Abstract
Purpose:
Baclofen is a muscle relaxant that could carry the risk for neurological adverse events (nAEs). We aim to analyze the nAE profile of frequently used baclofen for its clinical application.
Methods:
Our research is a disproportional analysis based on the FDA Adverse Event Reporting System (FAERS) database. We obtained adverse event reports of baclofen from January 2004 to June 2023 from the FAERS database. Reporting odds ratio (ROR) and Bayesian confidence propagation neural network (BCPNN) were performed to find nAE associated with baclofen.
Results:
We extracted a total of 9305 nAE cases with baclofen as the primary suspect. The patients were mostly under 18 years old (45.49%), with markedly more males (43.72%) than females (36.52%). In all, 7275 (78.18%) reports were from the United States. There were 2023 of 9305 (21.74%) reports that occurred serious adverse events (SAE). The median onset time of reports with baclofen-related nAE was 10 days (interquartile range (IQR) 1–112). Intrathecal injection (75.78%) was more than oral (23.06%). We detected 119 significant signals, among which the higher frequencies were somnolence (ROR: 4.69, 95% CI (4.37, 5.03); IC: 2.17, IC025 (2.06)), tremor (ROR: 2.76, 95% CI (2.50, 3.04); IC: 1.43, IC025 (1.29)), lethargy (ROR: 6.37, 95% CI (5.71, 7.10); IC: 2.61, IC025 (2.45)), status epilepticus (ROR: 8.71, 95% CI (7.07, 10.73); IC: 2.98, IC025 (2.69)), generalized tonic-clonic seizure (ROR: 3.17, 95% CI (2.54,3.95); IC: 1.62, IC025 (1.30)], and cerebrospinal fluid leakage (ROR: 229.56, 95% CI (197.76, 266.47); IC: 6.61, IC025 (6.43)]. Unexpected significant nAE might also occur, such as intracranial hypotension (ROR: 428.52, 95% CI (355.18, 517.00); IC: 6.75, IC025 (6.56)], cognitive disorder (ROR: 2.65, 95% CI (2.21, 3.19); IC: 1.38, IC025 (1.11)], anterograde amnesia (ROR: 7.35, 95% CI (2.74, 19.72); IC: 1.69, IC025 (0.90)], metabolic encephalopathy (ROR: 14.77, 95% CI (10.40, 21.00); IC: 3.55, IC025 (2.93)], and myoclonus (ROR: 5.98, 95% CI (4.70, 7.59); IC: 2.46, IC025 (2.13)].
Conclusion:
Given the wide use of baclofen, clinicians should be well-informed about important potential nAE. Although disproportional analysis is a refinement approach, it is still necessary to be vigilant about the nAE of baclofen. It is extremely crucial to early monitoring, especially in minors and the initial stage following the commencement of use.
Introduction
Chronic spasticity can impede all facets of a patient’s daily independence, affecting their ability to perform basic tasks such as hygiene, ambulation, comfort, and sleep. Long-term spasticity can lead to serious and debilitating injuries, potentially stripping patients of their mobility. Baclofen has remained a primary pharmacological therapy for the treatment of spasticity for a long time, particularly for individuals suffering from multiple sclerosis and spinal cord injury.1,2 Baclofen, as a lipophilic compound, functions as a γ-aminobutyric acid (GABA) derivative, efficiently interfering with the release of excitatory amino acids by binding to presynaptic aminobutyric acid receptors at the nerve endings of the spinal cord. This results in the suppression of both monosynaptic and polysynaptic reflexes of the spinal cord, thereby providing relief from spasticity in skeletal muscles. 3
As baclofen becomes increasingly utilized, people’s recognition of its safety concerns is steadily growing, prompting greater clinical attention toward the rising number of neurological adverse events (nAE) attributed to its use. A real-world study by Campbell et al. 4 in the United States showed that there were 126 adverse events, most of nAE, during the use of baclofen in 21 patients. The research in South Korea discovered that the risk of nAE was found to be 7.0% due to baclofen administration in patients with renal impairment. 5 A randomized controlled study spanning 1 year revealed that among 17 children using baclofen, 3 experienced severe nAE, resulting in significant disability. 6 Cases of neurotoxicity associated with baclofen often result from iatrogenic causes. Despite several studies highlighting the link between baclofen and nAE, unfortunately, providers have failed to carry out adequate and comprehensive research on nAE, leading to a widespread lack of awareness among individuals regarding the signs and symptoms of baclofen-induced neurotoxicity.7–10
The US FDA Adverse Event Reporting System (FAERS) gathers adverse event reports from medical practitioners, pharmacists, pharmaceutical corporations, and consumers worldwide, and serves as the main source of data for post-marketing safety surveillance and drug evaluation. 11 FAERS, a spontaneous reporting system, has broad population coverage and good cost-effectiveness. 12 Analyzing these reports in a more natural environment can yield more realistic results, offering crucial evidence to safeguard or enhance the safety of approved drugs.13,14 The disproportionality analysis emphasizes differences in the proportion of AE reports. If no relevance exists between the drug in question and the AE, the reporting rate will be similar to the average reporting rate for all other drugs. Conversely, a higher-than-average reporting rate suggests an association between the drug and AE. 15 To systematically understand the nAE related to baclofen and ensure the safe treatment of patients, we mined the nAE related to baclofen using disproportionality analysis in FAERS.
Materials and methods
Data sources and processing
This is a disproportionality analysis based on FAERS. The data of this study were obtained from the FAERS database. The adverse events in the FAERS database are updated quarterly. The FAERS database consists of seven datasets, including patient demographic (DEMO), drug information (DRUG), report sources (RPSR), indications for drug administration (INDI), reaction (REAC), therapy (THER), and outcome (OUTC). 16 DEMO dataset contains patient demographic and administrative information, a single record for each event report. The DRUG dataset contains drug/biologic information for as many medications as were reported for the event. RPSR dataset contains report sources for the event. INDI dataset contains all “Medical Dictionary for Regulatory Activities” (MedDRA) terms coded for the indications for use (diagnoses) for the reported drugs. REAC dataset contains all MedDRA terms coded for the adverse event. THER dataset contains drug therapy start dates and end dates for the reported drugs. OUTC dataset contains patient outcomes for the event. We imported data from DEMO, REAC, DRUG, OUTC, and THER into MySQL, and deduplication was applied before conducting any analysis. In line with the FDA’s recommendations, we excluded duplicate reports through the following steps. First, for records with identical CASE IDs, we selected the one with the most recent FDA_DT. Second, in cases where both the CASE ID and FDA_DT were the same, we opted for the record having the higher PRIMARYID.
All reports submitted to the FAERS database between January 1, 2004 and June 30, 2023 were downloaded. Subsequently, the raw data were managed locally using My SQL (version 8.0.28). In our study, we enrolled reports with the generic name of baclofen, as well as its trade names, including LIORESAL, GABLOFEN, OZOBAX, LYVISPAH, and FLEQSUVY. Only reports where the role code was the primary suspect were retained for further analysis
The MedDRA (version 25.0) was used to classify nAE reports. MedDRA has five hierarchical levels including system organ class (SOC). 17 Only adverse events with primary SOC being nervous system disorder were selected to ensure uniqueness in SOC classification in this study.
We established specific inclusion and exclusion criteria. Inclusion criteria were as follows: (1) generic name is baclofen, trade name including LIORESAL, GABLOFEN, OZOBAX, LYVISPAH, and FLEQSUVY; (2) Only reports where the role code was the primary suspect; (3) adverse events with primary SOC are nervous system disorder. Exclusion criteria were as follows: (1) reports where the role code was the secondary suspect, concomitant, and interacting and (2) adverse events with primary SOC are not nervous system disorder.
Signal detection methods
Disproportionality analysis is a widely adopted method for detecting signals of adverse events globally, which can be divided into frequency method and Bayesian method according to different statistical principles. The frequency method, such as the reporting odds ratio (ROR) method, boasts high sensitivity and a straightforward principle; however, its specificity is relatively low, making it susceptible to generating false-positive signals. Although the Bayesian method, such as the Bayesian confidence propagation neural network (BCPNN) method, exhibits average sensitivity, it possesses high specificity and is capable of yielding effective results even in the presence of missing data, thereby minimizing false positive signals.18,19 To reduce bias caused by a single method and enhance the reliability of the results, we used two methods for signal detection, including ROR and BCPNN. 20 The criteria of positive signals are as follows: the number of nAE reports is greater than or equal to 3, the lower 95% confidence interval of ROR is greater than 1, and IC025 is greater than 0.
Statistical analysis
Disproportionality analysis was used to analyze the nAE related to baclofen in the FAERS database. My SQL (version 8.0.28) and SPSS (version 26.0) were used to process the original data. All baseline data underwent descriptive statistical analysis utilizing SPSS. Our focused analysis concentrated on the onset time and dosage, where we employed mean and standard deviation (SD) if they followed a normal distribution. Conversely, if they did not follow a normal distribution, we utilized the median and interquartile range (IQR).
Results
Clinical characteristics
The characteristics of nAE-related reports submitted for baclofen are described in Table 1. From January 1, 2004 to June 30, 2023, a total of 21452 adverse event reports with baclofen as the primary suspected drug were included in the FAERS database, of which 9305 cases (43.38%) were reported as nAE. An analysis of the gender subset revealed that a higher percentage of male patients (43.72%) reported nAE associated with baclofen compared to female patients (36.52%). More cases of baclofen-related nAE were observed in the under 18-year-old patient group (45.49%). The United States has reported the highest report number of nAEs related to baclofen (78.18%). The FDA defines serious adverse events (SAE) as death, disability, life-threatening, hospitalization—initial or prolonged, congenital abnormalities. 21 In the nAE related to baclofen, a total of 2023 cases (21.74%) were classified as SAE.
Demographic characteristics of the nAE related to baclofen.

Events onset time
We have analyzed the onset time of nAE caused by baclofen defined as the difference between start-dt and event-dt, as shown in Figure 1(a). It is worth noting that nAE mostly occurred within the first week (47.59%), with a median onset time of 10 days (IQR 1, 112) followed by 8.65% for 8–14 days, 6.66% for 15–30 days, 4.83% for 31–60 days, 14.48% for 61–180 days, 5.66% for 181–365 days, and 13.64% for more than 365 days.

Events onset time and the dosage in different administration routes. (a) Events onset time for baclofen-associated nAE; (b) the dosage in different administration routes for baclofen-associated nAE. mcg: micrograms; mg: milligrams.
Administration and dose
We conducted a statistical analysis of the administration and dose in the nAE related to baclofen. Among them, a total of 1314 (23.06%) reports were reported for oral, 4317 (75.78%) were reported for intrathecal injection, and 66 (1.16%) were reported for unknown routes due to incomplete data. The median dose of intrathecal injection was 317.40 micrograms (IQR 125, 661), while for oral and other routes, the median dose was 30 mg (IQR 15, 100). The dose of baclofen in different administration routes is shown in Figure 1(b).
Signal analysis results
A total of 119 positive nAEs were detected by ROR and BCPNN, as shown in Supplemental Table S1. The top 30 occurrence frequencies of baclofen-associated nAE were analyzed, resulting in detection signals as depicted in Figure 2. The top five ranked signals of event count are muscle spasticity, hypertonia, somnolence, hypotonia, and encephalopathy (muscle spasticity and hypertonia are also indications of baclofen). We not only detected nAE as stated in the label such as somnolence, sedation, tremor, status epilepticus, and cerebrospinal fluid leakage but also detected intracranial hypotension, cognitive disorder, anterograde amnesia, metabolic encephalopathy, and myoclonus that were not included in the label.
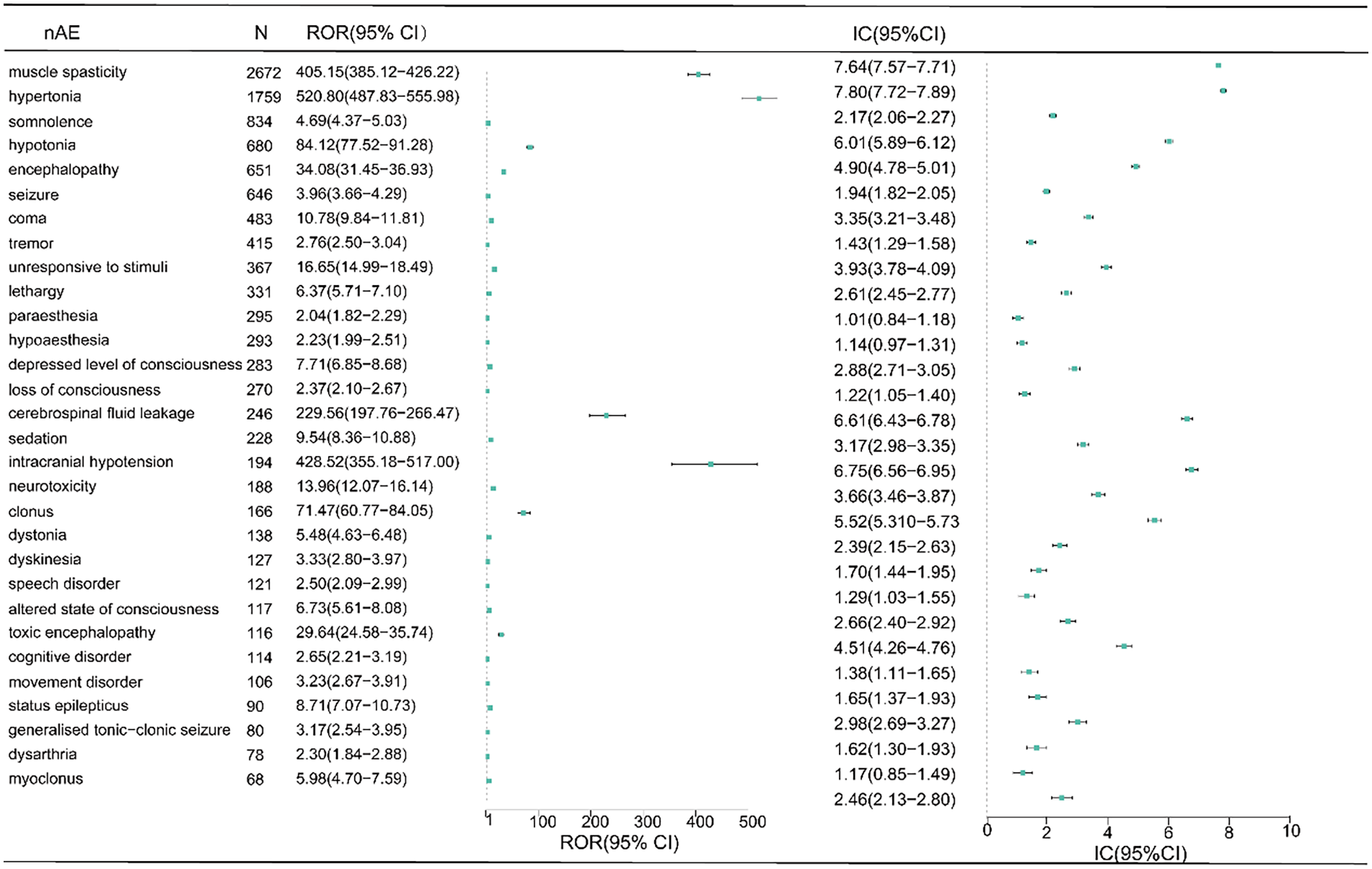
Ranking of top 30 occurrence frequencies of baclofen-associated nAE.
We generated an encyclopedic scatter plot to examine the relationship between the ROR and significant differences, focusing on the association between baclofen and nAE. The generated scatter plot is shown in Figure 3. In Figure 3, the x-axis represents the natural logarithm of the ROR (lnROR). The positive x-direction indicates that nAE associated with baclofen was reported more than other nAE. The y-axis represents the negative logarithm of the p-value (−log10(p-value)] from Fisher’s exact test. The positive y-direction represents a strongly significant difference. In other words, for nAE plotted in the upper right side of this figure, both the ROR and the significant differences were larger. We defined such nAE as high-signal nAE. 22 From the scatter plot, clonus, neurotoxicity, altered state of consciousness, toxic encephalopathy, and dystonia appear as high-signal nAE. In addition, status epilepticus, myoclonus, and metabolic encephalopathy are also considered as nAE with significant ROR and statistical differences.

Scatter plot of nAE related to baclofen. This figure shows the relationship between nAE and baclofen suspected to have caused them. We constructed this scatter plot by plotting the negative logarithm of the p-value (Log10p) from Fisher’s exact test on the y-axis and the natural logarithm of the ROR (lnROR) on the x-axis. In this scatter plot, the signals are larger for the points (drugs) plotted in the upper right corner. The purple-to-yellow colors represent a more significant statistical significance of the nAE.
Discussion
This study showed that there was a total of 21,452 cases of adverse events with baclofen as the primary suspect drug, of which 9305 (43.38%) were reported as nAE. Serious nAE was found in 2023 (21.74%) of 9305 patients, indicating that baclofen-related nAE was serious. Kimberly conducted a study on the trends in baclofen exposure reported to US poison centers from 2014 to 2017, which revealed a 43% increase in intentional suspected suicide attempts involving isolated baclofen exposure. 23 A study utilizing the FAERS public dashboard compared the incidence rates of dizziness, syncope, and confusion between tizanidine and baclofen and found that the incidence rates of dizziness (18.55% versus 16.07%), syncope (3.43% versus 3.28%), and confusion (8.47% versus 3.61%) were higher for baclofen compared to tizanidine. 24 This emphasizes the need for medical professionals to monitor these nAE closely.
Some studies showed that baclofen-related nAEs are more likely to happen to elderly patients who have kidney disease. 25 It is worth noting that the results of this study showed that patients under 18 years old account for the highest proportion (45.49%). We suspect that the possible reason for this phenomenon is drug abuse. The abuse of baclofen constitutes a relatively prevalent phenomenon, occurring not only in the United States and Singapore but also in the United Kingdom and several European nations.26,27 An Italian teenage girl experienced repeated comas due to an overdose of baclofen consumed for recreational purposes. 28 Baclofen is known on the Internet as “an interesting drug that feels pretty good to some extent.” 29 In Perry’s study, 8 out of 14 adolescents consumed an unknown dose of baclofen for entertainment purposes. 30 A literature review from France showed that between 1983 and 2010, there were five articles describing adolescents using baclofen for entertainment, with a total of 19 patients and an average age of 17 years old. 31 The cheap price and online sales of baclofen increase drug abuse. 32 The Internet’s promotion of baclofen, along with its widespread and affordable availability, may inadvertently increase its misuse among minors. This misuse may further lead to baclofen dependence among minors and escalate the risk of nAE among them. Furthermore, patients under the age of 18 are more susceptible to adverse drug events compared to adults, owing to distinct differences in drug pharmacokinetics and pharmacodynamics, their immature organ function which increases their vulnerability to external factors, varying pharmacokinetic and pharmacodynamic characteristics across different age and weight groups, as well as potential disparities in disease characteristics, progression, and etiology. 33
Our study suggested that the onset time of nAE related to baclofen varies, with the shortest being 1 day and the longest being 5075 days. 47.59% of patients’ onset time was within 7 days of taking baclofen, while 32.94% of patients’ onset time was within 1 day of taking baclofen. Considering that the onset time is mostly within 7 days, we believe this could be caused by baclofen withdrawal syndrome. Diagnosing baclofen withdrawal can be challenging due to its nonspecific symptoms, which mimic those of sepsis and serotonin syndrome. 34 Following the interruption of baclofen therapy, patients typically experience withdrawal symptoms within a few hours to days. These symptoms often manifest as excessive neurologic, autonomic, and psychiatric excitation due to the abrupt cessation of baclofen's inhibitory effects on the central nervous system.35–37 Furthermore, although 50% of nAE occur within 10 days, 75% of nAE take place within 112 days. This suggests that in clinical practice, we should not only pay attention to nAE in the early stage of treatment but also be vigilant about the possibility of delayed nAE occurring after taking baclofen. Gracies’ research suggests that compared to oral, small doses of intrathecal can reduce the incidence of adverse events in the nervous system. 38 However, our research indicates that both oral and intrathecal injection can cause nAE, and intrathecal injection causes more nAE than oral. This phenomenon may occur due to the abrupt termination of intrathecal injections, resulting from human errors, pump malfunctions, or knotting of the infusion tube. Any of these scenarios can cause baclofen to be abruptly discontinued, leading to excessive excitation of the central nervous system and severe muscle spasms.39,40 The recommended dose of oral baclofen for the treatment of spasticity is 5 mg three times a day, with a maximum daily dose of 80 mg. 41 The screening dose for intrathecal injection is 50 µg, with a maximum screening dose of 100 µg. Adult maintenance dose increases by 10% to 30% daily, while child maintenance dose increases by 5% to 15% daily. 42 Studies have shown that the nAE of baclofen not only occurs in excess but also occurs at routine doses. 43 Our research findings also indicate that nAE may occur at any dose, whether oral or intrathecal injection. The high lipophilicity of baclofen allows for it to easily cross the blood–brain barrier even at low doses, potentially causing nAE. 44 Khazneh et al. 45 reported a case of encephalopathy in a patient 12 h after taking a single 25 mg tablet of baclofen, whereas Dias et al. 46 described toxicity in a patient with normal renal function after ingesting 20 mg of baclofen.
Among the detected positive signals, somnolence, tremor, lethargy, and other peripheral nervous systems were more common. Status epilepticus, generalized tonic-clonic seizure, and cerebrospinal fluid leakage included in the label also had higher occurrence frequency and stronger signals. Baclofen, a derivative of GABA, has antispasmodic effects by activating GABA receptors to inhibit the transmission of synaptic reflexes. It has antiepileptic effects at lower concentrations, but the mechanism by which it induces epilepsy is unknown. In 1992, The Lancet reported the first case report of baclofen overdose-induced status epilepticus in a patient with focal seizures followed by systemic seizures, with an incubation period of 6 h. 47 Subsequent cases have reported that baclofen causes seizures, including generalized tonic-clonic seizure, myoclonus, non-convulsive status epilepticus, and myoclonic status epilepticus.48–50 Previous research suggests that baclofen may cause seizures, possibly due to its influence on both presynaptic and postsynaptic inhibitory interneurons, shifting the neuronal balance toward excitation and lowering the seizure threshold.43,51 Once epilepsy occurs, anti-epileptic symptomatic treatment and drug withdrawal can be taken to deal with it. In severe cases, peritoneal dialysis or hemodialysis should be taken. The frequency of cerebrospinal fluid leakage is high and the signal is strong. A study from 2023 concluded that cerebrospinal fluid leakage caused by intrathecal baclofen would limit the effectiveness. They investigated 157 patients who received intrathecal injections of baclofen, and the incidence of cerebrospinal fluid leakage was 19%. Tobacco use and younger age were risk factors for cerebrospinal fluid leak. 52
Unexpected nAEs include intracranial hypotension, cognitive disorder, anterograde amnesia, metabolic encephalopathy, and myoclonus. The incidence of perioperative adverse events was increased due to the occurrence of a cerebrospinal fluid leak. Intracranial hypotension arises due to decreased cerebrospinal fluid volume with a variable clinical presentation ranging from postural headaches to coma, which can be caused by spontaneous or post-traumatic dural lacerations leading to CSF leakage. Iatrogenic factors contributing to this condition include myelography, lumbar puncture, excessive drainage of ventricular or spinal shunts, spinal anesthesia, or the placement of a baclofen pump. Sometimes, the symptoms of intracranial hypotension can be resolved through conservative management. However, if they persist, epidural blood patching has proven to be highly effective in addressing them. 53 Amnesia has been observed as a rare adverse effect in clinical trials involving intrathecal baclofen, albeit without further elaboration. In addition, memory impairment has been occasionally noted as a symptom of chronic baclofen intoxication syndrome. 54 Grande reported a case of severe anterograde amnesia caused by intrathecal baclofen. Until that time, transient global amnesia associated with baclofen had not been reported in the literature. They believe that the anterograde amnesia was caused by an abrupt reduction in hippocampal nitric oxide (NO) synthesis, leading to a prolonged decrease in NO concentration. This decrease in NO concentration acted as a physiological switch, disrupting hippocampal GABAergic neurotransmission and memory acquisition. These episodes were sometimes triggered by the release of stress hormones following sympathetic stimuli. Intrathecal baclofen contributed to this process by reducing baseline GABAergic synaptic transmission in the hippocampus. The patient may have inherited a vulnerability to anterograde amnesia within the NO-GABA signaling pathway. 55 The precise mechanism underlying anterograde amnesia linked to baclofen requires additional research for definitive confirmation. Metabolic encephalopathy is a statistically significant nAE with a high signal value (ROR = 14.77, 95% CI (10.40, 21.00); IC = 3.55, IC025 = 2.93]. In the early stages of metabolic encephalopathy, global symptoms encompass disturbances in consciousness, confusion, disorientation, and delirium. Furthermore, a range of autonomic nervous system symptoms may appear, including insomnia and nausea. Subsequently, it may progress to epileptic seizures, oral and facial automatisms, pathological reflexes, myoclonus, tremor, and coma. In the most severe stages, decerebrate or decorticate rigidity may arise, leading to a deeper coma and ultimately, death. 56 A literature study of 14 patients with metabolic encephalopathy after taking baclofen concluded that even at moderate doses baclofen can cause metabolic encephalopathy. It is especially important to discontinue the use of baclofen and benzodiazepines after the development of metabolic encephalopathy. 43
Currently, there are no specific guidelines to guide the precise dosage adjustment of baclofen to prevent the occurrence of nAE. For patients with renal impairment, some studies suggest that the dosage of baclofen should be adjusted according to the estimated glomerular filtration rate (eGFR) to reduce adverse events. For patients with mild chronic kidney disease (eGFR > 60 mL/min), they recommend adhering to the lowest effective dose of baclofen as indicated in the manufacturer’s labeling. Conversely, for patients with moderate or severe chronic kidney disease (eGFR < 60 mL/min), due to insufficient evidence to support safe dosing practices, they advise against the use of baclofen and encourage the exploration of safer alternative options if available. 57 Furthermore, in case of an adverse event, hemodialysis can be employed to eliminate baclofen. A hemodialysis session lasting 4 h is capable of removing 79% of the drug, effectively reducing its half-life from 15.5 h to just 2.06 h. 41
Our research also has some limitations inevitably. First, spontaneous reporting tends to underreport adverse events, especially for less common or SAE Second, due to the inherent limitations of the spontaneous reporting system, we were unable to collect specific laboratory test values, comorbid conditions of patients, and drug–drug interaction. Missing data and incorrect adverse event coding can lead to biased results. Third, factors such as competition bias, co-prescription bias, region bias, and channeling bias can all contribute to some degree of bias in the final results. 58 In addition, due to the high number of missing values for factors such as gender, age, and indications in this study, an analysis adjusted for potential confounders was not conducted. Finally, the signal detected by the disproportionality analysis can only indicate that the drug has a certain statistical correlation with the signal. The specific causal relationship needs to be determined by more prospective research such as cohort studies or randomized controlled studies.
Conclusion
Widespread use of baclofen has led to concerns about safety issues, particularly regarding nAE. Despite some limitations, the FAERS database can effectively identify nAE associated with baclofen and provide crucial clinical insights. The common nAEs (somnolence, tremor, lethargy, cerebrospinal fluid leakage, status epilepticus, and generalized tonic-clonic seizure) are consistent with the label. Furthermore, our research identified several unexpected nAEs, such as intracranial hypotension, cognitive disorder, anterograde amnesia, and metabolic encephalopathy, each necessitating meticulous clinical differentiation and diligent management. It is extremely crucial to early monitoring, especially in minors and the initial stage after taking baclofen. This study can offer valuable insights for informed prescribing practices. In the future, it will remain necessary to conduct long-term, large-sample follow-up observational studies on nAE associated with baclofen, while paying close attention to SAE, onset time, dosage, and drug–drug interaction.
Supplemental Material
sj-docx-1-smo-10.1177_20503121251331812 – Supplemental material for Neurological adverse events associated with baclofen: A disproportionality analysis based on FDA Adverse Event Reporting System
Supplemental material, sj-docx-1-smo-10.1177_20503121251331812 for Neurological adverse events associated with baclofen: A disproportionality analysis based on FDA Adverse Event Reporting System by Ni Zhang, Tingting Jiang, Yanping Li, Pei Guo, Yumeng Liu, Yu Zhang and Yao Liu in SAGE Open Medicine
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
