Abstract
Objectives:
In 2018, the Food and Drug Administration approved pegvaliase-pqpz as an enzyme replacement therapy to lower blood phenylalanine levels in adults with phenylketonuria. However, its potential side effects have not been fully explored in a real-world setting. This study aimed to examine pegvaliase-pqpz-related adverse events and investigate their associated risk factors to offer important insights into clinical drug applications.
Methods:
We extracted reports on adverse events from Food and Drug Administration Adverse Event Reporting System database from 2018Q2 to 2024Q3. The analyses included demographic data, time-to-onset, sex, age, dosage, reporting personnel, and subgroup signals. The Reporting Odds Ratio, Proportional Reporting Ratio, Bayesian Confidence Propagation Neural Network, and Multi-item Gamma Poisson Shrinker were used for disproportionality analysis. A screening procedure was implemented for designated medical events, focusing on medication-related adverse events.
Results:
A total of 6264 reports were identified. Arthralgia was the most frequently reported adverse events. Lymphadenopathy, serum sickness, and tunnel vision were identified as unexpected signals. Weibull distribution analysis revealed that the incidence of adverse events decreased over time.
Conclusions:
Our study provides a deeper insight into the safety profile of pegvaliase-pqpz, thereby aiding healthcare professionals in clinical surveillance and risk identification.
Introduction
Phenylketonuria (PKU) 1 is an orphan disease caused by a monogenic autosomal recessive inborn error in amino acid metabolism caused by an enzymatic defect in the metabolic pathway of phenylalanine (Phe), which prevents the conversion of Phe to tyrosine and increases the levels of Phe in the blood (usually >363 μmol/L (6 mg/dL)). If left untreated, it can lead to mental retardation, light hair and skin colour, eczema, seizures, extreme arousal, sweat, a distinctive and pungent urine smell, and other major clinical features.
Approximately 450,000 individuals are affected by PKU globally 2 and the neonatal incidence varies among different races and regions. 3 Among them, Turkey has the highest, with approximately 1 in 2600 newborns, 1/10,000 in Northern Europe and East Asia, and 1 in 10,000 to 2 in 30,000 in the United States. The lowest reported prevalence of PKU is in Thailand, with approximately 1 in 230,000 newborns. However, PKU remains incurable. Early diagnosis and administration of a low-Phe diet can result in normal intellectual development but must strictly adhere to the lifelong diet plan. Otherwise, mental decline and neurological and psychiatric problems may occur. This form of treatment significantly impairs patients’ quality of life.
In May 2018, pegvaliase-pqpz 4 (Palynziq®, PAL) was approved by the Food and Drug Administration (FDA) for the treatment of adult PKU, and it was also approved for marketing in the EU in 2019. PAL is a PEGylated recombinant enzyme that transforms Phe into ammonia and transcinnamic acid. It addresses insufficient phenyalanine hydroxylase (PAH) activity in patients with PKU and reduces Phe levels in the blood, making it the only enzyme replacement therapy currently available for PKU. 5 However, there is limited population-based research evidence on the adverse events (AEs) associated with PAL. Thomas et al. 6 and Harding et al. 7 conducted a phase III clinical trial of PAL for the treatment of PKU to evaluate its safety and efficacy. A total of 261 participants were enrolled in this study. Blood Phe levels, attention deficit hyperactivity disorder rating scale, and mood state profile were used to observe changes in blood Phe levels, attention improvement, and adverse drug events. They found that PAL treatment could significantly reduce blood Phe concentration levels and improve inattention symptoms in patients with PKU and that arthralgia was the most commonly associated AE. To further assess the safety of PAL in a larger population range, our study utilized the Food and Drug Administration Adverse Event Reporting System (FAERS) to analyze AEs signals related to PAL, conduct a pharmacovigilance analysis, and offer an understanding of clinical application of PAL from a practical viewpoint.
Materials and methods
Data sources
Study data were obtained from the FAERS database, a system developed to facilitate the FDA’s post-marketing safety oversight of all marketed drugs and therapeutic biologic products. 8 This database collects information on millions of real-world AEs and medication error information from healthcare professionals, patients, and drug manufacturers. Each report contained information on variables such as the sex, age, duration of drug use, time to onset (TTO), drug dose, and causality. The information structure of FAERS was based on the International Safety Reporting Guidelines (ICH E2B) released by the ICH, and the terms for AEs and medication errors in the report were coded according to the International Medical Language Terminology (MedDRA). AEs were coded using the preferred term (PT). A specific PT can be assigned to multiple higher-level terms, higher-level group terms, and system organ class (SOC). We retrieved the time range from the second quarter of 2018 (drug approval time) to the third quarter of 2024. Figure 1 shows the method used to identify and analyze PAL-related AEs in the FAERS database.

The process of searching and analyzing PAL-related AEs from FAERS.
Data analysis
As the FAERS database relies on spontaneous reporting, there will inevitably be duplicate (the same report submitted by different sources) and multiple (subsequent follow-up of the same case and provision of supplementary and updated information) reports. Therefore, we removed duplicate data according to the following standards recommended by the FDA 9 to improve the reliability of the study results: If the report ID number (CASEID) was the same, the most recent reporting date (FDA_DT) was selected; if both CASEID and FDA_DT were the same, the record with the higher PRIMARYID was retained, and the rest were considered duplicate reports and deleted. The onset of adverse drug reactions was defined as the difference between the time at which the patient first took the drug and the time at which the first adverse reaction occurred. Therefore, reports with missing “date of drug initiation,” “onset of AE,” or with “date of drug initiation” later than the “onset of AE” were considered abnormal reports and excluded from the analysis.
To ensure that all relevant reports on PAL were extracted from the database, we utilized the National Center for Biotechnology Information Medical Subject Headings (NCBI MESH) to identify and organize all the names of PAL and used four measures of disproportionality to identify possible PAL-AE links: reporting odds ratio (ROR), proportional reporting ratio (PRR), Bayesian confidence propagation neural network (BCPN), 10 and multi-item gamma-Poisson shrinker (MGPS) (Table 1). A significant pharmacovigilance signal is identified when the AE rate associated with the target medication exceeded the background frequency and met the established thresholds of the four indices (Table 2). To assess serious and special safety events related to PAL, we conducted a comparative analysis using the designated medical event (DME) list of the European Medicines Agency to identify valuable positive signals. To determine whether the incidence of AEs was statistically different, we used a hierarchical logistic regression analysis, subdividing the data into different groups, including sex, age, dosage, and reporting personnel groups. R software version 4.2.2 11 was used for all the analyses. Statistical significance was set at p < 0.05.
Algorithm of four disproportional analysis.

EBGM: empirical Bayesian geometric mean; IC: information component; SD: standard deviation; IC-2SD: the lower limit of the 95% CI of the IC; E (IC): the IC expectations; V (IC): the variance of IC; EBGM05: the lower limit of the 95% CI of EBGM; 95% CI: 95% confidence interval.
Two-by-two contingency table for disproportionality analysis.

Weibull distribution
The incidence of AEs often changes over time owing to the different mechanisms of action of the drugs, whereas AEs unrelated to drug treatment occur at a constant rate. We used the Weibull distribution test to determine the proportion of changes in the incidence of AEs. The Weibull distribution is a continuous probability distribution whose probability density function and cumulative distribution function can be defined by the scale parameter (
Results
General characteristics
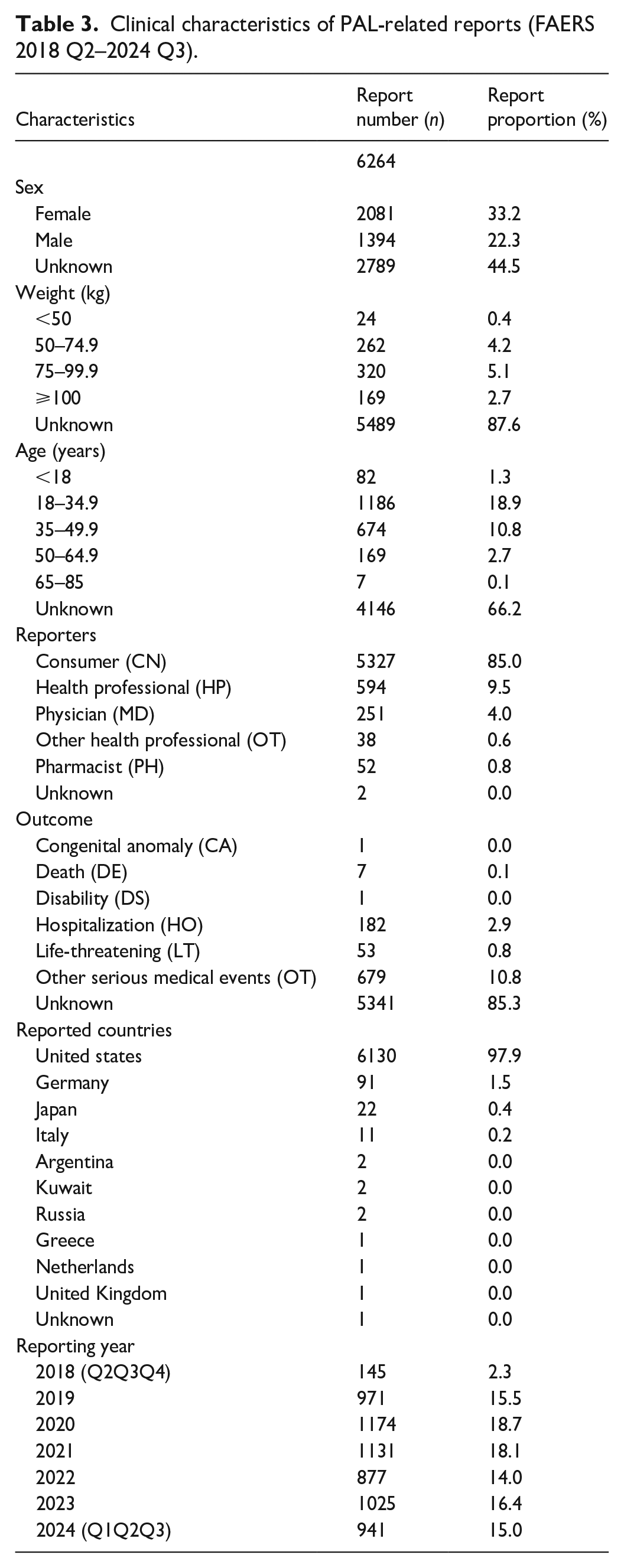
In total, 11,327,051 reports were extracted from the FAERS database. After eliminating duplicates, 9,617,619 reports were included in the final analysis, of which 6264 were PAL-related AEs. Table 3 provides a comprehensive overview of the patients’ baseline characteristics. After excluding cases with incomplete data, a higher proportion of PAL-related AE reports involved females (33.2%) than males. Most individuals (5.1%) weighed between 75 and 99.9 kg. Among those with a known age, the 18–34.9 years old age group accounted for the highest percentage of AE reports (18.9%). Most AE reports (85.0%) were self-submitted by the patients, a pattern consistent with other treatments that require long-term medication. The most frequently reported serious outcomes were “Other Serious Medical Events” (OT, 10.8%) and “Hospitalization” (HO, 2.9%). The United States contributed the largest number of reporting countries (97.9%). Throughout the study period, the number of PAL-related AE remained relatively stable after the second year of drug approval and peaked in 2020.
Clinical characteristics of PAL-related reports (FAERS 2018 Q2–2024 Q3).
Signal detection results
We calculated the PAL signal reporting at the SOC level (Figure 2(a)). Our analysis revealed that 27 SOCs were implicated in PAL-related AEs, General Disorders and Administration Site Conditions showing the highest incidence rates (32.4%). Notably, the SOCs that conformed to the criteria set by the four disproportionality algorithms included Musculoskeletal and Connective Tissue Disorders and Immune System Disorders. Additionally, General Disorders and Administration Site Conditions, Skin and Subcutaneous Tissue Disorders, Respiratory, Thoracic, and Mediastinal Disorders were also identified as significant SOCs, conforming to the ROR algorithm. A forest plot illustrating the SOC signals based on the reported numbers is presented in Figure 2(b).

(a) The bar chart of AEs at the SOC level and (b) forest plot of signal detection at the SOC level.
At the PT level, a total of 934 PTs were included in the follow-up analysis after excluding categories such as social circumstances, COVID-19, product issues, and other PTs unrelated to the drug. Among them, 79 cases of significantly disproportionate PTs simultaneously conformed to all four algorithms (Figure 3(a)). According to the adverse reaction descriptions in drug instructions and previous studies, common adverse reactions encompass anaphylaxis, tachycardia, angioedema, rash, urticaria, pruritus, and gastrointestinal symptoms (such as vomiting, nausea, and diarrhea). In our study, most AEs, such as Injection Site Erythema (PT: 10022061), Rash (PT: 10037844), Heart Rate Increased (PT: 10019303), and Oropharyngeal Pain (PT: 10068319), were consistent with findings from clinical trials. Interestingly, some significant AEs not previously anticipated were also identified in the list, including Lymphadenopathy (PT: 10025197, n = 36, ROR: 4.49; 95% CI: 3.23–6.23), Serum Sickness (PT: 10040400, n = 7, ROR: 14.08; 95% CI: 6.69–29.61), and Tunnel Vision (PT: 10045178, n = 5, ROR: 12.04; 95% CI: 5–29.01). The forest plot in Figure 3(b) shows the top 50 reported PTs.

(a) Venn plot of identification of 79 effective PTs achieved through the four different methods (b) Forest plot of PTs with a Top 50 cases. The blue arrows indicate that the 95% confidence interval’s lower boundary for the ROR is above 50.
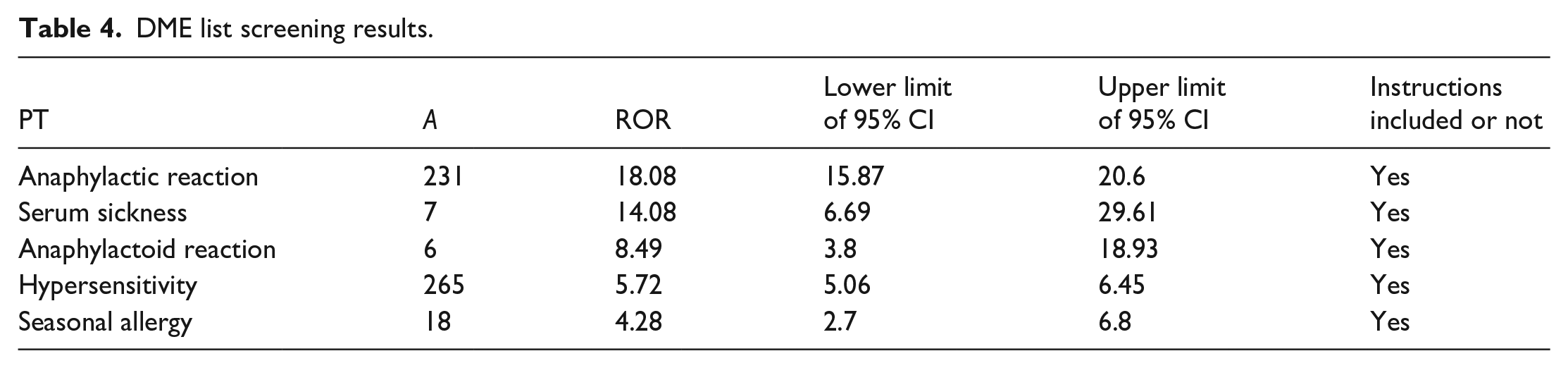
DME list screening
Among the 79 positive signals, 2 corresponded to the DME list, specifically Anaphylactic Reaction and Anaphylactoid Reaction. These signals were categorized as Immune System Disorders at the SOC level and as Hypersensitivity at the Narrow SMQ level. In addition, there were five positive signals related to AEs associated with immune system disorders (Table 4). Notably, the highest signal value was for the Anaphylactic Reaction, with an ROR of 18.08.
DME list screening results.
Time to onset
After discarding reports with incomplete information, 797 AEs were compiled for analysis of the onset time of AEs occurrences. The median TTO was 15 days (interquartile range[IQR]: 9–39 days), with most cases (n = 548, 68.76%) occurring within the first 30 days after PAL therapy initiation. The incidence of AEs progressively decreased over time, with 118 AEs (14.81%) reported between 31 and 60 days, 37 AEs (4.64%) between 61 and 90 days, and 43 AEs (5.40%) between 91 and 180 days. Notably, 3.76% of the reports indicated that AEs still manifest after 1 year of PAL treatment (Figure 4(a) and (b)). To further substantiate the temporal patterns observed in the risk of PAL-related AEs, a Weibull distribution analysis was conducted. From the overall analysis, a shape parameter (β) of 0.65 was derived, with the 95% confidence interval’s upper limit at 0.68. Values less than 1 indicate an early failure-type model, suggesting that the rate of AEs decreases over time (Table 5), which aligns with the findings of previous analyses.

(a) Bar chart of time to onset of PAL-related AEs and (b) curve plot of AEs onset time.
Weibull distribution of the PAL-related AEs.

Subgroup analysis
Gender subgroup analysis
To ascertain whether sex influenced the incidence of PAL-related AEs, we used the ROR method to assess the significant disproportionality of AEs rates between males and females. In the male cohort, 61 instances of significant disproportionality were identified, conforming to all four algorithms. Conversely, the female cohort exhibited 54 instances of significant disproportionality, according to the same algorithms. After intersecting the male and female subgroups, 39 significantly disproportional PTs were identified (Figure 5(a)). Arthralgia emerged as the most prevalent PT in both genders. We also use volcano plot to analyze the gender-specific differences in PAL related AEs It revealed that women had a higher likelihood of experiencing Injection Site Pruritus (PT: 10022093, ROR = 1.5, 95% CI: 1.08–2.08), whereas Chills (PT: 10008531, ROR = 0.47, 95% CI: 0.3–0.73) were more frequently reported in men (Figure 5(b)).
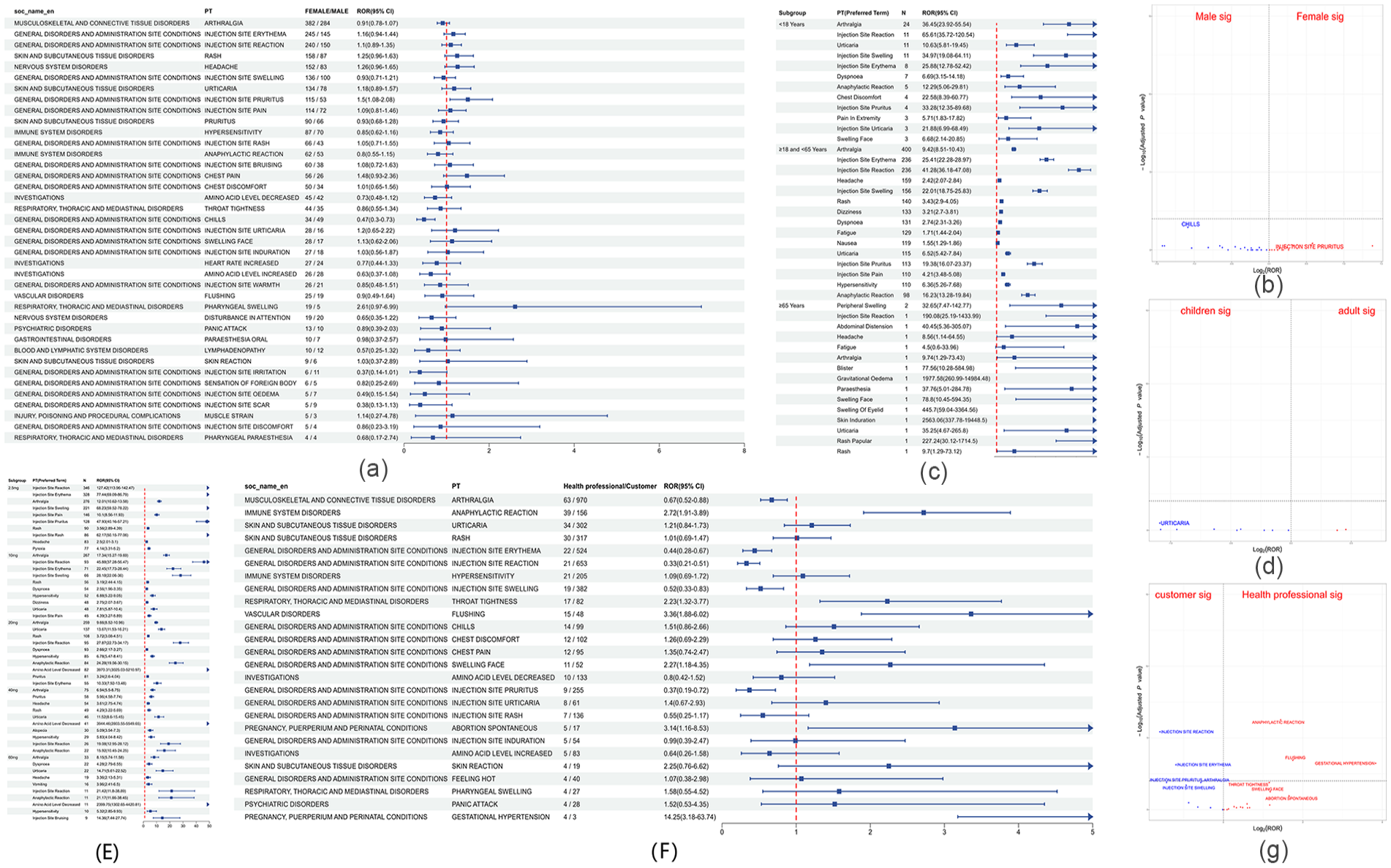
(a) Forest plot of gender-differentiated risk signals in PAL. (b) Volcano plot of gender-differentiated risk signal in PAL. (c) Forest plot of top 10 age-differentiated risk signals in PAL. (d) Volcano plot of age-differentiated risk signal in PAL. (e) Forest plot of top 10 dose-differentiated risk signals in PAL. (f) Forest plot of reporters-differentiated risk signals in PAL. (g) Volcano plot of Reporters-differentiated risk signal in PAL. PTs were displayed in descending order by number of case reports. The blue arrows indicate that the 95% confidence interval’s lower boundary for the ROR is above the maximum scale value.
Age subgroup analysis
To examine the relationship between age and PAL-related AEs, we categorized the participants into three age groups: minors (<18 years), adults (18–65 years), and older adults (>65 years). From 6264 reports, we identified 2118 reports with complete and pertinent data. These reports included 82 minors, 2029 adults, and 7 older adults. In the minor group, SOC levels conforming to all four algorithms were associated with Musculoskeletal and Connective Tissue Disorders. The top five PTs with high frequencies that also conformed to all four algorithms were Arthralgia, Injection Site Reaction, Urticaria, Injection Site Swelling, and Injection Site Erythema (Figure 5(c)). No life-threatening adverse reactions were reported in the minors.
For the adult group that conformed to the standards of the four algorithms, the identified SOC levels were Immune System Disorder. PTs associated with this class include Hypersensitivity, Anaphylactic Reaction, Seasonal Allergy, and Anaphylactoid Reaction. The top five PTs with the highest frequencies according to the four algorithms were Arthralgia, Injection Site Erythema, Injection Site Reaction, Headache, and Injection Site Swelling (Figure 5(c)). Due to the limited size of the elderly group, further comparative analyses were not feasible. Volcano plot analysis revealed a significant difference in the signal for Urticaria (PT: 10046735) in the minor group compared with that in the adult group (Figure 5(d)).
Dosage subgroup analysis
The relationship between PAL-related AEs and the injectable dose was examined. The injection doses were stratified into five subgroups: 2.5, 10, 20, 40, and 60 mg. After data consolidation, the numbers of reports included in the analysis for each subgroup were 1206, 1028, 1728, 713, and 258, respectively. The PT signal forest plot for each subgroup is shown in Figure 5(e). In the 2.5-mg subgroup, at the SOC level, the four algorithms identified General Disorders And Administration Site Conditions and Musculoskeletal And Connective Tissue Disorders as positive signals. At the PT level, the top three most frequent terms according to the four algorithms were Injection Site Reaction, Injection Site Erythema, and Arthralgia. In the 10-mg subgroup, the SOC level was consistent with those in the2.5-mg group. At the PT level, the three most frequently used terms were Arthralgia, Injection Site Reaction, and Injection Site Erythema. In the 20-mg subgroup, Immune System Disorders conformed to the criteria of the four algorithms at the SOC level. At the PT level, the three most frequent terms that met all four algorithms were Arthralgia, Urticaria, and Rash. In the 40-mg subgroup, Skin and Subcutaneous Tissue Disorders and Immune System Disorders at SOC level were consistent with the four algorithms. At the PT level, the three most frequent terms were Arthralgia, Pruritus, and Headache. In the 60-mg subgroup, Immune System Disorders were identified as positive at the SOC level. At the PT level, the three most frequent terms were Arthralgia, Dyspnoea, and Urticaria.
Reports subgroup analysis
We investigated the relationship between AEs associated with PAL and the categorization of reporting personnel. Participants were divided into two primary categories: consumers and healthcare professionals. The consumer category comprised self-reports from patients, with 5327 reports included in the analysis. The healthcare professionals category included physicians, pharmacists, health professionals, and other health professionals, and 935 reports were included in the analysis. The PTs reported in both groups that conformed to the criteria of the four algorithms were screened, resulting in the inclusion of 26 cases for further analysis (Figure 5(f)). At the SOC level, the consumer subgroup categorized Immune System Disorders and Musculoskeletal and Connective Tissue Disorders as positive. Pregnancy, Puerperium and Perinatal Conditions, and Immune System Disorders among healthcare professionals. At the PT level, the three most frequent terms in the consumer subgroup were Arthralgia, Injection Site Reaction, and Injection Site Erythema. In the healthcare professionals group, the top three symptoms were Arthralgia, Snaphylactic reaction, and Urticaria. Volcano plot analysis revealed that Injection Site Reaction, Injection Site Erythema, Injection Site Pruritus, Arthralgia, and Injection Site Swelling were more prevalent among the self-reported consumers. Conversely, the healthcare professionals group was more frequently affected by Anaphylactic Reaction, Flushing, Gestational Hypertension, Throat Tightness, Facial Swelling, and Spontaneous Abortion (Figure 5(g)). The PT signals were significantly different between the two groups (p < 0.05).
TTO subgroup analysis
The TTO of AEs was analyzed based on sex, age, and individual reporting (Figure S1). The median TTO was 15 days as reported by consumers (IQR: 7–70.25 days) and (IQR: 9.75–38 days) for healthcare professionals. Significant differences were observed in the TTO of AEs among subgroups with different informants (p = 0.056). However, no significant differences were identified between subgroups stratified by age or sex.
Discussion
PKU is an autosomal recessive genetic disorder resulting from mutations in the phenylalanine hydroxylase (PAH) gene, leading to impaired amino acid metabolism. The PAH gene encodes the PAH enzyme phenylalanine 4-hydroxylase (EC 1.14.16.1) and is located on chromosome 12q23.2. 14 There are more than 1500 variants of PAH, more than 50% of which are caused by missense mutations. 15 Patients with various PAH mutations may display diverse phenotypic manifestations of PKU. These manifestations range from classic PKU (blood Phe levels exceeding 1200 μmol L−1) to milder forms of the disorder. In these milder forms, some residual enzymatic activity is retained, resulting in less severe hyperphenylalaninemia (HPA). These milder phenotypes are categorized as mild PKU and mild HPA, with blood Phe concentrations from 600–1200 μmol L−1 to 120–600 μmol L−1, respectively. On May 11, 2018, the National Health Commission of China, along with five other departments, jointly formulated the “First List of Rare Diseases” in China, wherein PKU was included. When parents of a child with PKU are carriers of the disease-causing gene, the mother has a one-in-four chance of giving birth to a child with PKU in each pregnancy. A child can be identified through routine newborn screening within the first few days of birth and can receive timely and regular treatment. 16 If left untreated, most patients would suffer from severe intellectual disability, and their average life expectancy is only 3 or 40 years.
It is essential to prevent intellectual disability and prolong life by strictly controlling a low Phe diet in the treatment of PKU as the main and most effective method. In addition to strict dietary control, medication is required. Initially, the main drug used for the treatment of PKU in clinical practice was sapropterin dihydrochloride (Kuvan), 17 which was approved by the EU and FDA in 2008. Kuvan was the first drug to be combined with dietary adjustment, which reduces the concentration of Phe in the blood and brain of patients with PKU to prevent the destructive effects of PKU. It is suitable for all age groups with BH4 deficiency and for PKU patients who have a drug response and are over 4 years old. Considering the age at PKU onset, the safety and efficacy of Kuvan in key patients under 4 years of age are still unclear in various previous studies.
To meet the increasing demand for effective PKU treatment drugs, PAL, 18 also developed by Biomarin Co., Ltd., was approved by the FDA for marketing in May 2018. It is injected as a solution stored in prefilled syringes for subcutaneous injection. It is suitable for PKU patients with inadequate blood Phe control aged 16 years and older. PAL was the first and only enzyme replacement therapy for PKU. In early clinical trials of this drug, major adverse reactions were found to be allergic reactions. They can occur at any stage of treatment. In addition, injection site reactions, arthralgia, gastrointestinal symptoms, headache, systemic skin reactions, nausea, cough, sore throat, itching, nasal congestion, fatigue, and anxiety6,7,19,20 are also major adverse reaction symptoms. Considering the lack of drugs for PKU treatment and the potential serious side effects caused by PAL, it is necessary to pay attention to drug reactions in key populations. However, data regarding the management of adverse reactions to this drug are limited. Sacharow et al. 21 reported a clinical model of 46 adult patients with PKU who received PAL treatment for 1.5 years. Approximately 70% of patients had blood Phe < 360 μmol/L on starting PAL, with an average decrease of 68 ± 24% compared to baseline. Although the patients experienced mild side effects, they were effectively managed and treated in most cases. Patrawala et al. 22 reported a case of a PAL-induced allergic reaction and successful desensitization. A 13-step desensitization protocol was carried out in an outpatient setting using three concentrations of PAL with premedication with diphenhydramine and prednisone. The patient tolerated the desensitization treatment and was able to continue using the PAL. Lah 23 reported a clinical model of 43 patients with PKU who received PAL treatment for 1 year. 35.1% of patients maintained blood Phe levels < 360 μmol/L (6 mg/dL) without Kuvan assistance or medical food. The time and dose required to achieve a response were unrelated to the patient’s weight, initial Phe levels, initial diet, or combined treatment with Kuvan. A total of 21.6% reported allergic reaction episodes, which usually occurred shortly after the dose increase. However, the drug records for children under 16 years of age are still incomplete.
To further analyze the AEs of PAL in the PKU treatment, we investigated the AEs associated with PAL and their differences in different subgroups using the FAERS database. The FAERS database contains global adverse drug reactions and AE reports from 1968.24,25 These data were derived mainly from two reporting systems. One is the MedWatch online voluntary reporting system, where the data mainly come from individual case safety reports (ICSR) submitted by healthcare professionals and patients, and the other is ICSR submitted by pharmaceutical companies and drug distributors through database-to-database transmission and the secure reporting portal in electronic form. Our study retrospectively analyzed this information to further explore the potential PAL risk factors, AEs in high-risk populations, and emerging clinical safety issues.
Overall scenario
The study revealed that women (33.2%) had a higher incidence of PAL-related AEs than male counterparts. Previous studies have not found significant differences in the efficacy or side effects of PAL treatment between sexes. A possible factor contributing to this difference may be that the clinical research population in the FAERS database was limited, which may have led to undetected differences. Additionally, a study by Falcão et al. 26 showed that women absorb more medication than men, but did not explicitly confirm that sex affects the pharmacokinetics of PAL. Qi et al. 27 discussed the pharmacokinetic, pharmacodynamic, and immunogenicity basis of the optimal PAL dosing regimen for the treatment of PKU in adults, but did not directly mention the specific differences in the efficacy of PAL in PKU patients of different sexes. However, this finding is worthy of further attention, as it may be a cause of increased AEs in the female population. As the FAERS reporting system is voluntary, most of our target cases lacked age and weight information. However, among cases with complete record information, the proportion of patients who experienced AEs was the highest among those aged 18–35 (18.9%) and those with a weight of 75–100 kg (5.1%). This age range is in young adulthood, and the weight range is also wide. A potentially unhealthy lifestyle, high daily protein intake, and other factors that may increase the risk of AEs are worth noting. Serious adverse outcomes, including death and life-threatening conditions, constituted a minor proportion (0.9%) of the PAL-related outcomes. Our findings are consistent with those of previous clinical trials, which demonstrated that most adverse reactions were mild or moderate. The United States accounted for the vast majority (97.9%) of the reported cases. Previous studies have indicated that PKU can occur across all ethnic groups although its prevalence varies significantly by geographical region. Consequently, AEs may vary different countries or racial groups. To validate our findings further, we aimed to identify AE reports from other countries. Scala et al. 28 reported the experience of 18 patients with PKU in Italy who received PAL treatment. Of the patients, 66.7% experienced AEs, including injection site reactions, erythema, edema, and arthralgia. Most adverse reactions were mild to moderate. Only one patient required temporary suspension of the dosage because of recurring adverse reactions. Ishige et al. 29 analyzed the experiences of 12 PKU patients from Japan, all of whom reported at least one AE, the most common being injection site erythema and edema (n = 10, each 83.3%), without AE leading to discontinuation of treatment, no deaths, and no allergic reaction events. Gregoric et al. 30 reported the experience of a 28-year-old white male from Slovenia who did not respond to PAL and discontinued H1 antihistamine medication. However, a sudden allergic reaction occurred that was successfully resolved by daily antihistamine administration closer to the PAL injection site. In China, as PAL has not yet been approved for marketing, it is difficult to determine any relevant AEs or analyze the differences between races and regions. We also observed that the number of PAL-related AEs peaked in 2020 and has since shown a downward trend. According to Biomarin’s financial reports, global sales of PAL are growing at an annual rate of approximately 8%–10%, and the surge in drug consumption has not resulted in a steady rise in AEs. This may be due to the more effective use of the PAL under the guidance of medical professionals.
At the SOC level, AEs related to PAL (n = 6264) were distributed across 27 SOCs, with the highest proportion being General Disorders and Administration Site Conditions (n = 2029). The other two SOCs meet the threshold criteria of the four algorithms. These included Musculoskeletal and Connective Tissue Disorders and Immune System Disorders. This is consistent with the most common side effects mentioned on the drug labels, such as hypersensitivity reactions and arthralgia. We further analyzed the impact of different subgroups on specific SOCs. Univariate logistic regression analysis found that, compared with other systemic AEs, the age, weight, sex, and time of onset of the adverse reaction had no statistically significant correlation with the three SOCs mentioned above (p > 0.05) (Supplemental Table S1–S3).
At the PT level, we identified 79 significant disproportionations in the PT that conformed to all four algorithms. The most reported symptoms were Arthralgia, Injection Site Reaction, Injection Site Erythema, Injection Site Swelling and Rash, which were consistent with the drug instructions. In addition, we identified unexpected major AEs, including Lymphadenopathy, Serum Sickness and Tunnel Vision. They belong to Blood and Lymphatic System Disorders, Immune System Disorders and Nervous System Disorders at the SOC level. The possible reason for these unexpected signals is, first, because of the characteristics of hypersensitivity reactions. Type III hypersensitivity reactions are caused by the deposition of soluble immune complexes at multiple sites on the capillary basement membrane in local or systemic tissues, which activate the complement system and involve platelets, basophils, neutrophils, and other cells, resulting in an inflammatory response and tissue damage characterized by edema, local necrosis, and neutrophil infiltration. Severe cases can result in severe Serum Sickness. However, it may also be due to the symptoms of PKU itself, causing intellectual disability and abnormal cognition and behavior in patients. Drug tolerance and poor treatment efficacy may lead to neurological and immune system disorders. These unexpected signals require further investigation.
To assess the severity and specific safety events related to PAL, we delved deeper into the DME list to identify important positive signals at the PT level. Two signals, Anaphylactic Reaction and Anaphylactoid Reaction, were consistent with the PT signal on the DME list. The SOC level focused on Immune System Disorders, which was consistent with the adverse reaction indications on the drug’s label. To determine whether the risk of PAL-related AEs related to PAL presented a temporal trend, we conducted a Weibull distribution test. Most cases occurred within the first month of PAL (68.76%), and the incidence was considered to decreased over time. Considering the dosage and administration of the drug, this phenomenon may be related to the increased dose of PAL. The recommended initial induction dose of PAL was 2.5 mg, followed by gradual titration until a daily subcutaneous injection of 20 mg was reached. PAL has a high immunogenicity potential and often causes an immune reaction in the early stages of treatment before it slowly desensitizes, which is the goal of the titration process. Twenty milligrams was the most commonly used maintenance dose. Therefore, we obtained a relatively large number of reported cases from subgroups of these two categories. If the patient did not show a significant response to treatment within 16–24 weeks, an increasing dose was considered up to a maximum dose of 60 mg. The use of PAL is a long-term process and AEs rates decrease over time.
Subgroup scenario
To investigate the factors influencing the incidence of PAL-related AEs, we conducted further analyses by subgrouping the patients according to age, sex, dosage, and reporting personnel. In sex subgroups, the most frequent AEs at the PT level was Arthralgia. Women were more likely to be affected by Injection Site Pruritus, whereas Chills were more common in men. Both PTs were classified under General Disorders And Administration Site Conditions at the SOC level, which was found to be the SOC with the highest incidence of PAL-related AEs in our study. In the age subgroups, the high-value SOCs for the minor group that conformed to all four algorithms were Musculoskeletal and Connective Tissue Disorders, while the adult group had Immune System Disorder as high-value SOC. Arthralgia was the most frequent AEs at the PT level. Seven patients aged > 65 years old were reported in the database who received drug treatment. Consumers reported six cases. The AEs were mostly located at the injection site, with the most severe case reporting six AEs. Unlike other age groups, the elderly should pay special attention to Abdominal Distension and Mental Impairment. Further analysis revealed that the minor group was more susceptible to Urticaria than the adult group, which was classified under the SOC of Skin and Subcutaneous Tissue Disorders, likely due to allergic or autoimmune issues.
Currently, PAL treatment is primarily offered to patients diagnosed in early infancy through screening and treatment, at least in the first few years of life. Although the earliest newborn screening began in Massachusetts and Oregon in the United States in 1963, these patients are now in their early 60s. Therefore, all the patients in the elderly group were born before the advent of neonatal PKU screening. However, there are no relevant reports on PAL treatment in the elderly. Therefore, reported cases in elderly patients require further verification.
In the dose group, the PT level, consistent with the four algorithms, was associated with arthralgia in the top three. This indirectly indicates that AE occurred independently of the different doses. Analysis of the important SOC from different dose groups revealed that in the low-dose group, which was also the initial stage of conventional therapy, most adverse reactions were concentrated in the administration site conditions and skin and subcutaneous tissue disorders, including Injection Site Reaction, Injection Site Erythema, and Pruritus. As the dose or duration of medication increases, adverse reactions, including Dyspnea and Headaches, penetrate deeper into the body and are concentrated in the respiratory, immune, and nervous systems. In the reporting personnel group, 85% of the reports were from patient self-reporting. The remaining 15% were healthcare professionals, including physicians, pharmacists, and other healthcare professionals. Among the AEs reported in both subgroups with statistically significant differences, the symptoms reported by patients were mostly intuitive symptoms, such as Arthralgia, Injection Site, whereas the symptoms reported by healthcare professionals were mostly those that required medical diagnosis, such as Anaphylactic Reaction and Flushing. Among the AEs reported by healthcare professionals, Gestational Hypertension and Abortion Spontaneous may be adverse reactions that occur during the treatment of pregnant patients. The drug label also clearly emphasizes that the use of the drug in pregnant patients may cause harm to the fetus, but the approved label does not contraindicate its use during pregnancy. There are insufficient data on the drug-associated risk of adverse developmental outcomes. Pregnant women were excluded from the clinical trials of PAL, and all preclinical pregnancy studies included on the label were performed on laboratory animals without PKU. There have been several reports of favorable outcomes in infants with PKU treated with PAL during pregnancy,31 –33 indicating that PAL is effective in maintaining safe maternal blood Phe levels during pregnancy without deleterious effects on the mother or child. However, experience with PAL during pregnancy remains limited, and additional caution is required.
With regard to the use of drugs in children younger than 16 years of age, the safety and effectiveness of PAL have not been established on drug labels. Surprisingly, we identified 14 patients with PKU aged <16 years who were treated with PAL in the FAERS database. The dosages with the most adverse reactions were 20 and 2.5 mg, and the AEs with the highest frequency were Arthralgia, Injection Site Reaction, Urticaria, Injection Site Erythema, and Injection Site Swelling. However, the clinical use of PAL in adolescents remains under investigation. There are several clear reports in the literature on the treatment effects of drug use in children. Alfadhel and Albarakati 34 reported the case of a 12-year-old patient with PKU who received PAL treatment and was closely monitored for 1 year. Positive therapeutic responses became apparent 6 months after treatment initiation. However, caution is still required regarding adverse reactions.
Based on the ages reported in the FAERS database, we found three cases reported in patients under 1 month of age. Two cases were reported by the physicians. Patient 1 (Primary ID: 156414132), age 7.5 days, experienced AEs, including Injection Site Erythema, Injection Site Pain, Injection Site Reaction, Injection Site Warmth, and Pruritus; however, the treatment outcome was not documented. Patient 2 (Primary ID: 181831991), aged 5 days, developed Foetal Growth Restriction and reported other serious medical events (OT) outcomes. Patient 3 (Primary ID: 197436112), aged 0.5 days, developed Pyloric Stenosis and Vomiting, which resulted in HO. Combined with previous clinical treatment experiences, the three cases reported here were most likely infants born to mothers treated with PAL during pregnancy. Given the limited data sample, it was not possible to draw conclusions regarding the incidence of AEs, and further research in the field of pharmacovigilance is warranted.
Furthermore, our analysis included the TTO of AEs across various subgroups. Statistically significant variations were observed in the TTO of adverse reactions among different reporter types (p = 0.056). Possible explanations for these differences may be primarily due to differences in the medical knowledge and understanding of symptoms between patients and healthcare providers.
Drug combination scenario
Cetirizine and Famotidine were the most frequently prescribed medications in conjunction with PAL, suggesting that healthcare providers aim to mitigate AEs associated with PAL. The analysis included 549 patients who received PAL in combination with Cetirizine. On comparing the outcomes of PAL monotherapy with those of combination therapy, Arthralgia remained the most frequently reported AE at the PT level. According to the ROR and PRR algorithms, there were 17 significantly disproportional PT items, with the top 5 being Throat Tightness, Palpitations, Weight Increase, Hyperhidrosis, and Pharyngeal Swelling. The SOC levels identified using the ROR and PRR algorithms were used for Cardiac Disorders. No significant incongruities were observed between SOC and PT across any of the four criteria. AEs were significantly reduced compared with medication alone (Figure S2A). On the other hand, the analysis included 911 patients who received PAL in combination with Famotidine. Similar to the Cetirizine group, Arthralgia was the most frequently reported AE when PAL monotherapy and combination therapy were compared. The ROR and PRR algorithms identified 25 significantly disproportional PT items, with the top five being Chest Discomfort, Throat Tightness, Facial Swelling, Paraesthesia, and Weight Increase. The SOC terms conforming to the ROR and PRR criteria were Cardiac Disorders and Congenital, Familial And Genetic Disorders (Figure S2B).
Limitations
This study had certain limitations. First, the disproportional analysis was simple and sensitive, but susceptible to false positives, indicating that the identified AEs signals represent potential correlations that require further investigation. Second, the FAERS database contains limited and often incomplete information, which may introduce population bias. For instance, in this study, consumers were the primary reporters (85.0%), but they lacked professional medical knowledge; 87.6% of the reports lacked body weight data, and 85.3% of the reports did not report case outcomes, which might have led to reporting bias. In addition, the number and reliability of drug reports in children, pregnant women, and the elderly are insufficient, and the results of the vigilance analysis for drugs may require more information. Therefore, additional clinical studies and long-term data are required to confirm these findings.
Conclusion
In conclusion, we performed a comprehensive and extensive analysis of drug reactions to PAL using the FAERS database’s spontaneous reporting system, which encompasses a broad population and substantial data volume. The signals of drug reactions worthy of clinical attention were revealed, and the primary AEs identified included Arthralgia, Injection-site reactions, and Hypersensitivity. Our analysis also highlighted several unexpected signs, such as Lymphadenopathy, Serum sickness, and Tunnel vision. In addition, we conducted a subgroup analysis to discuss the use of PAL in key populations, such as minors, the elderly, and pregnant women. Given the limited number of reported cases, continued monitoring and further experimental and clinical studies are needed to comprehensively evaluate and ensure the safe use of PAL in all patients.
Supplemental Material
sj-docx-1-smo-10.1177_20503121251330187 – Supplemental material for Evaluating adverse events of pegvaliase-pqpz in phenylketonuria treatment: A comprehensive safety assessment
Supplemental material, sj-docx-1-smo-10.1177_20503121251330187 for Evaluating adverse events of pegvaliase-pqpz in phenylketonuria treatment: A comprehensive safety assessment by Kai Yan, Li Zhang, Guang Yang, Lixuan Yan, Li Wu, Peifeng He and Qi Yu in SAGE Open Medicine
Supplemental Material
sj-tif-2-smo-10.1177_20503121251330187 – Supplemental material for Evaluating adverse events of pegvaliase-pqpz in phenylketonuria treatment: A comprehensive safety assessment
Supplemental material, sj-tif-2-smo-10.1177_20503121251330187 for Evaluating adverse events of pegvaliase-pqpz in phenylketonuria treatment: A comprehensive safety assessment by Kai Yan, Li Zhang, Guang Yang, Lixuan Yan, Li Wu, Peifeng He and Qi Yu in SAGE Open Medicine
Supplemental Material
sj-tif-3-smo-10.1177_20503121251330187 – Supplemental material for Evaluating adverse events of pegvaliase-pqpz in phenylketonuria treatment: A comprehensive safety assessment
Supplemental material, sj-tif-3-smo-10.1177_20503121251330187 for Evaluating adverse events of pegvaliase-pqpz in phenylketonuria treatment: A comprehensive safety assessment by Kai Yan, Li Zhang, Guang Yang, Lixuan Yan, Li Wu, Peifeng He and Qi Yu in SAGE Open Medicine
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
