Abstract
Objectives:
Avoiding inadvertent hypothermia during surgery is important. Intravenous fluid warmers used intraoperatively are critical for maintaining euthermia. We sought to prospectively evaluate the performance of the parylene-coated enFlow™ intravenous fluid warmer in patients undergoing surgery.
Methods:
This was a prospective two-center observational clinical trial performed in inpatient surgical services of two large academic hospital systems. After written informed consent, patients were enrolled in the trial. All patients were adults scheduled for a surgery that was expected to last for at least 1 h with the administration of at least 1 L of fluid warmed prior to infusion. Patient temperature was recorded in the preoperative unit, at the induction of anesthesia, and then every 15 or 30 min until the end of surgery. Temperature monitoring continued in the recovery unit. The parylene-coated enFlow™ intravenous fluid warmer was used in addition to the usual patient warming techniques. The primary outcome was the average core temperature, and secondary analyses assessed individual temperature measurements, temperature measurements during specific time periods, and rate of hypothermic events.
Results:
In all, 50 patients (29 males) with a mean age of 64 years were included in the analysis. The mean surgical time was 195 min and patients received an average of 1142 mL of fluids. Core temperature dropped by only 0.3°C approximately 60 min after induction and recovered back to the baseline level approximately 60 min later. There was no correlation between flow rate and measured core body temperature.
Conclusions:
The parylene-coated enFlow intravenous fluid warmer was able to warm fluids at all flow rates during prolonged surgery. The results showed that enFlow performed as expected.
The parylene-coated enFlow intravenous fluid warmer performed as expected in this prospective two-center clinical trial. Episodes of hypothermia were consistent with those reported for other devices, and there were no hyperthermic events.
Introduction
Perioperative hypothermia is a well-known and well-studied risk factor associated with anesthesia and surgery. Current guidelines support the use of surgical techniques and devices to minimize core temperature fluctuations and avoid hypothermia.1–6 During general anesthesia, normal vasoregulatory mechanisms are blunted, leading to a drop in the core temperature secondary to vasodilation. Hypothermia is more likely in a cold operating room environment, infusion of relatively cold intravenous (IV) fluids, and certain surgeries. 7 Perioperative hypothermia has been associated with delayed post-anesthetic recovery, increased blood loss during surgery with the need for transfusion, increased incidence of surgical wound infections, impairment of antibody- and cell-mediated immune defenses, and increased postoperative adverse myocardial events.8–13 To reduce these events, current techniques to maintain normothermia include draping and covering the patient during surgery, active heating with forced air warming devices or heated water underbody pads, use of heated irrigation fluids in major body cavities, and warming of IV fluids as they are infused. 14 In particular, warmed IV fluids are used for avoidance of intraoperative hypothermia. Studies have shown that patients managed using fluid warmers maintained core body temperatures of 0.41–0.74 °C warmer using fluid warmers compared with other standard controls throughout the operative procedure with a relative risk of postoperative shivering of 0.39. 14
The enFlow® (Vyaire Medical, Mettawa, IL, USA) is an active IV fluid warming system that attaches in series to the IV infusion line, which has a cartridge capable of heating fluids as they are infused into the patient (see Figure 1). The enFlow cartridge recently underwent recall, redesign, and re-release secondary to concerns regarding the release of aluminum and other elements into the infusate. 15 The newer cartridge has been extensively tested in a bench environment for the release of aluminum, other potentially toxic elements, and organic molecules with favorable results.16–22 Additional bench testing also confirmed that the newer parylene-coated enFlow design warmed fluids as efficiently as the previous design; however, the performance of the parylene-coated enFlow has not been evaluated in a clinical setting. 16

The enFlow™ cartridge.
To further assess this improved design in a clinical setting, this trial evaluated the efficacy of the new parylene-coated enFlow IV fluid and blood warming system in maintaining normothermia in patients undergoing surgery. Our hypothesis was that the parylene-coated enFlow cartridge would provide adequate warming of the infusate during general anesthesia in adult patients for surgeries lasting over an hour and for patients receiving at least 1 L of fluid. In this observational trial, we evaluated the heating performance of the new enFlow at different flow rates and surgery durations. The primary outcome measure will be the mean of the temperature measurements taken during the course of surgery.
Methods
Ethics
Ethical approval for this trial was provided by the Ethics Committee of Helsinki and Uusimaa Hospital District (HUS/3206/2020) and the HUS Ethical Commission III, Helsinki, Finland (Chairperson Lauri Tammilehto) on 11 January 2021. The committee of 13 participants reviewed the clinical investigation plan, including amendments, the participant information leaflet, and written informed consent. This trial conforms to the Strengthening the Reporting of Observational Studies in Epidemiology standard for observational studies.23,24 This trial was conducted in accordance with the ethical principles in the Declaration of Helsinki. Written informed consent was obtained from all subjects participating in the trial prior to their participation. The trial was registered prior to patient enrollment at clinicaltrials.gov (NCT04709627, Principal investigator: Mikko Lax, MD, Date of registration: 14/01/2021.)
Trial format
Subjects with scheduled surgeries were recruited for enrollment in the trial consecutively from patients undergoing general anesthesia. The enrollment period was June 2021 through February 2022, when the number of qualifying cases was attained. Patients were enrolled if they were 18 years of age or older and required surgery for at least 1 h. Other inclusion criteria included an expectation of the perioperative need for at least 1 L of infused fluids warmed during infusion, and the ability and willingness to provide written informed consent. Exclusion criteria included pregnancy, a recent history of fever, or if the attending physician did not believe that participation of the patient would be in their best interest.
This was a noninterventional observational trial. Participants were managed according to the routine care protocols of physicians and hospitals, receiving fluids warmed by the parylene-coated enFlow device. The device is packaged with a 7.5 cm extension set that is then connected via a 3-way stopcock connector to the patient (10 cm in length). Core temperature was monitored using a SpotOn™ forehead temperature monitor (3M Healthcare Division, St. Paul, MN, USA). For patients that had a urinary tract catheter placed for therapeutic reasons, bladder temperature measurements were also recorded at the same time points using the temperature sensor on the catheter. Surgical patients were also warmed using forced air warming, warming blankets, and control of the operating room temperature according to the hospital’s usual protocol. During the investigation period, a research operator acquired temperature measurements and collected data on the surgical and warming practices. Skin temperature measurements were taken at baseline (i.e., 15–0 min prior to induction), 15, 30, 60, and 90 min post-induction, and then every 15 min until the end of surgery. A final measurement in the operating room was recorded at the end of surgery, followed by a reading upon arrival to the post-anesthesia care unit (PACU) and 30 min after arrival to the PACU. The level of and duration of high or low infusate temperatures were recorded for all patients. The levels of unacceptable infusate temperatures include the following: “Flashing Blue”: <33°C; “Solid Blue”: ⩾33°C and <35°C; “Solid Yellow”: >42°C and ⩽45°C; and “Flashing Red”: >45°C.
This trial was conducted at two hospital sites, Department of Anesthesiology, Helsinki and Uusimaa Hospital District, Helsinki University Hospital, Helsinki, Finland, and Department of Anesthesiology, South Karelia Central Hospital, Lappeenranta, Finland. The clinical data were recorded using an electronic data capture system and transferred to the research administrator for analysis. No treatment or control group analysis was performed, as non-use of an IV fluid warmer (i.e., a control group) would be contrary to these hospitals’ usual care protocols. In particular, warmed IV fluids are used for avoidance of intraoperative hypothermia. In the two hospitals where the clinical study was conducted using IV was standard of care.
Statistical analysis
Power analysis and sample size determination
The sample size for this trial was determined based on the primary outcome of the average core temperature during surgery. We reviewed the related Cochrane Collaboration report of 209 subjects from nine studies with standard care practices. 14 We concluded that the core temperature during surgery could be expected to be maintained at 36.4°C (standard deviation (SD) 0.6°C). Assuming the same mean temperature and variation in this investigation, with 42 subjects, there would be a 99% chance that the control of the mean core temperature would be between 36.1 and 36.7°C. Our review also found that the mean core temperature (99% confident interval) among the studies was between 36.0 and 36.9°C, which is wider than the expected results of our investigation. Thus, our investigation was reasonably powered with 42 participants. To investigate the performance during longer surgeries, the protocol specified a total investigation size of 50 subjects, with at least 42 completing at least 60 min of surgery, and at least 12 subjects completed at least 120 min of surgery.
Data and statistical analyses
The primary end point for each subject was the mean and SD of the core temperature SpotOn measurements taken during the surgery. To compare the performance of the enFlow device in this trial with previous studies, a noninferiority test was conducted to compare the mean patient temperature during surgery with the 209 patients compiled from nine studies in the Cochrane Collaboration report.
14
This test evaluated the null hypothesis that the difference in mean temperature in this trial (µ1) and the mean temperature from the Cochrane report (µ2 = 36.4°C) is less than or equal to opposite of a predefined noninferiority margin (
To visualize how temperature changed over the course of a surgery, the mean and 99% confidence interval temperature across patients were calculated at each time point after the start of induction. Time points with less than 50% of the population were not included in the visualization. A repeated measures analysis of variance (ANOVA) with Dunnett’s test 27 was conducted to compare mean temperatures at each time point to the baseline temperature.
The trial was subdivided into four predefined study periods, and the mean and SD of measurements during periods were calculated: Baseline (i.e., 15–0 min prior to induction), induction period (i.e., 15, 30, 60, and 90 min after start of induction), and post-induction (i.e., additional measurements taken after 90 min after start of induction). A repeated measures ANOVA was performed to compare the mean temperatures at baseline, induction, post-induction, and PACU periods. Multiple comparison post-hoc tests were conducted for pairwise comparisons between periods if the ANOVA identified a significant difference between periods.
Hypothermic and hyperthermic events were identified for each patient during the entire surgery as well as separately during the induction and post-induction periods. Hypothermia was defined as a patient’s temperature less than 36.0°C and hyperthermia as a temperature greater than 38.0°C. We quantified hypothermic and hyperthermic events using two different methods. The first method counted the total number of individual temperature measurements which were within the hypothermic or hyperthermic temperature ranges. The second method grouped hypothermic and hyperthermic episodes as continuous time periods where the temperature was within the hypothermic or hyperthermic ranges. These episodes were classified based on which study period they started in, and the duration of each event was calculated. The rate of these episodes was calculated as the number of episodes divided by the period duration. Finally, we quantified the proportion of patients that experienced at least one of these events during that study period.
To examine the relationship between the infusate flow rate and patient temperature, we examined the correlations between IV fluid flow rates and patient temperatures for measurements recorded during surgery. Robust linear regressions with bisquare weight functions and outliers removed (i.e., points greater than three SDs from the mean) were run for each relationship. For flow rates, we looked at the maximum and mean flow rates for each patient. For patient temperature, we included the minimum and mean temperatures during surgery for each patient. Furthermore, the same regressions were conducted looking at minimum and mean temperature versus change in patient temperature from their baseline temperature. For these regressions, we included the largest decrease in temperature during surgery (i.e., minimum temperature—baseline) and the mean change in temperature during surgery for each patient.
For patients with bladder temperature measurements, a Bland–Altman analysis was conducted to compare time-aligned temperature measurement pairs from the SpotOn system with bladder temperature measurements.
For a subset of patients, a second fluid heater was used in parallel with the enFlow fluid warmer (HOTLINE Blood and Fluid Warmer or Level 1 H-1200 Fast Flow Fluid Warmer). To explore the effect of the second fluid heater, we compared the results of patients whose fluids were exclusively heated with the enFlow fluid warmer (“enFlow Only”) with the patients who had a second fluid warmer (“Two Warmers”). Categorical data were compared using Fisher’s exact tests or chi-squared tests, and continuous data were compared using two-sample t-tests for normally distributed data and Wilcoxon rank sum tests otherwise.
To account for multiple comparisons, we used a significance level of α = 0.005 for each statistical test to maintain an overall type I error of 5%.
Results
A total of 54 patients were recruited for the trial. Of the 54 patients, data from four patients were excluded because they did not meet the inclusion criteria. Therefore, 50 patients (29 males) completed the trial and were included in the analysis. The mean surgical time was 195 min (range: 64–434) with all 50 included subjects having a surgical duration of at least 60 min and 38 subjects having a surgery lasting at least 120 min. Surgery started an average of 55 (SD: 11) min after the start of induction. The mean age of the patients was 64 years (SD: 12) with a mean body mass index (BMI) of 27.0 kg/m2 (SD: 5.7). The demographic characteristics of the patients and their summary statistics are shown in Table 1. The average ambient temperature in the surgical suites was 21.5°C (SD: 1.0°C) at baseline and 21.8°C (SD: 1.1°C) at the end of surgery. The infusate temperature remained in the acceptable operating range (i.e., 35–42°C) for all patients and all times.
Demographic characteristics and summary of results (N = 50 subjects).

ASA, American Society of Anesthesiologists; BMI, body mass index.
Across the entire duration of surgery, patients received a mean total infusion volume of 1142 mL (SD: 792) at an average rate of 343 mL/h (SD: 125) (Table 2).
Infusion flow rates and total volume infused during surgery (N = 50 subjects).

As part of perioperative care, all patients were also warmed with one or more other additional methods of warming including forced-air patient warming, heated mattress, and alternative fluid warmers (Table 3).
Forced-air patient warming (3M Bair Hugger System); heated mattress (Astopad); fluid warmer (HOTLINE Blood and Fluid Warmer or Level 1 H-1200 Fast Flow Fluid Warmer) (N = 50 subjects).

During surgery, the mean temperature across all patients was 36.5 (99% confidence interval: 36.4°C, 36.7°C). A noninferiority test established noninferiority of the enFlow to maintain patient temperature during surgery compared to previous clinical studies 14 with a noninferiority margin (δ) of 0.23°C. Specifically, the 99% confidence interval of the difference between the mean temperature in this trial and those compiled from previous clinical studies was −0.070°C to 0.30°C. Noninferiority was established because the lower limit of this confidence interval is greater than −δ (i.e., −0.070 > −0.23).
Figure 2 shows the mean and 99% confidence intervals for SpotOn temperature measurements obtained before, during, and after the surgery. There were statistically significant decreases in the core temperature from baseline at 30, 60, 90, and 105 min after the start of induction (p ⩽ 0.0002). Temperature reached a minimum at 60 and 90 min after the start of induction, with a temperature of 0.3°C below the baseline temperature. Core temperature then recovered back to the baseline temperature for the remainder of the surgery and into the PACU.
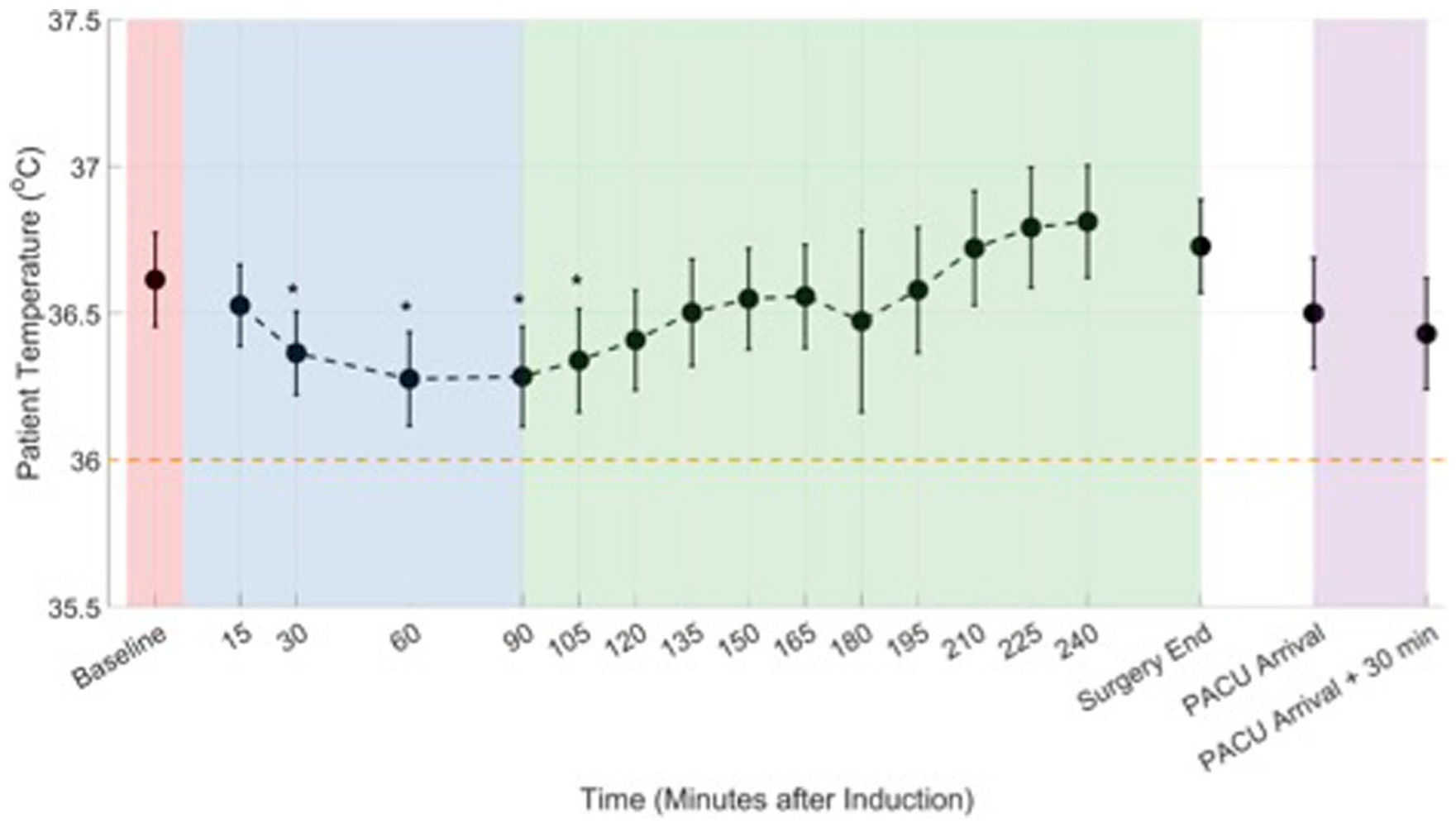
Patient temperature before surgery (baseline, shaded red), induction (shaded blue), post-induction (shaded green), and in the PACU (shaded purple). The threshold for hypothermic events (i.e., 36°C) is also shown (horizontal dashed orange line). Data are presented as mean (marker) and 99% confidence interval (error bars).
Table 4 summarizes patient temperatures for each of the four study periods. Patient temperature dropped significant from baseline (36.6°C) during the induction phase (36.4°C, p = 0.0001) and then rebounded back during the post-induction phase (36.6°C, p = 0.78) which continued into the PACU (36.5°C, p = 0.041).
Descriptive statistics of average patient temperatures during four study periods.

PACU, post-anesthesia care unit.
There were 819 SpotOn temperature readings recorded during this trial. Of the 819 temperature readings recorded, there were 81 (10%) below the hypothermic threshold of 36.0°C and zero were above the hyperthermic threshold of 38.0°C (Table 5). The minimum single temperature measurement during surgery was 35.1°C, and the maximum single temperature measurement was 37.5°C.
Number of hypothermic and hyperthermic temperature measurements by study period.

Grouping consecutive temperature measurements below the hypothermic threshold into continuous episodes, a total of 19 hypothermic episodes were experienced in 16 (32%) patients. In all, 13 of these episodes started during the induction study phase while six started in the post-induction study phase. During surgery, patients experienced an average of 0.09 hypothermic episodes per hour (i.e., one episode every 11.1 h) (Table 6). These hypothermic events lasted for a median duration of 15 min with a minimum duration of 0 min (i.e., single temperature measurement) and maximum duration of 210 min.
Summary population statistics of hypothermic episodes commencing during the induction and post-induction periods.

To examine the relationship between infusate flow rate and patient temperature, we ran linear regressions between IV fluid flow rates and patient temperatures for measurements recorded during surgery (Figure 3). The linear regressions revealed no association between fluid flow rates and patient temperature (p ⩾ 0.37).

Correlations between fluid flow rate and patient temperatures. Each circle represents a single patient, with outliers indicated as open circles. Solid lines represent linear regressions. No significant associations were found.
As an additional analysis, we explored the correlation between IV fluid flow rate and change in patient temperature from their baseline temperature recorded prior to surgery (Figure 4). These linear regressions also revealed no association between fluid flow rates and change in temperature from baseline (p ⩾ 0.12).
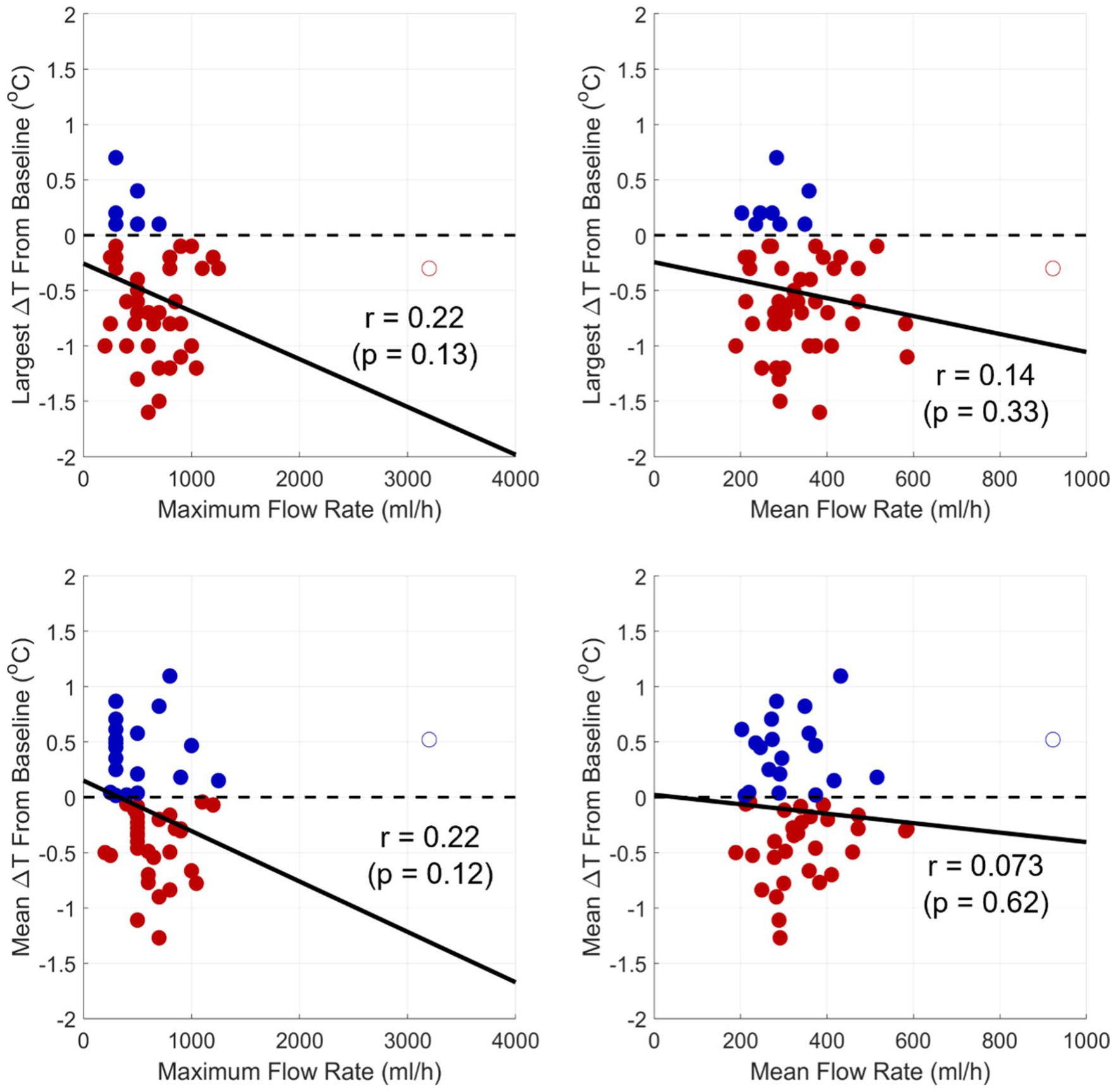
Correlations between fluid flow rate and patient temperatures. Each circle represents a single patient, with outliers indicated as open circles. No temperature change is shown as a horizontal dashed line. Decreases in temperature are shown as red markers and increases as blue markers. Solid lines represent linear regressions. No significant associations were found.
The Bland–Altman analysis comparing time-coincident temperature measurements by the SpotOn and bladder temperature patients showed good agreement between the two measurement techniques (Figure 5). SpotOn temperatures were on average 0.21°C higher (95% limits of agreement: −0.79°C to 1.19°C) than bladder temperature measurements for the 400 measurement pairs from the 22 patients with a urinary tract catheter.

Bland–Altman plot comparing non-invasive temperature measurements (SpotOn) and bladder temperature measurements for patients with a urinary tract catheter (N = 22 patients). Each marker represents a pair of temperature measurements (400 measurement pairs) coincident in time. Measurements from the same patient are represented by a marker of the same color and symbol. Solid horizontal line represents the mean bias and dashed lines are the 95% limits of agreement (LoA).
As shown in Table 3, eight patients had fluids warmed with a secondary fluid warmer in conjunction with the enFlow fluid warmer. We performed subgroup analyses to compare the results of patients whose fluids were heated with only the enFlow (enFlow Only, N = 42) and patients with a second fluid warmer (Two Warmers, N = 8). Patient characteristics and ambient temperatures listed for the entire population in Table 1 were not significantly different for the two subgroups (p ⩾ 0.05). Infusion flow rates listed for the entire population in Table 2 were also not significantly different between the subgroups (p ⩾ 0.029). For example, mean infusion rates were 357 and 272 mL/min for the enFlow Only and Two Warmers subgroups, respectively (p = 0.029). Furthermore, other patient heating methods were not significantly different between the two subgroups. Specifically, a similar percentage of patients in the two subgroups had forced air patient warming (98% versus 88%, p = 0.18) and a heated mattress (81% versus 63%, p = 0.25). The mean durations of these heating methods were also not significantly different between the two subgroups (p ⩾ 0.85). The primary end point of the study (i.e., mean temperatures during surgery) was not significantly different between the enFlow Only subgroup (36.5°C ± 0.4°C) and the Two Warmers subgroup (36.7°C ± 0.4°C, p = 0.22). Noninferiority tests performed with data from the subgroups established noninferiority of the warmers used in this study compared to previous clinical studies 14 with a noninferiority margin (δ) of 0.23°C. The 99% confidence intervals for the differences between this study and previous clinical studies were (−0.11°C, 0.28°C) and (−0.18°C, 0.71°C) for the enFlow Only and Two Warmers subgroups, respectively. The two subgroups did not have significantly different hypothermia episode frequencies during surgeries (0.10 versus 0.03 episodes per hour, p = 0.23). Finally, the percentage of patients which experienced at least one hypothermia episode (36% versus 13%) was not significantly different (p = 0.41).
Two adverse events were identified through the trial, both unrelated to the study device. Specifically, one consented patient was tachycardic with weakening vitals and the procedure was canceled. In the second patient, the cannula perforated the patient’s vein, resulting in local infiltration. The devices were used within the indications stipulated in manufacturer’s instructions for use. 28
Discussion
In this clinical observational trial, core patient temperature dropped by an average of 0.3°C approximately 60 min after the start of induction of general anesthesia for patients receiving warmed flows with the enFlow fluid warmer. Patient temperature returned to baseline levels approximately 60 min later. This magnitude drop in temperature is consistent with prior studies and noninferiority was established compared with compiled results from nine clinical studies. 14 In a study from 1993 using healthy volunteers undergoing general anesthesia without the use of active warming techniques or application of heating blankets, core body temperature decreased by approximately 1.5°C within 1 h. 10 A drop in core body temperature to this extent has been shown to lead to significant adverse clinical events, including a twofold increase in cardiac events, 8 delayed wound healing with increased risk of infection, 11 and an increase in surgical blood loss and use of perioperative blood product transfusions, even with smaller drops in core temperature.12,29 In a Cochrane Review of the effect of IV fluid warmers, Campbell et al. 14 found that, with a fluid warmer, core temperature was about half a degree higher compared to patients not given warmed fluids. This suggests a typical decrease in core temperature of 1.0°C using other methods of warming fluids. 14 The results of this trial show an average decrease in core temperature of only 0.3°C (Figure 2), indicating success in mitigating the risk of incidental hypothermia associated with surgery and anesthesia. Only 32% of patients experienced any temperatures below the hypothermic threshold (Table 6). Furthermore, these episodes typically quickly resolved with a median duration of 15 min.
The exact degree to which warmed IV fluids prevent hypothermia or warm patient core temperatures is subject to discussion, but several researchers have attempted to quantify such therapies. Sari et al. demonstrated that up to 79% of patients experienced incidental hypothermia when no active fluid-warming systems were employed, with a significant increase in postoperative shivering in patients who experienced intra-operative hypothermia. 30 While these results are particularly important during high-risk abdominal and cardiac surgery, they also translate into lesser-risk ambulatory and sedation procedures, even when more modest infusion volumes are expected (<1 L). Kim et al. evaluated ambulatory urologic procedures under monitored anesthesia care and showed a significantly reduced drop in core temperature after induction, at the end of the procedure, and in recovery compared with patients who did not receive warmed IV fluids (p = 0.004, p = 0.02 and p = 0.008, respectively). 31 Hasankhani showed similar results in ambulatory orthopedic surgery, where the final core temperature averaged 36.4°C when IV fluids were warmed compared to 35.9°C in controls (p = 0.001). 32 This aligns with the results of a meta-analysis of literature covering the use of warmed IV fluids during anesthesia, showing a mean core temperature of 0.41–0.79°C warmer in patients who received warmed IV fluids compared to controls. 14
Active fluid warming systems have significant limitations in the amount of heating they can produce alone, as there is a ceiling limit on how warm the infusate can be without causing cellular or organ injury. As such, it is agreed that the main contribution of active fluid warming perioperatively is to reduce the effects of heat loss to the environment rather than true heating of a hypothermic patient, converse to active cooling of a patient where infusate temperatures may be given at 30°C–40°C lower than body temperature. 33 Mathematical thermodynamic models have been created to predict core temperature variations based on the temperature of the infusate and the baseline temperature of the patient. Barthel and Blumenberg separately designed models that predict that the average adult core temperature is likely to decrease by approximately 0.3°C–0.4°C with every liter of room temperature IV fluid, whereas the rise in core temperature using the same volume of warmed infusate is calculated at 0.1°C.33,34
Our results corroborated these predicted outcomes. Not only did the patients in this trial demonstrate a less significant decrease in core temperature after induction of anesthesia and throughout surgery compared with previously published data of non-warmed IV fluids, 14 but the degree to which temperature dropped in these patients mirrors data previously described with other active fluid warming devices 35 and in the proposed mathematical models. 33
Overall, patients in this trial experienced a modest decrease in core temperature compared to baseline upon induction of anesthesia (approximately 0.3°C), with recovery of core temperature back to baseline within 3 h. Compared with other published data and mathematical models predicting patient body temperature changes with infused fluids, our results suggest that the enFlow fluid warming system was successful in reducing the degree of change in body temperature associated with anesthesia and surgery and the incidence of hypothermia during anesthesia, along with a more rapid return of core body temperature to baseline.
This trial has several limitations. This was an uncontrolled and unblinded study design and there was no control group of patients not receiving warmed IV fluids. Such intraoperative care would deviate from our own policies of using fluid warmers in these cases. Further studies may include a control group from another surgical site that did not require the use of fluid warmers. This was also not a comparison study to see if enFlow with its parylene coating worked better than any other IV fluid warmer, so there was no comparator group. All patients were co-managed with force-air warming and warming blankets, measures which were not controlled by the research protocol. Clearly, these measures contributed to the avoidance of hypothermia. However, the previously mentioned studies also utilized ancillary warming devices but without IV fluid warming, and marked decreases in core body temperature were measured. In addition, Campbell et al. showed that warmed IV fluids versus room temperature IV fluids increased core temperature by 0.5°C independent from other temperature management factors. 14 Eight patients included in the study had a secondary fluid warmer which was used in conjunction with the enFlow fluid warmer. However, subgroup analyses demonstrated the patient characteristics, patient temperatures, and hypothermia frequencies were not significantly different between patients with and without a secondary fluid warmer. Furthermore, a noninferiority tests performed on the subgroup of patients whose fluids were heated exclusively with the enFlow fluid warmer established noninferiority compared with previous clinical studies. Core temperature in this trial was estimated using the SpotOn temperature measurement system. This measurement technique has been validated against pulmonary artery catheter measurements with a mean error of −0.23°C. 25 Furthermore, we showed good agreement between SpotOn temperatures and bladder temperature measurements in the subset of patients with a unitary tract catheter (Figure 5). We considered adding temperature sensors at each end of the warming cartridge to evaluate the impact of the device. However, the specifications of the device state that the output fluid temperature is 40°C ± 2°C. The enFlow heater has a temperature sensor which monitors the output fluid temperature and alarms if it is outside of the range. This project did not seek to confirm that the device performs within the technical specifications. Finally, the trial was not adequately powered to fully clarify overall safety of the device nor the correlation between infusate flow rate and core body temperature.
Combined with previous laboratory-based bench studies demonstrating the efficacy of the newer-designed enFlow system compared to its historical design in warm-infused fluids, this trial provides substantial evidence that the parylene-coated enFlow system operates as intended in living patients undergoing surgery and anesthesia and should be considered when actively warming patients at risk for hypothermia.
Conclusions
In this prospective multicenter trial, the newly designed parylene-coated enFlow IV fluid warming system provided active warming to patients undergoing surgery and anesthesia consistent with previous studies of similar devices across a wide range of infusate flow rates. Even with the new parylene coating on the heating blocks, the enFlow system performs as intended.
Supplemental Material
sj-docx-1-smo-10.1177_20503121231182517 – Supplemental material for Verification of an intravenous fluid warmer: A prospective, two-center observational trial
Supplemental material, sj-docx-1-smo-10.1177_20503121231182517 for Verification of an intravenous fluid warmer: A prospective, two-center observational trial by Mikko Lax, Seppo T. Mustola, Kimmo Repo, Jari Järvinen, Danielle K. Bayoro, Steven H. Cataldo, Viivi Karhinen, Edward A. Rose, Herman Groepenhoff and Andreas D. Waldmann in SAGE Open Medicine
Footnotes
Acknowledgements
None.
Authors’ contributions
ML, SM, and AW contributed to design of the work, drafting the manuscript, and final approval of the version to be published, and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. KR, JJ, DB, VK, SC, ER, and HG contributed to the collection and analysis of the data, drafting the manuscript, and final approval of the version to be published, and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ML, SM, KR, JJ, and VK have no competing interests. The actual research including case extraction and data analysis were performed by an outside clinical research organization, Clinius Oy (VK). DB, ER, HG, and AW are affiliated with Vyaire Medical. SC is an independent consultant. This manuscript and all attached tables and figures were reviewed and approved by all authors.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
Ethical approval for this trial was provided by the Ethics Committee of Helsinki and Uusimaa Hospital District (HUS/3206/2020) and the HUS Ethical Commission III, Helsinki, Finland (Chairperson Lauri Tammilehto) on 11 January 2021. The committee of 13 participants reviewed the clinical investigation plan, including amendments, the participant information leaflet, and written informed consent. This trial conforms to the STROBE statement. This trial was conducted in accordance with the ethical principles in the Declaration of Helsinki. Written informed consent was obtained from all subjects prior to participating in the trial.
Ethics approval
Ethical approval for this study was obtained from Ethics Committee of Helsinki and Uusimaa Hospital District (HUS/3206/2020) and the HUS Ethical Commission III, Helsinki, Finland.
Informed consent
Written informed consent was obtained from all subjects before the study.
Consent for publication
Not applicable.
Trial registration
The trial was registered prior to patient enrollment at clinicaltrials.gov (NCT04709627, Principal investigator: Mikko Lax, MD, Date of registration: 14 January 2021.)
Availability of data and materials
The datasets used and/or analyzed during the current trial are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
