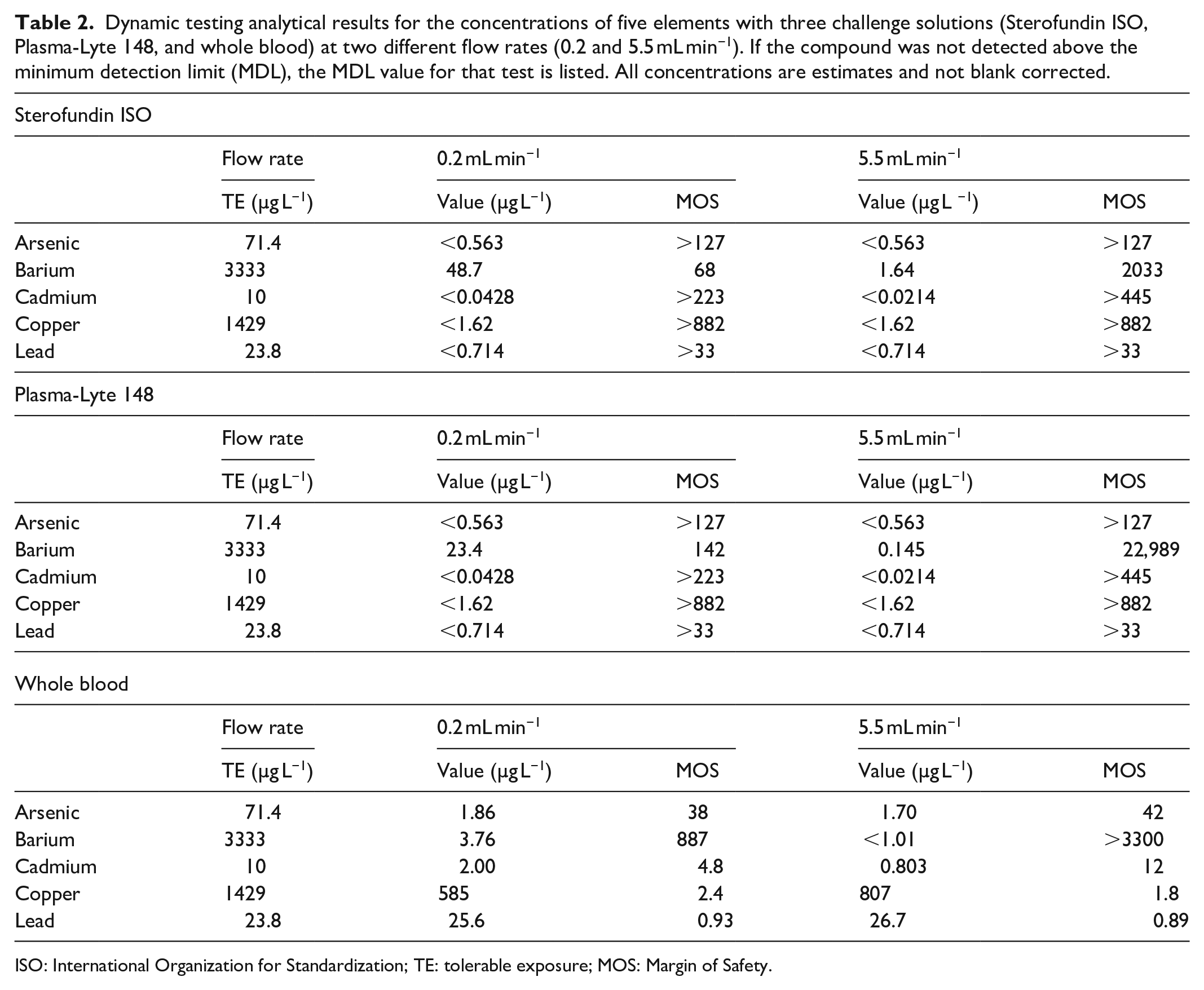
ISO: International Organization for Standardization; TE: tolerable exposure; MOS: Margin of Safety.
For most elements, the highest concentrations were detected when whole blood was used as the challenge solution. Specifically, cadmium concentrations of 2.00 and 0.803 µg L−1 were detected at 0.2 and 5.5 mL min−1 flow rate tests with whole blood, corresponding to Margins of Safety of 4.8 and 12, respectively. Copper concentrations were 585 and 807 µg L−1 for the two flow rates representing Margins of Safety of 2.4 and 1.8, respectively. Finally, we noted lead levels that were slightly above the TE level for infants (defined as 23.8 µg L−1) in the fluids that were flowed at the lower (25.6 µg L−1) and the higher (26.7 µg L−1) flow rates. These concentrations were not blank corrected so that the source of the lead is unclear.
Quasi-static bench test
For the quasi-static tests, the total content of the five elements within the enFlow cartridge were measured after 72 h exposure within a heated cartridge for 16 different challenge solutions. The measured contents were compared to the FDA’s parenteral PDE for each element. The Margin of Safety values were above a value of 1.0 for all five elements for all 16 challenge solutions. Table 3 lists the measured content for the challenge solution which resulted in the maximum content for each of the five elements. For example, the maximum lead concentration was 3.38 µg device−1 (Margin of Safety = 1.5) with the human buffy coat challenge solution. Also included is the TE for each element which was used to calculate the Margins of Safety. Most of the elements had Margins of Safety much higher than 1.0, and lead was the only element with a Margin of Safety less than 55.
Maximum concentration of each element recorded for any of the 16 challenge solutions. All concentrations are estimates and not blank corrected. The parenteral Permitted Daily Exposure (PDE) 18 is listed for each element and used to calculate the worst-case Margin of Safety for that element.

Table 4 lists the complete results from the quasi-static testing for the five elements for all 16 challenge fluids. The maximum concentration for each element across the challenge solutions are indicated in bold. Figures 1–5 show the graphical representation of element extraction by enFlow in each challenge fluid compared to the parenteral PDE as set by the FDA. 18 Figures only include data for levels that were detected above the minimum detection limit (MDL) for that test.





Elements found in challenge IV solutions after warming with enFlow with Margin of Safety compared to parenteral PDE per FDA Q3D guidance document. 18

PDE: Permitted Daily Exposures; USP: United States Pharmacopeia.
The maximum concentration for each element across challenge solutions is indicated in bold. Elements that were not detected above the minimum detection limit (MDL) are marked as “–.”
Discussion
The results of this study found low toxicological risk of exposure to five elements with the use of the parylene-coated enFlow IV/blood warmer when used in both acute and chronic exposures with a wide range of challenge solutions. This study builds upon previous work which found low toxicological risk of aluminum exposure when using the parylene-coated enFlow system. 9 The previous study found a Margin of Safety of at least 1.7 compared to the FDA’s maximum recommended concentration of aluminum in parenteral fluids and also included a pilot animal study which simulated use of the enFlow system in mice and found no evidence of toxicity. 9
Despite their common occurrence in our environment and in industrial and electronic processes, data on mineral leaching from medical devices are scarce. Arsenic is found in food, water, soil, and air, with a common method of exposure being the inhalation of dusts. 35 Islam et al. 36 reported on the arsenic content of certain dental materials. Studies on arsenic trioxide in coronary stents have provided toxicological information for clinicians. 37 This study identified levels of arsenic in multiple fluids that were well below the FDA’s PDE.
Because of its high reactivity and toxicity, barium metal is seldom used in industry. Barium sulfate is radio-opaque and has long been used in radiology as a radiocontrast agent. Some researchers have investigated its use to facilitate visualization of catheters used in uterine artery embolization. 38 Barium titanate is incorporated into piezoelectric circuits within some medical devices. 39 There were negligible amounts in warmed infusate after exposure to the warming cartridge.
Cadmium is a common component in batteries, capacitors, coatings, and pigments. Some implantable devices are powered by cadmium-containing batteries. 40 Electroplating with cadmium and certain semiconductors may be a source of cadmium poisoning. In this study, there were no significant amounts of cadmium identified.
Copper is commonly used in electronics as an electrical conductor in wires and circuit boards. 41 As such, its proximity to the patient increases the chances of toxic exposures. Copper has antimicrobial properties and has been used in medical devices for this purpose. 42 It is commonly used in layering techniques during the manufacturing of medical devices to strengthen them and prevent failure. 43 There were no appreciable levels of copper in the warmed fluids after exposure to the enFlow.
Lead is extremely common in our environment and in manufacturing settings. It is very efficiently absorbed by the human body via ingestion, skin contact, or inhalation, and tends to be stored in bones, leading to bioaccumulation. Because of its ubiquity in water supplies and environmental air, it is not unexpected to find lead in medical devices. For example, lead can be found in dental cements 36 and piezoelectric components, 44 some of which may be found in newer microfluidic pumps used in medical devices. 45
Reported levels of all five elements were significantly higher when blood products were used as the challenge solution compared to saline solutions for both static (see Table 2) and dynamic tests (see Table 3). This observation is potentially explained by high baseline concentrations within the blood products from the donor’s exposures. Blood levels for the five elements reported in this study will, of course, vary from person to person based on their particularly environmental and dietary exposures. Normal blood levels for the elements measured in this study are arsenic: 10 µg L−1; 46 barium in human serum: 1–60 µg L−1; 47 cadmium: 0.5–2.0 µg L−1; 48 copper: 1000–1200 µg L−1; 49 and lead: 10–45 µg L−1. 50 These values are consistent with the concentrations measured in this study for the various blood products.
The reported concentrations of lead for the dynamic tests using whole blood were 25.6 and 26.7 µg L−1 for the two flow rates tested which are slightly higher than the TE for infants of 23.8 µg L−1. However, these concentrations are not blank corrected, and therefore, it is likely that some amount of the lead within the whole blood was present prior to flowing it through the enFlow cartridge. In adults, one study found average whole blood levels of 21 µg L−1 (range = 10–45 µg L−1) in 100 individuals without occupational exposure. 50 These tests were all run against a single donor’s blood, and a measure of these elements in the donor’s blood prior to infusion would have been helpful. These data suggest the whole blood used in this study contained a significant baseline level of lead which contributed to the measured levels after exposure within the enFlow cartridge. Further studies should address this concern by measuring all elements in the blood prior to infusion through the enFlow cartridge.
A potential limitation of this study is the assessment of elements on an element-by-element basis and not as a mixture. Also, insufficient information exists on the toxicological significance of these possible interactions. Extracted compounds from the device may exhibit effects via a common toxicological mechanism of action and may have additive effects. Any assumptions on the toxicological significance of the mixture of compounds extracted from the flow path of the cartridge of the enFlow device are impractical. Concentrations are not blank corrected and might be overestimates if the matrices contained inherent levels of the elements. Despite these limitations, this risk assessment had rigor from its use of an extensive array of challenge fluids, the extended duration of warming periods, the multiple analytic chemistry technologies, and the referencing from multiple extractable chemical characterization studies and published literature on the detected minerals.
Conclusion
The results indicate that observed toxicological risk levels associated with the enFlow for warming blood, blood products, and electrolyte solutions intended for IV use in patients were below those set by the FDA and other regulatory bodies and suggest use of the enFlow with a variety of IV solution types and in different therapeutic scenarios. Further investigations of other minerals as well as organic compounds may provide additional safety information on this device.
Footnotes
Acknowledgements
The authors acknowledge Vyaire Medical, the manufacturer of enFlow, who supported this study.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: D.B., H.G., and A.D.W. are the employees of Vyaire Medical. M.P. is an employee and medical director of Vyaire Medical. E.R. is an independent consultant retained by Vyaire Medical for this project.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.