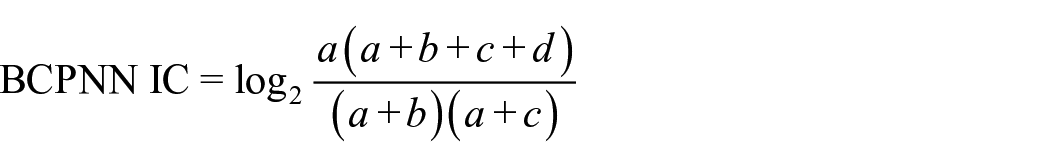Abstract
Background:
The potential association between new antineoplastic drugs and an increased risk of suicide-related adverse drug reactions remains unclear. This study aims to utilize the FAERS public database to analyze suicide-related adverse drug reactions associated with common antitumor drugs and to investigate potential risk signals for such adverse drug reactions.
Methods:
This study was a retrospective analysis utilizing the FAERS database. The FAERS database was examined for reports of suicide-related adverse events associated with antitumor drugs, spanning from 2004 to 2023. To identify and verify adverse event signals, we employed reporting odds ratios, proportional reporting ratios, and Bayesian methods (Bayesian Confidence Propagation Neural Network). Additionally, logistic regression analysis was performed to assess outcomes in tumor patients.
Results:
A total of 223,781 suicide-related adverse event reports were screened, of which 3790 involved common antitumor drugs. The top five drugs reported were tretinoin (n = 1220), methotrexate (n = 664), celecoxib (n = 505), rituximab (n = 107), and imatinib (n = 105). Risk signal analysis indicated that, with the exception of tretinoin (ROR = 6.317), the reporting odds ratio values for the other drugs were below 2. Among cancer patients, the most frequently reported adverse events included suicidal ideation (n = 233), completed suicide (n = 131), and suicide attempts (n = 97). Regression analysis revealed that risk factors for patient death included indication (OR = 0.967, p < 0.01), gender (OR = 0.57, p < 0.01), and type of adverse event (OR = 4.644, p < 0.01).
Conclusion:
The findings suggest that antineoplastic drugs may not statistically increase the risk of suicide-related adverse events. However, specific tumor types and suicide-related adverse events may contribute to increased mortality in cancer patients. Further research is warranted to elucidate the risk of suicide-related adverse events in oncology patients.
Introduction
According to statistics from the International Cancer Center, there were 19.3 million new cases and 10 million cancer deaths worldwide in 2020. Cancer has become one of the major diseases that seriously threaten human health globally. 1 Under the influence of various cancer-promoting factors such as accelerated aging, deterioration of the ecological environment, changes in lifestyle, and genetics, the incidence of breast cancer and other tumors has been increasing year by year. 2 Multiple studies have found that, compared with the general population, cancer patients are more likely to experience psychological distress, depression, severe depression, and other mental and psychological problems, including suicidal thoughts.3,4 Cancer patients need to receive antitumor treatment, and with the update of antitumor treatment strategies, these treatments have played a key role in improving the survival rate and quality of life of tumor patients. However, for cancer patients receiving anticancer drugs, drug-related adverse events can increase the risk of psychiatric disorders. In recent years, with the strengthening of drug safety monitoring, more and more studies have begun to focus on the possible psychiatric adverse events caused by antitumor drugs, especially the risk of suicide.
As an extreme and serious outcome, the potential link between suicide and antineoplastic drug use deserves further study. Among cancer patients, suicide risk may be influenced by multiple factors, such as disease severity, psychological stress, pain management, lack of social support, and side effects of medications.5,4 For example, some neurotoxic antitumor drugs may directly or indirectly affect patients’ mental health. 6 Research shows that among cancer patients, the overall mortality rate from suicide is 1.49 times that of the general population. 7 The risk of suicide varies by cancer site, with patients suffering from malignant respiratory cancers having the highest risk. 7 Suicide among cancer patients is a major public health problem, but the risk factors remain unclear.
Therefore, this study aims to explore the relationship between antineoplastic drugs and suicide-related adverse events through a detailed search and analysis of the U.S. Food and Drug Administration (FDA) Adverse Event Reporting System (FAERS) database. We extracted suicide-related adverse event reports from the database, screened out reports related to tumor patients and antitumor drugs, and then conducted descriptive and risk signal analyses to explore the correlation between the two and identify potential risk factors for death.
Methods
Data sources and retrieval
This retrospective study used the U.S. FAERS as the main data source and queried the FAERS database through the open-source pharmacovigilance data extraction, mining and analysis tool OpenVigi l 2.1. Extract suicide-related ADE reports from the FAERS database from the first quarter of 2004 to the third quarter of 2023, without age restrictions, and the search objects are primary suspected drugs and secondary suspected drugs were antitumor agents. The research flow chart is shown in Figure 1.

Flowchart of the study process based on the FAERS database.
Inclusion and exclusion criteria
Inclusion criteria: (1) Reports involving patients with cancer who were treated with antineoplastic drugs; (2) Reports that identified suicide-related ADEs using keywords such as “completed suicide,” “suicidal ideation,” “suicide attempt,” and “suicidal behavior”; (3) Cases with clear documentation of the suspected drug and its role (either as a primary or secondary suspect).
Exclusion criteria: (1) Reports that did not involve oncology patients or antineoplastic drugs; (2) Reports with insufficient information on the drug or unclear identification of suicide-related events; (3) Duplicate or incomplete reports where outcomes or drug roles were not discernible.
Data processing and cleaning
Eliminate duplicate reports, exclude nontarget drugs and uncertain drugs, deal with missing or inconsistent data, and collect and analyze patient indications, gender, age, reporting region, and reporting year reported on antitumor drug ADE in the FAERS database. In the study, system organ classification (system Organ class (SOC)) is used to classify adverse drug event (ADE) data, and the preferred terms of the International Medical Dictionary for Regulatory Activities are used to code the ADE data.
Signal detection method
A fourfold table was used to calculate and compare the association between specific antitumor drugs and the occurrence of ADEs (Supplemental Table 1). Proportional Reporting Ratio (PRR), Reporting Odds Ratio (ROR), and Bayesian Confidence Propagation Neural Network (BCPNN) methods were used to evaluate the relationship between suicide risk and specific antitumor drugs. For PRR and ROR, when their values are greater than 2 and the target ADE report number of the target drug is ⩾3, it is considered that the drug may be related to a specific adverse drug reaction (ADR). 8 For BCPNN, the larger the information component (IC) value in this method, the higher the signal strength. The specific calculation formula is as follows 9 :
Statistical analysis
We conducted a descriptive analysis to identify basic trends and general characteristics of suicide-related adverse events caused by anticancer drugs. Including analysis of the frequency of adverse events, basic characteristics of patients (such as age, gender distribution), and clinical outcomes of suicide-related adverse events. To evaluate the potential characteristics (including tumor indications, type of suicide-related adverse events, age, gender) and other characteristics and death, we performed binary logistic regression analysis. Odds ratios (ORs) and their 95% confidence intervals were used to express the relationship between patient characteristic and the risk of suicide-related death, as the analysis was specifically conducted to assess suicide-related mortality, not all-cause mortality. An outcome model was constructed and the Receiver operating characteristic (ROC) curve was used to evaluate the sensitivity and specificity of the model. All analyzes were performed using Python 3.12 and Graph Pad 9. In this study, a p-value less than 0.05 was considered to be statistically significant.
Results
ADE reporting basic information
A total of 223,781 suicide-related ADE reports were extracted from the FAERS database. Analysis of reporting trends indicates a significant increase in the absolute number of suicide-related ADEs, peaking in 2020 (Figure 2). This trend suggests either an increase in the number of reports or a rise in the actual incidence of such events. Among these, 3790 ADE reports pertained to antineoplastic drugs. Specifically, 499 ADE reports were related to tumor indications, spanning the years 2004–2023. The most frequently reported cancer types were breast, lung, and colon cancer. Suicidal ideation was the most commonly reported event, followed by completed suicides and suicide attempts. Slightly more female patients were reported than male patients. Age information was available in 309 reports, with an average age of approximately 61.4 years, a minimum age of 15 years, and a maximum age of 98 years. The United States accounted for the highest number of reports. Approximately 70.3% of these patients had death as the final outcome (Figure 3).

Trend of suicide-related adverse drug events (ADEs) from the FAERS database.
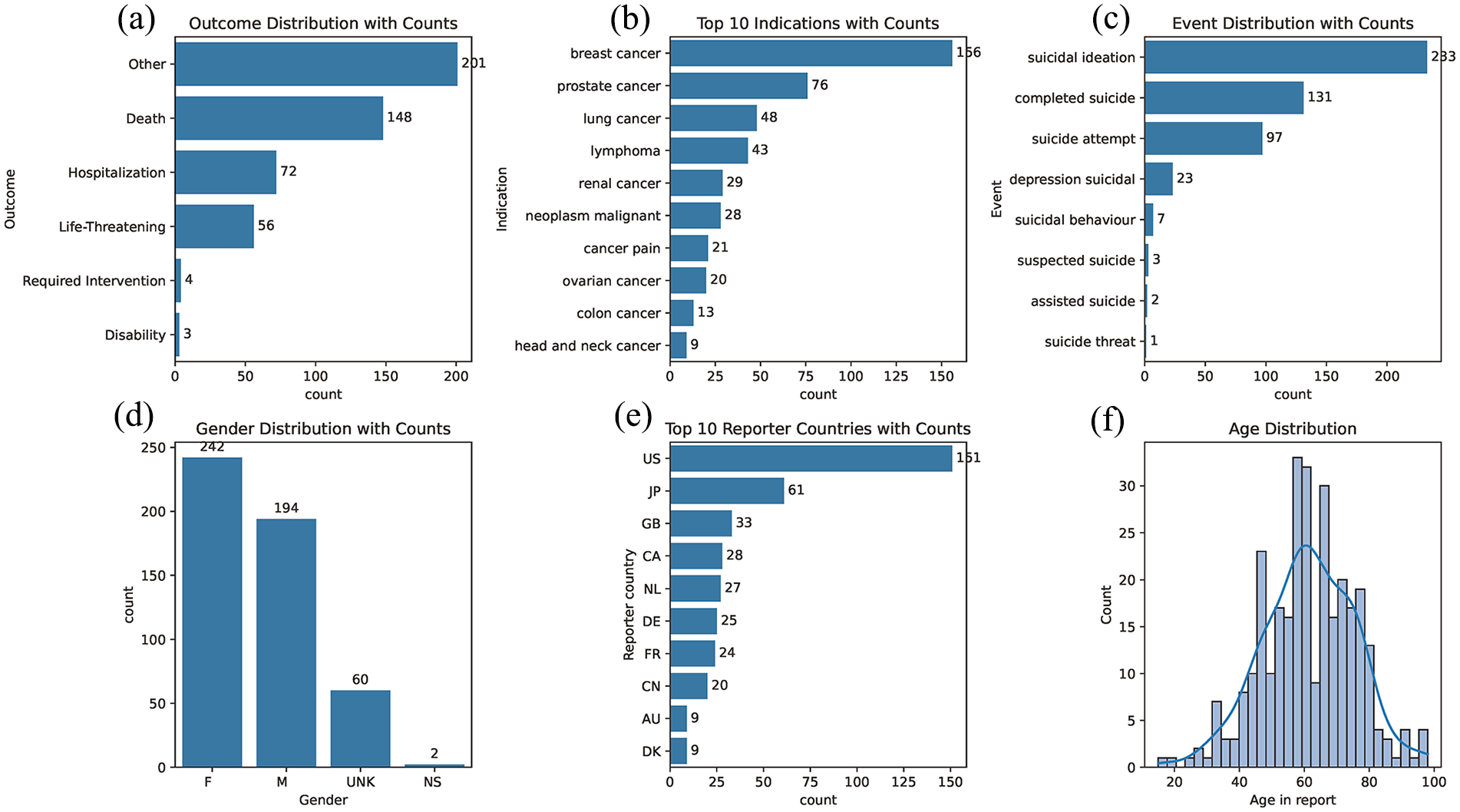
Adverse drug event (ADE) reports for antineoplastic agents: (a) outcome distribution with counts; (b) Top 10 indications with counts; (c) Event distribution with counts; (d) Gender distribution with counts; (e) Top 10 reporter countries with counts; (f) Age distribution
Disproportionality analysis for the association of suicide/self-injury with anticancer agents
A total of 3790 ADE reports related to antitumor drugs were obtained. Antitumor drugs were categorized according to the absolute number of ADEs, and the results are presented in Table 1. Additionally, we conducted an analysis based on the classification of antitumor drugs, including 162 reports for alkylating agents, 105 reports for antibiotics, 862 reports for antimetabolites, 430 reports for protein kinase inhibitors, 248 reports for monoclonal antibodies, 248 reports for platinum compounds, 84 reports for taxanes, and 79 reports for vinca alkaloids. Analysis of the signal intensity for these classified drugs revealed no new safety signals.
Suicide/self-injury signal strength reported at the level of anticancer drugs in the FAERS database.

Analysis of risk factors for death outcomes
After selecting patients whose indications are tumors, a regression analysis was performed to determine the association between various characteristics and the outcome of death. The characteristics analyzed included tumor type, suicide-related adverse event type, gender, age in the report, and reporter country. For the oncology disease category, bladder cancer served as the reference category; for the event type, “assisted suicide” was the reference category; and for gender, female was the reference category. The results of the logistic regression analysis are presented in Table 2. Compared with bladder cancer, the risk of death was significantly reduced for other cancer categories, with the difference being statistically significant (p = 0.027). Other types of suicide-related adverse events were associated with a statistically significant increased risk of death compared with assisted suicide (p < 0.001). Male patients showed a significantly reduced risk of death compared with female patients, with the difference being statistically significant (p < 0.001).
Analysis of risk factors for death outcomes.

P < 0.001 denotes a statistically significant difference.
ROC curve
The performance evaluation results of the model are as follows (Figure 4): The sensitivity was 0.9811, the specificity was 0.8636, Youden’s Index was 0.8448, and the area under the curve was 0.9430 (p < 0.01). The ROC curve demonstrates that the model possesses strong capabilities in distinguishing between positive and negative samples, indicating it is a highly performant model.

ROC curve.
Discussion
As a special group, cancer patients’ mental health is easily affected by the disease and its complications, leading to an increased risk of suicide. 10 Additionally, antitumor drug treatment for cancer patients may increase the risk of ADRs due to the complexity of medication regimens and high side effects. 11 Research suggested that PARP (Poly ADP-ribosepolymerase) inhibitors negatively impacted patients’ emotional and psychological states by affecting neurotransmitter metabolism and altering biomarker levels. These drugs possessed multitarget properties that not only affected tumor cells but also produced side effects on normal cells, potentially increasing the risk of depression and suicidal ideation among patients 12 ; therefore, the medication safety of this group should be taken seriously. In this study, we used FAERS (U.S. FAERS) cases involving suicide-related ADRs to generate basic statistics and red flag analysis.
All suicide-related ADR cases were mined and analyzed for antitumor drugs and tumor indications, including trends, male-to-female ratio, end-point outcomes, danger signal intensity. Our study found that suicide-related adverse events have been on the rise in recent years. Trend analysis ADRs that the percentage of suicide-related adverse events peaked in 2020, which may be related to external factors such as the COVID-19 pandemic and its impact on mental health. 13 After 2020, the number and percentage of cases declined slightly. Among different age groups, the 18-to 64-year-old group was identified as a high-risk group, with the proportion of women being higher than that of men. However, in some age groups (e.g., 3–11 years old), the proportion of men was higher. Further analysis of the data revealed that antineoplastic drugs did not significantly increase the risk of suicide-related adverse events.
Previous research has showed that despite significant advances in cancer treatment in recent decades, cancer patients remain at high risk for suicide. Data from 62 studies encompassing 46,952,813 patients were systematically reviewed and meta-analyzed. It was found that compared with the general population, the suicide mortality rate of cancer patients was markedly higher (standardized death ratio = 1.85, p < 0.05). Suicide risk is strongly associated with cancer prognosis, cancer stage, time since diagnosis, and geographic region. 11 Other studies have examined the risk of suicide death among cancer patients. Through a systematic review and meta-analysis, it was found that the standardized death ratio for suicide death in cancer patients was 1.55. The risk of suicide death varies among different cancer types, highlighting that cancer substantially elevate the risk of suicide and necessitates timely intervention to reduce this risk. 14 A retrospective analysis also found that among 78,650 cancer patients, 89 (0.1%) died by suicide. Of the 14,517 patients who completed the assessment, 69 (0.5%) reported suicidal intent, but none died by suicide. 15 These studies have shown that cancer patients have a significantly increased risk of suicide. In our study, the risk of suicide in oncology patients was not assessed because nononcology patients were not included. However, we examined the relationship between antitumor drugs and suicide-related adverse events. The results showed that no significant increase in suicide-related adverse events was found with either traditional antitumor drugs or newer agents such as immune checkpoint inhibitors and targeted therapies-related adverse events. This is consistent with the adverse events mentioned in the package inserts of these new antineoplastic drugs. It is suggests that the risk of suicide-related adverse events caused by new antitumor drugs is low, but it is necessary to pay attention to the potential impact of factors such as false negatives and low adverse event reporting rates on this result.
In terms of danger signal analysis, FAERS database data analysis shows that there are significant differences in the absolute values of suicide-related adverse events among different anticancer drugs. However, the ROR for these drugs are generally low, suggesting that they are associated with fewer suicide-related adverse effects. Nonetheless, a low ROR value does not imply a causal relationship, and clinical practice still requires adequate attention to the mental health of cancer patients. Additionally, this study analyzed factors related to the death outcomes of cancer patients. Through regression analysis, we found that the type of suicide-related adverse event, tumor type, and gender were significantly associated with the risk of death.
The limitation of this study is that the FAERS database is a spontaneous reporting system, and not all antitumor drug-related adverse events in this study were reported by professional medical personnel, which may result in lower credibility of the reported information. Additionally, the majority of patients in adverse event reports are from the United States, which poses a risk of racial bias. Second, the absence of a formal sample size calculation is another limitation, as the study included all eligible cases from the FAERS database. This approach may have led to an over- or under-representation of certain adverse events due to reporting biases inherent to the database. Third, this study conducted subgroup analysis, but due to limited data, the diversity of patient groups and antitumor treatment regimens may affect the reliability of the results. Fourth, because observational data are used, causality cannot be determined, and the true incidence of adverse events cannot be reflected. Furthermore, the stage of cancer was not considered in this study, which is another significant limitation, as the cancer stage could substantially impact the risk of suicide-related ADRs. A more detailed analysis incorporating cancer stage could provide deeper insights into the risk factors for suicide-related ADRs. Moreover, a key limitation is that the FAERS database does not clearly distinguish deaths due to disease progression from those related to ADRs or suicide. While we focused on deaths specifically linked to suicide-related events (e.g., “completed suicide” or “suicide attempt”) and excluded cases solely from disease progression, some confounding with cancer-related mortality may persist. Despite these limitations, this big data signal mining based on the FAERS database highlights the importance of mental health in cancer patients and supplements the safety data of these drugs postmarket to a certain extent.
Conclusion
This study analyzed the potential association between common antineoplastic drugs and suicide-related adverse events and found that antineoplastic drugs may not be significantly associated with suicide-related adverse events. However, among different cancer types, patients with respiratory cancers had a notably higher incidence of suicide-related adverse events, while patients with certain other cancers, such as bladder cancer, showed lower incidences. These findings suggest the need for targeted interventions based on cancer type. Real-world research is still needed in the future to further discover the impact of new antitumor drugs on the suicide risk of cancer patients.
Supplemental Material
sj-docx-1-smo-10.1177_20503121241308686 – Supplemental material for Analysis of the relationship between antineoplastic drugs and suicide-related adverse events based on the food and drug administration adverse event reporting system database
Supplemental material, sj-docx-1-smo-10.1177_20503121241308686 for Analysis of the relationship between antineoplastic drugs and suicide-related adverse events based on the food and drug administration adverse event reporting system database by Nan Zhao, Huimin Wang, Huilin Xu, Xixian Tang and Dedong Cao in SAGE Open Medicine
Footnotes
Acknowledgements
We are grateful to the U.S. Food and Drug Administration for providing access to the FAERS database, which made this research possible.
Author contributions
Dedong Cao: Conceptualization; Formal analysis; Investigation; Methodology; Data curation; Project administration. Nan Zhao: Conceptualization; Data curation; Writing – review and editing. Huimin Wang: Conceptualization; Methodology; Writing – original draft. Huilin Xu: Conceptualization; Writing – review and editing. Xixian Tang: Conceptualization; Writing – review and editing.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
The study has been approved by the Ethics Committee of Renmin Hospital of Wuhan University, with the approval number WDRY2022-K035. As it is a retrospective study using public databases, consent waiver was obtained from Institutional Review Board.
Informed consent
As it is a retrospective study using public databases, consent waiver was obtained from Institutional Review Board.
Consent for publication
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.