Abstract
Purpose:
This study aimed to investigate the pathogenic characteristics of Vibrio parahaemolyticus in Taizhou City, China, to provide a scientific basis for the diagnosis and control of Vibrio parahaemolyticus in the region.
Methods:
Bacterial strains were isolated from stool specimens of diarrheal patients at the First People’s Hospital of Wenling City. The virulence genes, serotypes, and antibiotic susceptibility of these strains were determined.
Results:
From June 1, 2020 to May 31, 2023, a total of 97 strains of Vibrio parahaemolyticus were isolated from stool specimens of diarrheal patients at the First People’s Hospital of Wenling City. The results of virulence gene detection showed that 87.6% of the strains were thermostable direct hemolysin (TDH; tdh)+, thermolabile hemolysin (tlh)+, and tdh-related hemolysin (trh)−, 11.3% were tdh− tlh+ trh−, and 1.0% were tdh+ tlh+ trh+. Six different serotypes were identified, with O3:K4 and O4:K4 being the most common, accounting for 42.3% and 32.0%, respectively. The other serotypes identified were O3:K6, O4:K8, and O1:KUT, accounting for 11.3%, 10.3%, and 3.1%, respectively. Furthermore, the results of antimicrobial susceptibility tests showed that most strains were sensitive to the tested drugs, while some strains exhibited resistance to cefuroxime (32.6%) and cefazolin (43.6%).
Conclusion:
The majority of the isolated Vibrio parahaemolyticus strains carried the tdh gene, and the O3:K4 serotype dominated in Taizhou City from 2020 to 2023, followed by the O4:K4 serotype. Furthermore, some strains exhibited resistance to cefuroxime and cefazolin.
Introduction
Vibrio parahaemolyticus is a halophilic gram-negative bacterium widely distributed in temperate and tropical marine environments and coastal waters worldwide, often associated with foodborne gastroenteritis.1,2 Outbreaks of V. parahaemolyticus have occurred globally, significantly increasing the medical burden.2,3 However, not all V. parahaemolyticus strains are pathogenic. 4 The tdh or tdh-related hemolysin (trh) is considered the major virulence factor of V. parahaemolyticus and plays a crucial role in V. parahaemolyticus-induced gastroenteritis.5,6 Studies have shown that most clinical isolates of V. parahaemolyticus are associated with strains carrying tdh and/or trh.7–9 Infections related to V. parahaemolyticus are caused by multiple serotypes, and their pathogenicity varies with the serotype. Since 1996, the O3:K6 serotype has started to spread widely and has dominated globally. 10 Subsequently, serotypes have been continuously evolving, and other dominant strains with epidemic potential have emerged in different regions, even replacing the position of O3:K6,2,11,12 Interestingly, pandemic serovariants such as O1:KUT, O1:K25, and O4:K68 exhibit the same genetic and molecular characteristics as pandemic strains, still carrying markers such as tdh+ and toxRS/new+. 13 In Beijing, China, O10:K4 was detected as the dominant serotype in 2021. However, all 64 tdh+ strains belonged to the pandemic clone, carrying the tdh+ trh− toxRS/new+ markers. 14 In general, for severe cases of V. parahaemolyticus infection, antibiotics are usually used for treatment. However, the overuse of antibiotics in human and aquaculture systems has led to the emergence of multidrug-resistant strains of V. parahaemolyticus, including resistance to antibiotics such as ampicillin (ABPC).2,15,16 During the period from January 2017 to December 2021, Zhang et al. 17 detected 598 positive samples of V. parahaemolyticus (5.36%) from 11,166 fecal specimens in Huzhou, China, with 86.8% of the isolated strains being resistant to ABPC. Xie et al. 18 evaluated 900 food samples collected from China between 2017 and 2019, and identified 101 strains of V. parahaemolyticus. The results indicated that most of the isolated strains from aquatic products were resistant to aminoglycoside drugs and exhibited multidrug resistance. These data highlight the need for monitoring and controlling the spread of drug-resistant V. parahaemolyticus in clinical and environmental samples. Therefore, understanding the pathogenic characteristics of V. parahaemolyticus and timely surveillance of antibiotic-resistant strains are crucial for disease control.
Currently, V. parahaemolyticus has become a major cause of seafood-related gastroenteritis worldwide.1,19 In China, particularly in coastal areas, contamination of seafood products with V. parahaemolyticus is frequently reported.2,11,17 Between 2017 and 2019, the results of testing 900 market samples for V. parahaemolyticus showed that a total of 101 samples (11.22%) tested positive. Looking at the food categories, the positivity rates for ready-to-eat foods, shrimp, and fish were 3.67% (11 samples), 19.33% (58 samples), and 10.67% (32 samples) respectively. Yearly data indicated an increasing trend in contamination rates from 2017 to 2019, with rates of 10.33%, 11.00%, and 12.33% respectively. The testing revealed that the concentration of V. parahaemolyticus in the samples ranged from 1.50 to 100 MPN/g. 18 Taizhou is a prefecture-level city located in the southeast coastal region of China. Due to its active production and consumption of seafood products, the incidence of V. parahaemolyticus infections is high in Taizhou. However, research on V. parahaemolyticus in Taizhou is still limited. Given the genetic variability of V. parahaemolyticus, a comprehensive understanding of the pathogenicity of local strains is of significant importance for disease control and prevention.
Therefore, the aim of this study is to analyze the pathogenic characteristics of V. parahaemolyticus in the Taizhou region, providing essential insights for the diagnosis, treatment, and prevention of V. parahaemolyticus in this area. Additionally, this study contributes the latest information to V. parahaemolyticus-related research in the southeast coastal region of China.
Materials and methods
Clinical sample collection and V. parahaemolyticus cultivation
This study is an epidemiological investigation based on a specific region (Taizhou, China) and time period (2020–2023), in which clinical data were collected from 97 cases of diarrheal patients at the First People’s Hospital of Wenling City, Taizhou, Zhejiang Province, China, from June 1, 2020 to May 31, 2023. The collection of clinical samples was approved by the Ethics Committee of the Wenling first people’s Hospital. The approval number is KY-2020-1012-01. All patients were given detailed information and signed informed consent forms. The collected clinical information included white blood cell (WBC), C-reactive protein (CRP) levels, and fecal occult blood. A total of 97 strains of V. parahaemolyticus were isolated from patient stool specimens.
Isolation, identification, and culture of V. parahaemolyticus: Patient samples are directly streaked onto Thiosulfate Citrate Bile Salts Sucrose Agar (TCBS) selective medium and simultaneously inoculated into alkaline peptone water tubes. Incubate at 37°C overnight, then use an inoculating loop to streak the enriched broth onto TCBS selective medium and incubate at 37°C for 24 h. Suspicious colonies are picked and streaked onto nutrient agar plates for purification. Initial screening is done using oxidase and salted gelatin, retaining strains that are oxidase-positive and do not grow in salted gelatin for further concentration screening in semi-solid medium. Further screening involves sucrose and glucose gas production, 6% NaCl gelatin, and Arabic gum, with strains positive for Arabic gum, negative for glucose gas production, negative for sucrose, and growing in 6% NaCl gelatin subjected to 20E biochemical system identification to differentiate from other Vibrios and pseudomonads.
This study collected clinical samples from a specific region (Taizhou, China) and timeframe (2020–2023) to analyze the distribution and epidemiological characteristics of V. parahaemolyticus, constituting an epidemiological investigation.
Inclusion criteria: Patients who presented with acute diarrhea; V. parahaemolyticus was successfully isolated and confirmed from their stool specimens.
Exclusion criteria: Patients whose diarrhea lasted more than 14 days, or who had a history of chronic intestinal diseases; patients who had used antibiotics within 48 h prior to sample collection.
Virulence factors analysis
Genomic DNA from the 97 strains of V. parahaemolyticus was extracted using the QIAamp DNA Mini Kit (Qiagen, Germany). Templates were prepared, and Polymerase Chain Reaction (PCR) were set up using the 2× Taq PCR Master Mix (Tiangen, China) in the Veriti 96-Well Thermal Cycler (Applied Biosystems, USA). The PCR reaction conditions are as follows: 37°C for 2 min, 94°C for 2 min, one cycle, followed by 93°C for 15 s, 60°C for 60 s, repeated for 45 cycles. After electrophoresis on a 1% agarose gel containing GelRed (Biotium, USA), the gel images were captured using a gel imaging system. The sequences used (5ʹ to 3ʹ) are as follows 20 : Tlh, F-ACTCAACACAAGAAGAGATCGACAA, R-GATGAGCGGTTGATGTCCAA; Tdh, F-TCCCTTTTCCTGCCCCC; R-CGCTGCCATTGTATAGTCTTTATC; Trh: F-TTGCTTTCAGTTTGCTATTGGCT; R-TGTTTACCGTCATATAGGCGCTT.
Serotyping
Serotyping was performed using the agglutination test. Overnight cultures of the bacterial strains were added to 3% sterile physiological saline to prepare a uniform bacterial suspension. Initially, a polyvalent K antiserum was used for detection, and if agglutination reaction occurred, individual antisera were then used for further testing. Furthermore, another aliquot of the aforementioned bacterial suspension was transferred to a centrifuge tube. After being subjected to high pressure at 121°C for 1 h, the resulting lysate was used for O antigen identification. The serotype was represented by the combination of O antigen type and K antigen type (O:K). K antigen that could not be typed was denoted as untypeable (UT). V. parahaemolyticus O diagnostic antiserum (YDTJ331, Shanghai Yuduo Biotechnology Co., Ltd., China) and V. parahaemolyticus K diagnostic antiserum (JL0004, Denka Seiken, Japan) were used for testing.
Antimicrobial susceptibility tests
Drug susceptibility of the isolated 97 strains of V. parahaemolyticus was determined using the Kirby-Bauer disc diffusion method. 21 The results were interpreted according to the Clinical and Laboratory Standards Institute (2019) guidelines. The quality control strains include Escherichia coli ATCC25922 and Staphylococcus aureus ATCC25923. A total of 17 antibiotic discs were used, including amikacin (AMK), ABPC, sulfamethoxazole-trimethoprim (ST), ciprofloxacin (CPFX), chloramphenicol (CP), meropenem (MEPM), piperacillin/tazobactam, gentamicin (GM), tetracycline (TC), cefepime (CFPM), cefuroxime (CXM), cefotaxime (CTX), ceftazidime (CAZ), cefoxitin, cefazolin (CEZ), imipenem (IPM), and levofloxacin (LVFX).
Statistical analysis
Statistical analysis was performed using SPSS 27.0.1 software (IBM, Armonk, NY, USA). Categorical data were presented as frequency and percentage (%). The median was used to represent patient age.
Results
Characteristics of diarrheal patients
Data from 97 cases of diarrheal patients were collected at the First People’s Hospital of Wenling City of Taizhou City from June 1, 2020 to May 31, 2023. The characteristics of these patients are presented in Table 1. Among the 83 cases where WBC levels and CRP levels were tested, 63 patients (75.9%) had significantly elevated WBC levels above the normal range of 4 − 10 × 109/L, and 40 patients (48.2%) had CRP levels higher than 5 mg/L, exceeding the normal range for CRP. Additionally, among the 97 diarrheal patients, 80 patients (82.5%) had fecal occult blood, accounting for 82.5% of the total cases.
Characteristics of diarrheal patients.

WBC: white blood cell; CRP: C-reactive protein.
Virulence genes and serotypes of V parahaemolyticus strains
The tlh gene is considered a characteristic molecular marker of V. parahaemolyticus strains. 22 In this study, all 97 isolated strains were positive for tlh, confirming their identification as V. parahaemolyticus. Furthermore, the virulence genes tdh and trh were determined. Among the strains tested, 85 isolates (87.6%) were positive for tdh and tlh but negative for trh (tdh+ tlh+ trh−), 11 isolates (11.3%) were positive for tlh but negative for tdh and trh (tdh− tlh+ trh−), and 1 isolates (1.0%) was positive for tdh, tlh, and trh (tdh+ tlh+ trh+). These results indicate that tdh and tlh is the predominant potential virulence factors carried by the 97 V. parahaemolyticus strains (Table 2).
Distribution of virulence genes and serotypes of V. parahaemolyticus.

+: present; −: absent; KUT: K untypeable.
The results of this study revealed that among the 97 isolated V. parahaemolyticus strains, 3 out of 13 O serotypes were detected, and 4 out of 73 K serotypes were identified. In total, six different combinations of O and K antigens were identified, including O1:KUT, O3:K4, O3:K6, O4:K4, O4:K55, and O4:K55. Among them, O3:K4 (41) and O4:K4 (31) were the predominant serotypes found in patients, accounting for 42.3% and 32.0%, respectively. The other serotypes identified were O3:K6 (11), O4:K8 (10), O1:KUT (3), and O4:K55 (1), representing 11.3%, 10.3%, 3.1% and 1.0% of the cases, respectively (Table 2).
Antimicrobial resistance of V. parahaemolyticus strains
Further analysis of the drug susceptibility of the isolated 97 V. parahaemolyticus strains is summarized in Table 3. All strains were found to be sensitive to AMK, ST, CP, MEPM, piperacillin-tazobactam, TC, CFPM, CTX, CAZ, and IPM. Some strains exhibited resistance to CXM (32.6%) and CEZ (43.6%). A small number of strains showed resistance to ABPC (3.8%), CPFX (4.1%), GM (4.1%), and LVFX (3.7%).
Distribution of antimicrobial susceptibility of V. parahaemolyticus strains.
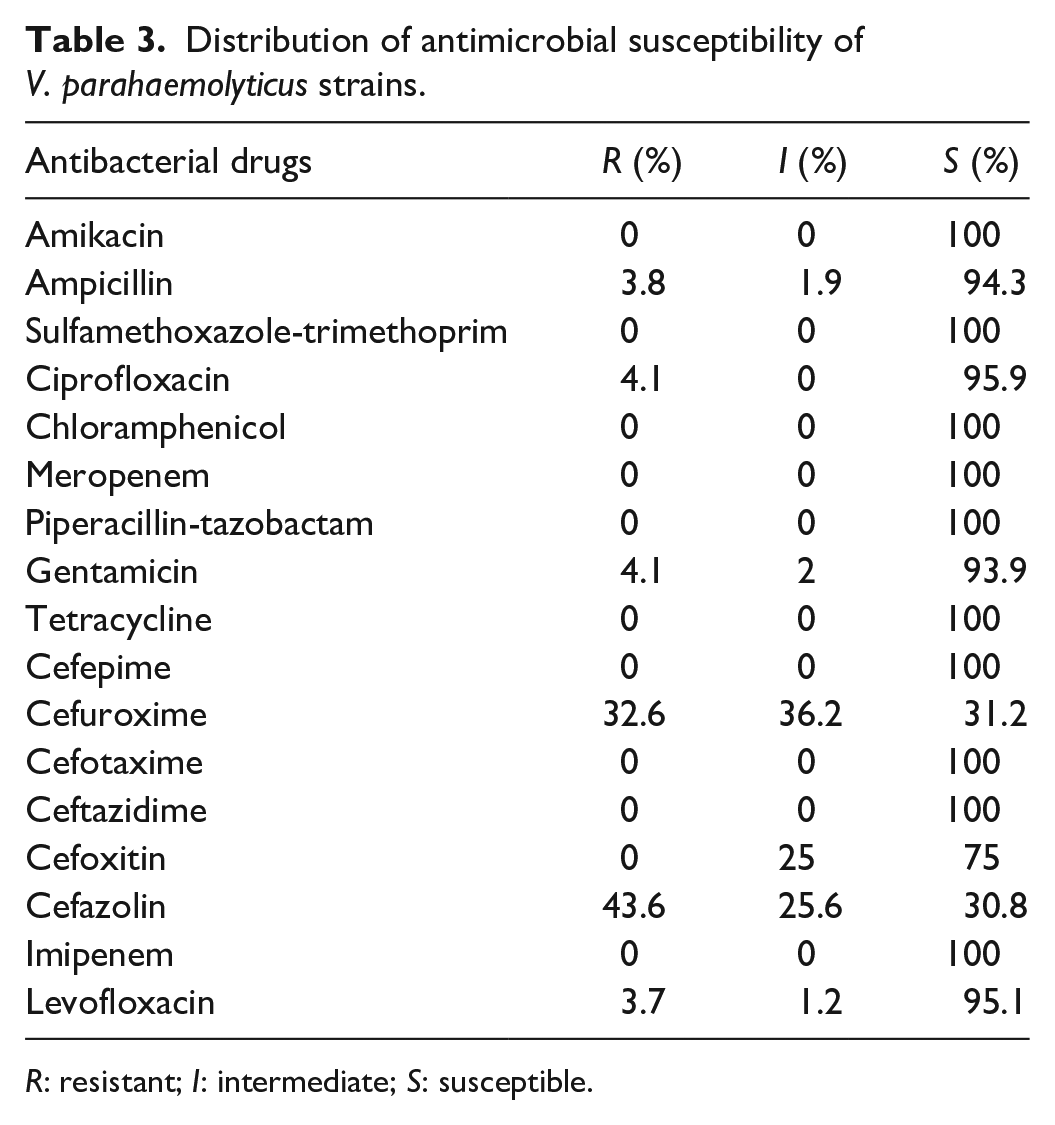
R: resistant; I: intermediate; S: susceptible.
Discussion
The major virulence genes of V. parahaemolyticus are tdh and trh, with up to 90% of clinical isolates testing positive for the tdh and/or trh genes.22–24 The TDH produced by V. parahaemolyticus causes β-hemolysis in a special blood agar medium called Wagatsuma agar, a hemolytic activity known as the Kanagawa phenomenon. The Kanagawa test refers to a positive result where β-hemolytic zones are produced on O-type human or rabbit blood agar plates containing high salt (70 g/L) and mannitol, indicating the presence of the Kanagawa phenomenon. 25 TDH is known to mediate the Kanagawa phenomenon, which determines the pathogenicity of V. parahaemolyticus, while trh and tdh shares approximately 70% homology and exhibits similar toxicity. 26 TDH has been shown to possess hemolytic, enterotoxic, and cytotoxic properties, 27 which may contribute to increased levels of WBCs, elevated CRP levels, and presence of occult blood in the stool of some patients. In this study, highly pathogenic strains were identified, with 87.6% carrying the tdh+tlh+trh− genotype and 1% carrying the tdh+tlh+trh+ genotype. However, there were also 11 isolates with tdh−tlh+trh− strains, indicating that these isolates may carry other virulence genes.13,28 Firstly, V. parahaemolyticus strains in different environments may experience different selection pressures, resulting in the loss or retention of virulence genes. Therefore, V. parahaemolyticus strains present in the same region at different time periods may exhibit different pathogenic characteristics. Secondly, V. parahaemolyticus exhibits high genetic diversity and genome plasticity, and virulence genes can be transferred between strains through mobile genetic elements such as plasmids, bacteriophages, or transposons. The frequency and efficiency of this gene-level transfer can affect the distribution of virulence genes in strains. 29
The classification of V. parahaemolyticus strains is primarily based on the serotyping of the somatic O-antigen and the capsular polysaccharide K-antigen. 6 To date, 13 O antigens and 71 K antigens have been identified. 17 It was first reported in the literature that the O3:K6 serotype was isolated from travelers returning from Indonesia to Japan in 1995. 10 Since then, the O3:K6 serotype of V. parahaemolyticus has become widespread in China and other countries. 8 The characteristics of O3:K6 include tdh+, trh−, toxRS/new+, and orf8+/−, belonging to pandemic clone 30. 30 Apart from O3:K6, serotypes such as O4:K68, O4:K8, O1:K25, and O1:KUT also belong to pandemic clones, carrying tdh+ and toxRS/new+, but trh−. 31 Subsequent studies have revealed an increasing number of non-O3:K6 serotype strains with epidemic potential.16,32 Monitoring V. parahaemolyticus in diarrhea patients from Taizhou, China, between 2020 and 2023, revealed that the predominant serotypes were O3:K4 and O4:K4, with O3:K6 accounting for only 11.3%. This differs from the overall prevalent serotypes in Zhejiang Province in recent years, where O3:K6 was the major serotype. 33 However, by 2021, the main serotype had transitioned to O10:K4. 33 This could be attributed to regional variations or the evolution of serotypes over time, as recombination around the O and K antigen gene clusters can lead to serotype conversions.34–36 This further emphasizes the diversity of V. parahaemolyticus serotypes.
The widespread use of antibiotics in clinical, agricultural, and aquaculture settings has contributed to the escalating problem of V. parahaemolyticus resistance. In this study, most of the 97 V. parahaemolyticus strains isolated from diarrhea patients in Taizhou City showed susceptibility to the majority of tested drugs. However, some strains exhibited resistance to CXM and CEZ, and a small percentage showed resistance to ABPC (3.8%). These results differ from other coastal cities in southeastern China.17,37 For example, in Huzhou City, most isolates were resistant to ABPC (86.8%). 17 In Shanghai, most isolates were resistant to ABPC (100.0%), sreptomycin (100.0%), cephazolin (92.9%), kanamycin (92.8%), and AMK (90.5%). 37 In addition, interestingly, in this study, penicillin-sensitive V. parahaemolyticus exhibited resistance to first-generation cephalosporins (43.6%) or second-generation cephalosporins (32.6%). This may be due to the higher specificity of certain β-lactamases that selectively hydrolyze cephalosporins rather than penicillin antibiotics. For example, some AmpC β-lactamases, such as those encoded by the blaCMY gene, predominantly hydrolyze cephalosporins, resulting in bacterial resistance to cephalosporins but potential sensitivity to penicillins. 38 Furthermore, environmental differences, different crop or aquaculture species, and preferences in antibiotic usage can also contribute to these results. The widespread use of cephalosporin antibiotics in clinical settings in Taizhou City may provide selective pressure on the presence of β-lactamase genes in the bacterial population, thereby increasing the frequency of these genes within the microbial community. Therefore, compared to penicillin, the frequent use of cephalosporin antibiotics in Taizhou City will lead to an increase in specific cephalosporin resistance. However, this study has certain limitations. The sample size was determined by the availability of clinical specimens during 2020–2023, and no formal sample size calculation was conducted. This may reduce the statistical power to detect rare traits (e.g., emerging antibiotic resistance) and limit the precision of prevalence estimates (e.g., wider confidence intervals). In future studies, priority should be given to priori statistical power calculations based on pilot data or regional epidemiological trends. Adopting a prospective multicenter design can enhance sample representativeness and ensure sufficient power to detect rare traits.
Conclusion
In conclusion, this study evaluated the pathogenic characteristics, including virulence genes, serotypes, and antibiotic resistance of V. parahaemolyticus isolated from diarrhea patients in Taizhou City between 2020 and 2023. The majority of the isolated strains carried the tdh gene, and the dominant serotypes were O3:K4 and O4:K4. Most of the V. parahaemolyticus strains tested were sensitive to the majority of the drugs, while some exhibited resistance to CXM and CEZ.
Footnotes
Acknowledgements
Not applicable.
Ethical considerations
The collection of clinical samples was approved by the Ethics Committee of the Wenling First People’s Hospital. The approval number is KY-2020-1012-01. All patients were given detailed information and signed informed consent forms.
Author contributions
Ding Ding: Conceptualization, Investigation, Methodology, Writing – original draft. Yi Jiang: Data curation, Formal analysis, Writing – review and editing. Yajing Lin: Data curation, Visualization. Ting Zhang: Methodology, Resources. Jiejie Guo: Investigation, Validation, Formal analysis. Tian Jiang: Visualization, Investigation. Linyao Huang: Data curation, Visualization. Jianxin Yan: Methodology, Validation. Long He: Conceptualization, Funding acquisition, Project administration, Resources, Writing – review and editing.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Zhejiang Provincial Medical and Health Science and Technology Program Project (No. 2021KY405) and Wenling City Science and Technology Program Project (No. 2020S0180089).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data used to support the findings of this study are available from the corresponding author upon request.
