Abstract
Objective:
This study aimed to determine the incidence of postoperative major adverse cardiac events for patients undergoing carotid endarterectomy.
Methods:
This single-center, retrospective study recruited 171 carotid endarterectomy patients between January 1999 and June 2018. Patients who received a carotid endarterectomy in conjunction with other surgery were excluded. The primary outcomes were the incidences of major adverse cardiac events (comprising myocardial infarction, significant arrhythmias, congestive heart failure, and cardiac death) within 7 days, 7–30 days, and > 30 days–1 year, postoperatively. The secondary outcomes were the factors related to major adverse cardiac events and the incidence of postoperative stroke. The patients’ charts were reviewed, and direct contact was made with them to obtain information on their status post discharge.
Results:
The incidences of major adverse cardiac events within 7 days, 7–30 days, and >30 days–1 year of the carotid endarterectomy were 3.5% of patients (95% confidence interval: 0.008–0.063), 1.2% (95% confidence interval: 0.004–0.028), and 1.8% (95% confidence interval: 0.002–0.037), respectively. The major adverse cardiac events occurring within 7 days were arrhythmia (2.3% of patients), cardiac arrest (1.8%), myocardial infarction (1.2%), and congestive heart failure (1.2%), while the corresponding postoperative stroke rate was 4.7%.
Conclusion:
The 7-day incidence of major adverse cardiac events after the carotid endarterectomy was 3.5%. The most common major adverse cardiac event during that period was cardiac arrhythmia.
Introduction
Carotid endarterectomy (CEA) is a treatment for patients with significant carotid artery stenosis. It was first performed successfully in 1953. 1 Several trials have concluded that CEA reduces the risk of ischemic stroke compared with other forms of medical treatment. 2 The North American Symptomatic Endarterectomy Trial (NASCET) 3 demonstrated a reduction in the 2-year postoperative stroke rate for patients with severe stenosis (70%–99%) to 22.2% using best medical treatment, but to only 9% using a CEA. Moreover, the European Carotid Surgery Trial (ECST) 4 found a 3-year stroke and death rate of 14.9% for CEA patients versus 26.5% for patients receiving best medical treatment. In addition, our institution reported that 100 patients who underwent CEA had a 1% perioperative mortality rate and a 3% perioperative, major, adverse neurological event rate. 5
However, the severity of carotid artery stenosis and the extent of coronary artery disease (CAD) are significantly correlated. 2 Importantly, major adverse cardiac events (MACEs)—including myocardial infarction (MI), cardiac arrhythmias, congestive heart failure (CHF), and cardiac death—are significantly correlated with CAD. 6 Consequently, those patients who have undergone a CEA have a higher perioperative cardiovascular risk and an increased risk of long-term mortality versus the general population because of the systemic nature of atherosclerosis.7,8 A previous study demonstrated a rate of 1.7% for postoperative MACEs for this population. 9
However, the variability of surgical and anesthesia techniques might affect cardiovascular outcomes. The investigators aimed to determine the postoperative incidences of MACEs at 7 days, 7–30 days, and >30 days–1 year for CEA patients at a tertiary-care center where the single surgical and anesthesia technique was performed. The secondary outcomes were the incidence of postoperative stroke and the factors associated with MACEs.
Materials and methods
This retrospective, descriptive study included patients’ age > 18 years, undergoing a CEA at a tertiary-care, university hospital between January 1999 and June 2018. The exclusion criteria were the patients who underwent the CEA in conjunction with other surgery, such as a coronary artery bypass graft or aortic surgery in the same setting. The final study population consisted of 171 patients (Figure 1). The patients’ data were retrieved from their medical charts and through direct contact with the individuals concerned to obtain information on their status post discharge. Before the research commenced, approval was obtained from the Siriraj Institutional Review Board, Faculty of Medicine Siriraj Hospital, Mahidol University (

Flow chart of patient enrollment.
Surgical management
Patients were enrolled to the surgical list according to NASCET criteria. The severity of carotid artery stenosis was assessed by either magnetic-resonance angiography (MRA) or computed tomography angiography (CTA). Ultrasonography was also used as an additional tool to assess the flow velocity at the lesion. Six surgeons at our institution performed the CEAs, with routine use of intravascular shunts across the clamp section of carotid artery, and saphenous vein or synthetic patching for arteriotomy closures.
Anesthesia management
The preoperative evaluations were performed in accordance with the 2014 ACC/AHA guidelines for the perioperative cardiac evaluation and management of patients undergoing noncardiac surgery. Cardiologist consultation and proper specific cardiac assessment were made following the guidelines. Every patient was admitted the night before the surgery. The CEAs were performed under general anesthesia, with monitoring following the protocol recommended by the American Society of Anesthesiologists, including non-invasive blood pressure, electrocardiography, pulse oximetry, and temperature. Arterial catheterization was performed before induction of the anesthesia. As to neurological monitoring, intraoperative electroencephalography was not available at our institution. Bispectral index (BIS) and near-infrared spectroscopy (NIRS) were used in some selected cases, depending on the judgment of the anesthesiologist in charge of the patient. The patients were extubated at the end of the surgery for immediate neurological evaluation; they were subsequently transferred to the intensive care unit for close monitoring for 24 h postoperatively.
Data collection
The variables collected were age, gender, body mass index (BMI), ASA classification, medical comorbidities, characteristics and severity of the carotid stenosis and contralateral carotid stenosis, preoperative medications, and laboratory investigations (hemoglobin and creatinine).
Patients without any neurologic symptoms within 6 months before the surgery were considered asymptomatic. The event which indicated surgery (transient ischemic attack (TIA) or stroke, including amaurosis fugax, in the preceding 6 months) was collected for symptomatic patients. Contralateral carotid stenosis was defined as a narrowing of the carotid arteries of ⩾ 50%, based on clinical investigation. Postoperative stroke was defined as any new neurological abnormalities after the surgery, within 7 days, 7–30 days, and 30 days–1 year, 10 excluding metabolic or anesthetics cause, either confirmed by initial computed tomography or clinical manifestation.
The intraoperative hypotensive time ratio was defined as the summation of the intraoperative hypotensive time over the total operative time (minutes). Intraoperative hypotension was defined as a systolic blood pressure < 90 mm Hg or a diastolic blood pressure > 60 mm Hg.
The definitions of the MACEs used were as follows11–13:
MI: ST or non-ST elevation MI; unstable angina
Significant cardiac arrhythmias: dysrhythmia that caused symptoms or hemodynamic instability (such as an atrial flutter, atrial fibrillation, supraventricular tachycardia, ventricular tachycardia, atrioventricular block, or symptomatic bradycardia)
CHF: symptoms, signs, or radiographic findings suggestive of heart failure, including dyspnea, orthopnea, increased jugular venous pressure, pulmonary rales, cardiomegaly, and pulmonary vascular engorgement
Cardiac death: any sudden death presumed to be of cardiac origin
Study objectives
The primary objective was to determine the presence of MACEs (MI, significant cardiac arrhythmia, CHF, or cardiac death) during the postoperative periods of 7 days, 7–30 days, and 30 days–1 year. The secondary outcomes were to describe the risk factors associated with the MACEs and the postoperative stroke rates for the corresponding time intervals.
Statistical analysis
Sample size calculation
Previous study from Go et al.
9
reported MACEs after cardiac endarterectomy were 1.7% in 30 days. After discussion with our statistician, the investigators performed sample size calculation with Association of Public Health Laboratories (APHL): Influenza Laboratory Testing Sample Size Calculation formula, Rare/Novel influenza event detection due to the low incidence of MACE after CEA; Formula: Sample size (
The baseline characteristics and categorical data, described as numbers and percentages, were compared for the patients with and without MACEs. The statistical package used was PASW Statistics for Windows (version 18; SPSS Inc., Chicago, IL, USA). For each time interval, the incidences of the primary and secondary outcomes were calculated, and the 95% confidence intervals (95% CI) were determined. The authors compared age, BMI, gender, ASA classification, characteristics and severity of carotid stenosis, medical comorbidities, preoperative laboratory investigation results, and the intraoperative hypotensive time ratio using chi-square tests and Fisher’s exact test. A
Results
The investigators established incidences of MACEs within 7 days, 7–30 days, and 30 days–1 year of 3.5%, 1.2%, and 1.8%, respectively. At 2.3%, cardiac arrhythmia was the most common MACE to occur within 7 days. Postoperative stroke (rate: 4.7%) only occurred within 7 days postoperatively (Table 1).
Incidence of postoperative, major, adverse cardiac events and stroke.

CHF: congestive heart failure; MI: myocardial ischemia.
As the MACEs were most commonly found within 7 days of surgery, the authors performed a univariate analysis using the 7-day data to identify the factors associated with postoperative MACEs. There were no statistically significant differences in terms of age, gender, BMI, and ASA classification. The majority of patients undergoing CEA were symptomatic with severe stenotic lesion, but with no difference from the asymptomatic patients in terms of the incidence of postoperative MACEs. There were also no differences in comorbidities, including CAD, peripheral arterial disease (PAD), or atrial fibrillation. Moreover, the preoperative medications of the two groups showed no statistically significant differences. Finally, although the intraoperative hypotensive time ratio was slightly higher for the MACE group, it was not statistically different (Table 2).
Characteristics of patients based on the presence of MACEs within 7 days postoperatively.
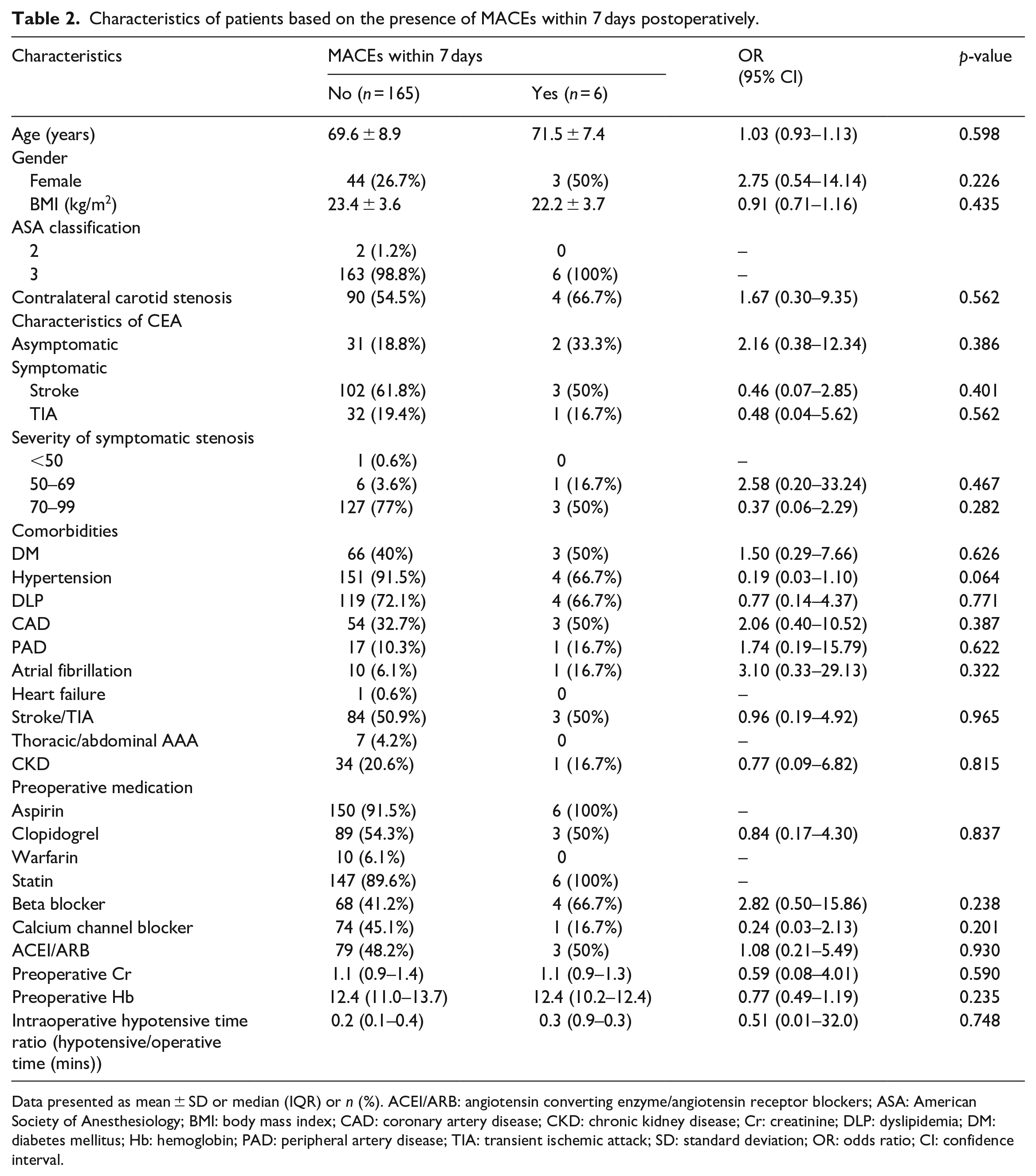
Data presented as mean ± SD or median (IQR) or
Most cardiac arrhythmias occurred early during the postoperative period (Table 3). The majority were corrected by medications and electrolyte supplementation. One patient developed bradyarrhythmia requiring cardiologist consultation; a coronary angiography that was subsequently performed showed coronary stenosis. Eventually, the patient did not survive a MI (Table 3).
Classification of cardiac arrhythmia within 7 days.

AF: atrial fibrillation; CAG: coronary angiography; PVC: premature ventricular contraction; RVR: rapid ventricular response; SVT: supraventricular tachycardia.
Discussion
In this retrospective study, the incidence of MACEs after CEA was highest 3.5% (95% CI: 0.008–0.063) within 7 days of the CEA surgery, followed by 1.2% (95% CI: 0.004–0.028) within 7–30 days, and 1.8% (95% CI: 0.002–0.037) within 30 days–1 year. Cardiac arrhythmia was the most common form of MACE to occur within 7 days of the procedure. As to the secondary outcomes, the incidence of stroke was also highest, and it was only found within 7 days postoperatively (4.7%).
There are some possible biological bases to explain the high incidence of MACEs observed by this study. First, it is related to inflammation generated by a CEA. Several inflammatory cytokines—such as tumor necrosis factor and interleukin, and the generation of byproducts of lipid oxidation—have hostile effects on the integrity and function of endothelial cells. They eventually lead to a prothrombotic state, which could favor the risk of postoperative MACEs and stroke after a CEA.14,15 Second, a CEA can cause hemodynamic instability because the procedure site is in proximity to the carotid sinus, which has a baroreceptor function. A reduced carotid sinus sensitivity caused by a CEA is associated with increased sympathetic activity; in turn, this can lead to cardiovascular diseases, including coronary vasoconstriction, platelet aggregation, impaired ventricular remodeling, and life-threatening arrhythmia.16,17 Finally, the concomitant existence of CAD in the CEA population, as aforementioned, contributes to MACEs.
In contrast, a 2015 study by Vilariño-Rico et al. found that the MACE rates at 1, 3, and 5 years were 3.1%, 9.3%, and 15.8%, respectively. 18 Moreover, other research by Go et al. showed MACE incidences of 1.7%, 18.4%, and 31.5% at 30 days, 5 years, and 10 years, respectively. 9 The lower incidences of MACEs found by this study would most likely be the result of its relatively smaller number of CEA cases and shorter follow-up time.
Moving on to the factors associated with MACEs after a CEA, earlier studies9,18,19 reported that previous CAD, PAD, and contralateral carotid artery stenosis represented more than 50% of the significant risk factors. In the case of the current investigation, the authors were unable to identify any statistically significant factors associated with the MACEs arising within 7 days of the surgery.
PAD has been found to be a prognostic factor for adverse cardiovascular events after CEA. From the Reduction of Atherothrombosis for Continued Health (REACH) Registry, patients with PAD were found to have a MACE rate of 21.14% 1-year postoperatively. 20 This is substantially higher than the corresponding rate revealed by the present research (1.8%). The relatively low MACE rate might partly be explained by the incidence of PAD in the current work’s study population (10%) being considerably lower than the REACH Registry’s PAD rate of 25%–30%. 20
The Stenting and Angioplasty with Protection in Patients at High Risk for Endarterectomy (SAPPHIRE) trial 21 found that a history of MI and diabetes was a high-risk factor for major adverse cardiovascular and cerebrovascular events. Consequently, that research team recommended increased attention for patients with such a history. By contrast, the present investigation did not find any significant relationship between MACEs and either a history of MI or diabetes. The inclusion of higher-risk patients in the SAPPHIRE study may explain the discordant results.
Interestingly, the authors found that cardiac arrhythmia was the most common complication within 7 days postoperatively. However, 75% of those events were minor (such as paroxysmal ventricular contraction and supraventricular tachycardia), occurred temporarily, and could be controlled with medications and supportive treatments (see Table 3). The results from the current study may shed more light on this issue as we found that close postoperative monitoring of the CEA patients to rapidly detect cardiac arrhythmia and provide prompt treatment was vital.
Some studies have mentioned that antiplatelets and statins9,22,23 demonstrated a protective effect for cerebrovascular disease in that they reduced the incidence of cardiovascular events in stroke and TIA patients. Due to the high prescription rate of those medications in the population used in the present research, the authors were not able to observe any such relationship.
In anesthesia perspective, preoperative cardiac assessment can be very helpful to screen and guide anesthesiologists to prevent perioperative MACEs for these high-risk patients. However, to the current knowledge, there is no specific cardiac screening guideline for this group of patients. In the author’s center, pre-anesthetic clinic uses the 2014 ACC/AHA guideline on perioperative cardiovascular evaluation and management of patients undergoing noncardiac surgery as a guide for cardiac or specific consultation. Interestingly, if the incidence of MACEs after major vascular surgery is widely reported, specific guideline for this group of patients should be developed. Preoperative antiplatelet especially clopidogrel and aspirin in patients with recent symptomatic carotid stenosis is more effective than aspirin alone in reducing asymptomatic embolization, which is markers of future stroke and TIA risk. 24 Regarding the anesthesia technique, Vaniyapong et al. 25 reported no statistical difference in choices of anesthesia (local versus general anesthesia) and mortality rate in patients undergoing CEA. Although, specific intraoperative neurological monitoring in patient population is well established to help reduce adverse neurological outcome,26,27 cardiac monitoring shows no significant benefit in reduction of MACEs. In terms of arterial blood pressure management, phase of surgery is important. Concerning about “watershed” area during carotid cross-clamping, arterial pressure should be elevated to between normal values and 20% above baseline. 28 However, intraoperative hypertension can also make surgical shunt placement more difficult and has been associated with intracerebral hemorrhage. So, on balance, it is prudent to avoid hypotension, particularly during the period of carotid cross-clamping, whereas after restoration of flow, it is preferable to avoid hypertension to prevent hematoma, myocardial ischemia, and cerebral hyperperfusion. 29
The strength of this single-center study was that the CEA procedures utilized similar surgical and anesthetic techniques. Therefore, other confounders were scant. However, the research had some limitations. First, it is a retrospective study, some data might be missing and no control of some important factors, such as BIS or NIRS monitoring. However, with the low number of patients performing this operation, retrospective study design is inevitable. Second, compared to previous studies, this study has a smaller sample size and a relatively short follow-up period. To the investigator’s knowledge, this is still the largest retrospective cohort study of MACEs after CEA in a developing country. The result about the risk of cardiac arrhythmias within 7 days postoperatively should be useful and benefit for improving the perioperative care of CEA patients.
Conclusion
The incidence of MACEs after CEA was 3.5% within 7 days, with the most common complication being cardiac arrhythmia. Consequently, patients undergoing CEA represent a high-risk group, and the need for close postoperative monitoring is emphasized.
Unfortunately, the investigators could not identify any significant risk factors associated with the MACEs occurring in the population during the 7-day postoperative period. Future studies drawing on larger sample sizes and longer follow-up periods are needed.
Footnotes
Acknowledgements
The authors thank Chayanan Thanakiattiwibun from Integrated Perioperative Geriatric Excellent Research Center, Faculty of Medicine, Siriraj Hospital, Mahidol University, Bangkok, Thailand who assisted in the submission of the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from the Siriraj Institutional Review Board, Faculty of Medicine Siriraj Hospital, Mahidol University (Si 556/2018).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research project was supported by the Faculty of Medicine Siriraj Hospital, Mahidol University, Grant No. (IO) R016239001.
Informed consent
Written informed consent was not obtained due to the retrospective nature of the study.
Trial registration
This study was registered at clinicaltrials.gov (NCT03596229).
