Abstract
Central nervous system involvement of dengue virus is increasingly reported from endemic areas. This study describes the clinical characteristics and laboratory features of a pediatric patient enrolled in a central nervous system illness study conducted in 2017–2018 to identify viral and bacterial etiologies in Indonesian children. Dengue diagnostics including molecular and serological testing were performed on an encephalitis patient who presented with both classical dengue and neurological clinical symptoms. Dengue virus serotype 1 RNA was detected in both cerebrospinal fluid and serum by serotype-specific reverse transcription polymerase chain reaction, and the E gene was successfully sequenced. Anti-dengue virus immunoglobulin M was detected in both admission and discharge sera, whereas anti-dengue virus immunoglobulin G was identified only in the discharge serum. This study describes the central nervous system complications in a case with dengue virus infection in West Java, Indonesia, and highlights the potential for dengue virus serotype 1, a serotype rarely associated with neurotropism, to cause encephalitis.
Introduction
Dengue is one of the most significant global public health concerns especially in tropical countries. Dengue viruses (DENV 1–4) are traditionally considered to be non-neurotropic viruses, but central nervous system (CNS) involvement in the forms of encephalopathy, encephalitis, and meningitis has been reported in an increasing number of studies worldwide. 1 Neurological manifestations account for 0.5%–5.4% of patients admitted with DENV infection in Southeast Asia. 2 All four serotypes of DENV are known to be neurotropic, but DENV-2 and DENV-3 are reported to be the more common viruses.1,3–5 This study describes the clinical and laboratory findings of a patient diagnosed with viral encephalitis confirmed to be due to DENV-1.
This individual was a participant in a prospective study conducted in two hospitals in Bandung, West Java, Indonesia, identifying common bacterial and viral etiologies of CNS infection in children between 6 months and 17 years during the period 2017–2018. Blood and cerebrospinal fluid (CSF) from suspected CNS infections were processed using serological, molecular and cell culture assays at the Eijkman Institute for Molecular Biology. Clinical and laboratory data were collected from the participating hospitals. The study was approved by the Institutional Ethics Committee on Medical Research Involving Human Subjects (LB.04.01/A05/EC/255/VIII/2017), and written informed consent was obtained from parents of the subjects.
The case
A 7-year-old boy presented to the Emergency Department of Hasan Sadikin Hospital with a history of difficulty arousing from sleep, confusion, and incoherent speech for 1 day before admission. The patient experienced sudden onset of fever for 2 days prior to admission accompanied by coffee ground vomiting. There was no history of other bleeding episodes or seizures. Physical examination showed an obese, drowsy patient with a Glasgow Coma Scale (GCS) of 13, fever of 38.3°C, normal blood pressure, and no circulatory problems. Abdominal examination revealed no signs of hepatomegaly, abdominal tenderness, or petechiae. Ocular tests revealed normal pupils. Neurological examination showed no facial asymmetry, motor weakness, abnormal movements, nor posture. Reflexes were normal, and the Babinski was negative. Nuchal rigidity, Kernig, and Brudzinski signs were absent.
Laboratory examination showed hemoglobin and hematocrit levels were within normal range with leukocyte count of 1.9 × 103/uL, and lower platelet count of 129 × 103/uL. Liver transaminases were high with 446 U/L for aspartate aminotransferase (AST) and 204 U/L for alanine aminotransferase (ALT). Patient electrolytes were within normal limits with sodium of 141 mmol/L, potassium of 3.9 mmol/L, and non-fasting blood glucose of 139 mg/dL. CSF showed clear fluid, negative for Nonne’s and Pandy’s test and protein of 26.9 mg/dL which indicated CSF protein was within normal limits, cell count of 4 cells/uL, and glucose of 98 mg/dL. CSF gram and acid-fast bacilli stains were negative. Neuroimaging was not conducted. A presumptive diagnosis of viral CNS infection was given based on neurological manifestations as well as laboratory parameters.
During follow-up, conscious level improved to GCS 15 on day 4 and defervescence occurred on day 3 of hospitalization. Hematocrit dropped to 34.3% on the day 5 while nadir platelet count occurred on day 3 of hospitalization. Supportive treatment was given with antipyretics and fluid replacement. The patient continued to improve and was discharged on the sixth day following admission with dengue encephalitis as the final diagnosis. On the 1-month follow-up, the patient had completely recovered without any neurological sequelae.
Additional laboratory examination confirmed the presence of DENV serotype 1 RNA by reverse transcription polymerase chain reaction (RT-PCR) in CSF taken on admission day and in the serum specimen taken on the second day of hospitalization (Table 1). Molecular screening was conducted by genus-specific flavivirus RT-PCR, followed by DENV serotyping RT-PCR6,7 and whole genome sequencing. Obtained sequence was submitted to the GenBank with accession number MW559478.1. Anti-DENV immunoglobulin M (IgM) 8 was positive in both serum specimens from the second and the sixth day of hospitalization, while samples collected on day 6 showed immunoglobulin G (IgG) seroconversion (Dengue Virus IgG DxSelect, Focus Diagnostics, USA). Other classical neurotropic pathogens including Japanese encephalitis virus (JEV), chikungunya virus, human herpesviruses, enteroviruses, Streptococcus pneumonia and Haemophilus influenzae were negative using PCR testing algorithms 9 as well as an anti-JEV IgM assay. 10 Both CSF and blood samples were inoculated onto Vero E6 cells to attempt for virus isolation; no virus was isolated from these efforts. A phylogenetic tree was constructed based on E gene aligned to the top 20 sequence matches in NCBI BLASTn database (Figure 1). The DENV-1 strain from this study belonged to genotype IV and appeared to be closely related to strains previously identified in Indonesia. None of the GenBank DENV-1 strains analyzed in the phylogenetic tree were reported to be associated with CNS manifestations.
Summary of clinical and laboratory findings.

AST: aspartate aminotransferase; ALT: alanine aminotransferase; DENV: dengue virus; JEV: Japanese encephalitis virus; CSF: cerebrospinal fluid; QNS: quantity not sufficient; ND: not done.
DENV IgM ELISA. 8
Dengue Virus IgG DxSelect (Focus Diagnostics, USA).
JEV IgM ELISA. 10
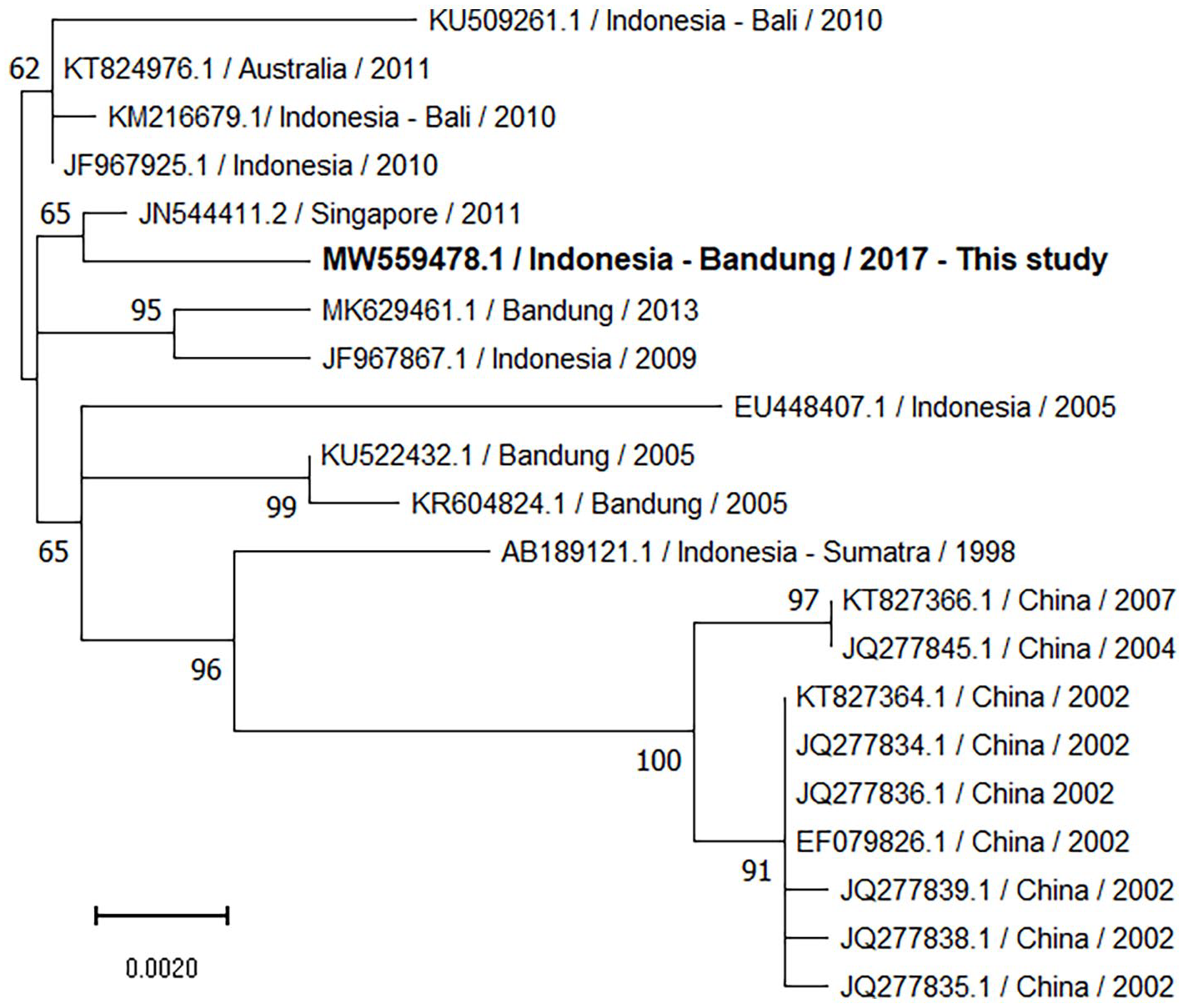
Phylogenetic tree comparing E gene of DENV-1 from the patient to the top 20 aligned sequences in NCBI BLASTn database. The tree was built using the Tamura–Nei model, and maximum likelihood method with 1000 bootstrap replicates. Bootstrap percentages (⩾50%) are shown on internal branches. Label indicates the accession number/country—city or region/collection year. The scale bar represents the number of nucleotide substitutions per site.
Discussion
Encephalitis is increasingly recognized to be associated with DENV infections in endemic areas. The mechanisms of CNS infection by DENV are not well understood; it could be a direct invasion followed by replication within the CNS inducing neuronal cytotoxicity and inflammation, or by crossing the blood–brain barrier during viremia as reported with other flaviviruses. 2 Neurological manifestations related to DENV infection have been reported in Indonesia,5,11 Vietnam, 1 Thailand, 4 and Malaysia. 3 This study case presented with dengue-like biphasic fever and decreased consciousness. Laboratory testing confirmed primary DENV infection based on IgM-positive/IgG-negative results on the serum specimen obtained within 2 days of symptom onset (Table 1); both primary and secondary dengue are reported to be associated with neurological complications. This case has fulfilled the four criteria of dengue encephalitis2,12 as demonstrated by: (1) the presence of fever with hemorrhagic manifestation and thrombocytopenia; (2) CNS involvement including altered sensorium; (3) the presence of DENV RNA in CSF and serum as well as IgM in serum; and (4) the exclusion of other causes of encephalitis.
The previously reported DENV infections with CNS manifestations have been mostly based on IgM serology without RNA or virus isolation. 4 A few studies have successfully isolated DENV in either CSF or blood and reported DENV-2, 1 DENV-3,3,5 and DENV-4. 13 DENV-1 is rarely associated with neurological disease and has only recently been reported from Cambodia 14 and Myanmar. 15 Some fatal cases of DENV CNS infection were reported to be associated with hemorrhagic manifestations. 2 However, this case of DENV-1 encephalitis with bleeding episodes was not associated with metabolic disturbances or neurological sequelae. The neurological manifestations were mild and self-limiting, although evidence of viral RNA in the CSF indicated that CNS invasion occurred. It is also interesting to note that the study case was an obese patient reported to be associated with more severe DENV infection. 16 DENV CNS infection might occasionally be associated with classical dengue manifestations as in this case. However, this case did not manifest lymphocyte pleocytosis which was commonly reported in other studies of dengue encephalitis.1,2,4
Conclusion
DENV has only recently been considered as an important cause of encephalitis. This study reports a case with fever and neurologic impairments and recovery of DENV-1 RNA in CSF from a child with encephalitis admitted to one referral hospital in West Java. Dengue virus should be considered as an etiology of CNS infection in endemic areas, especially in those presenting with relevant symptoms and laboratory findings. Further studies are warranted to clarify the clinical relevance of different serotypes and genotypes as well as the patient’s immune status, that is, primary versus secondary infection in CNS infection associated with DENV.
Footnotes
Acknowledgements
The authors thank the patient and family for agreeing to participate, the staff of Hasan Sadikin Hospital for their support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. The funders did not participate in study design, data collection or analysis, decision to publish, or preparation of the manuscript. The findings and conclusions expressed in this report do not necessarily represent the views of the Centers for Disease Control and Prevention.
Ethical approval
Ethical approval to report this case was obtained from The Institutional Ethics Committee on Medical Research Involving Human Subjects Faculty of Medicine Universitas Padjadjaran (LB.04.01/A05/EC/ 255/VIII/2017).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Research and Technology, Republic of Indonesia; Internal grant Faculty of Medicine, Universitas Padjadjaran and the US Centers for Disease Control and Prevention.
Informed consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
