Abstract
Objective:
The aim of this study was to evaluate survival in patients with COVID-19 and cancer, and to find factors associated with early mortality.
Methods:
Retrospective cohort derived from a registry of a referral center in Bogotá. Survival was analyzed according to the type of neoplasm using Kaplan–Meier method. A cox regression was performed to look for factors associated to higher risk of death.
Results:
Two hundred fifty-four patients were included with cancer and COVID-19, most of whom were women (median age 68 years; range 19–97). Cardiovascular comorbidities were frequent. Patients with hematologic neoplasms had higher survival than those with solid neoplasms (log-rank test, p = 0.024). C-reactive protein levels (hazard ratio 1.02; 95% confidence interval 1.00–1.03, p = 0.025), Charlson’s comorbidity index (hazard ratio 1.15; 95% confidence interval 1.06–1.26, p = 0.004) and respiratory failure (hazard ratio 4.83; 95% confidence interval 2.47–9.44, p = <0.001) were significantly associated with higher mortality. No interaction between active anticancer therapy and mortality was observed.
Conclusion:
In contrast to other reports, survival was worse in patients with solid tumors than in those with hematologic neoplasms. Increased C-reactive protein, Charlson’s comorbidity index and respiratory failure were associated with higher in-hospital mortality. This study reveals the complex impact of cancer and its treatment on COVID-19 outcomes, highlighting the persistent risks to cancer patients. It emphasizes monitoring C-reactive protein levels, comorbidities, and respiratory failure as key indicators of poor prognosis. Furthermore, we provide new insights into the differential impact of COVID-19 on cancer patients with solid organ versus hematologic neoplasms.
Introduction
The COVID-19 pandemic has had a profound economic and social impact worldwide. By the end of 2022, 636 million cases were reported, with nearly 6.61 million deaths. 1 Morbidity and mortality due to COVID-19 differs substantially depending on factors such as age and underlying comorbidities. 2 Since the dawn of the pandemic, it was recognized that cancer patients were disproportionately affected, with higher risk of severe COVID-19 (i.e., intensive care unit (ICU) admission and/or death) compared to the general population (39% versus 8%, respectively), 3 as well as delays in oncologic treatment and research. 4 However, this is a highly heterogeneous population, and the risk seems to be determined by other associated factors as well.1,2,4,5
For instance, hematologic malignancies have been associated with risk of death of 33%–37%6,7 as well as higher ICU admission (26% versus 19%) and higher need for ventilatory support (11% versus 10%) when compared to solid malignancies. 6 A British cohort (n = 1044) was one of the earliest studies to find an association between specific cancer types and outcomes in COVID-19; they reported that leukemia was associated with higher mortality as compared with other cancers (odds ratio 2.25, 95% confidence interval (CI) 1.13–4.57). 8
Multiple studies describe that the factors that predict mortality due to COVID-19 in the general population are also valid for cancer patients, such as age, male sex, cardiovascular disease (CVD), and hypertension.5,9 Likewise, there is evidence of a higher mortality risk in patients with worse functionality, higher rate of comorbidities, 9 hematological neoplasms compared to solid tumors,8,10–12 lung cancer, 13 metastatic solid tumors, 10 and low socioeconomic status. 14
Some studies have reported that active treatment with cytotoxic chemotherapy increases the risk of worse outcomes and mortality.9,10,15,16 However, other studies did not find the same association between receipt of cytotoxic chemotherapy and mortality,17,18 while other found a detrimental effect of chemotherapy, but no influence of other treatments such as immunotherapy, radiotherapy, or surgery. 19
Prognosis and acute treatment of patients with cancer was also complicated by the scarcity hospitalization and ICU resources during the worst periods of the pandemic; the highest occupancy of the ICU during the pandemic in Colombia was in June 2021 with 88%, although areas such as Bogota, Antioquia, and the Eje Cafetero reached figures between 90% and 95%. 20
This study aimed to evaluate mortality in patients with cancer and SARS-COV-2 infection and find possible associations with this outcome, such as type of oncologic treatment, type of malignancy, receipt of active oncologic treatment, functionality, comorbidities, among others.
Methods
We conducted a retrospective cohort study of patients older than 18 years with a confirmed diagnosis of cancer included in the COVID registry of Hospital Universitario San Ignacio in Bogotá, Colombia. Patients were admitted to the Emergency, Internal Medicine, and ICU departments between March 2020 and February 2021. Confirmation of SARS-COV-2 infection by polymerase chain reaction was required. Patients who were discharged or referred to other institutions within the first 48 h after admission were excluded. A total of 4553 patients with confirmed COVID-19 were available in the institutional COVID-19 registry and were screened for inclusion and exclusion criteria.
The study was approved by Comité de Investigaciones y Ética institucional (CIEI) of Pontificia Universidad Javeriana–Hospital Universitario San Ignacio (approval number 2021/155). An informed consent waiver was granted considering that data were retrospectively collected with no direct intervention to patients, and precautions were taken to preserve confidentiality, in accordance with Colombian law and international regulations.
The information was obtained from the institutional COVID-19 registry of the Hospital Universitario San Ignacio, where all patients treated with this diagnosis were registered in a standardized and systematic manner since the beginning of the pandemic. Sociodemographic variables, comorbidities, laboratories at admission, treatment for COVID-19, and outcomes were recorded in Research Electronic Data Capture (REDCAP®) software by trained medical and nursing staff. The quality of data was checked on a recurrent basis to minimize the rate of missing data and increase fidelity. For the purpose of this study, additional information had to be gathered from electronic health records, including indication for intensive therapy, intensive care therapy allocation according to resource availability, tumor type and stage, data on oncologic treatment, chronology of oncologic therapy, and Eastern Cooperative Oncology Group (ECOG) performance scale. 21 Quick sepsis-related organ failure assessment score 22 at admission, and Charlson’s comorbidity index 23 were calculated for every patient.
Statistical analysis
At the time of planning the study, there was scarcity of data regarding the expected differences in outcomes of COVID-19 infection for patients with solid and hematologic tumors. However, sample size was calculated to detect a hazard ratio (HR) of 0.65 in log rank test for overall survival function of solid versus hematologic patients, with a power of 80%, alpha error of 0.05, assuming a proportion of 3:1 in the number of solid versus hematologic tumors. Estimated sample size was 187. Descriptive statistics were used to present the sociodemographic data and clinical variables. For continuous variables, mean and standard deviation or median and interquartile range were used, depending on the distribution of the data. Shapiro–Wilk test was used to evaluate the assumption of normality. For categorical variables, frequency and percentage were reported. The difference between groups (hematologic versus solid cancer; and patients with active neoplasms and active treatment versus no active treatment) was evaluated according to the nature of the variables using Student’s t-tests, chi-square, or Mann–Whitney U tests. Active treatment was defined as receipt of systemic therapy, radiotherapy, and/or oncologic surgery within 8 weeks prior to admission.
Survival was analyzed using Kaplan–Meier curves and Log Rank test for comparison between groups. Multivariate Cox regression was performed to evaluate whether variables such as age, comorbidities, hospital stay, ICU treatment requirement, and cancer features were associated with higher mortality. Differences were considered statistically significant if p < 0.05. The cox proportional hazards assumption was evaluated with Schoenfeld residuals analysis and confirmed with the graphical analysis of log(−log(−log(S(t))) versus t or log(t). All tests were performed using Stata 12 StataCorp LP (College Station, TX, USA) and graphics were generated with RStudio.
Results
A total of 254 patients with cancer and COVID-19 met inclusion criteria and were included. Median follow-up was 8 days (range 2–112 days). The demographic and clinical characteristics according to the type of cancer are shown in Table 1. Most patients were female (52%), median age was 68 years (range 19–97).
Clinical characteristics and outcomes in patients with cancer and COVID-19 infection according to type of neoplasm.

IQR: interquartile range; HT: hypertension; CVD: cardiovascular disease; CKD: chronic kidney disease; CLD: chronic lung disease; ECOG: Eastern Cooperative Oncology Group; ARDS: acute respiratory distress syndrome; PE: pulmonary thromboembolism; DVT: deep vein thrombosis; CNS: central nervous system; ICU: intensive care unit; IMV: invasive mechanical ventilation; AKI: acute kidney injury; KDIGO: Kidney Disease Improving Global Outcome severity scale for AKI. Bold values are statistically significant.
p (Student’s t-tests, chi-square, or Mann–Whitney U tests).
Patients with solid tumors comprised 73.2% (n = 186) of the cohort, of which 46.2% had metastatic involvement, 38.7% had active disease under treatment, 26.9% had active disease without treatment, and 29% were in remission without treatment. The most frequent primary for this group was prostate (n = 29 (15.6%)), followed by cervix uteri (n = 27 (14.5%)), breast (n = 24 (12.9%)), thyroid and nonmelanoma skin cancer (n = 20 (10.8%)), colon and rectum (n = 13 (6.9%)), lung (n = 10 (5.5%)), stomach (n = 9 (4.8%)), and others (n = 54 [29.0%]).
Patients with hematologic malignancies represented 26.8% (n = 68), of which 61.8% had active disease under treatment, 20.5% had active disease without treatment, and 17.6% were in remission without treatment. The most frequent type of hematologic malignancy was non-Hodgkin lymphoma (n = 24 (35.3%)), followed by acute leukemia (n = 13 (19.1%)), multiple myeloma (n = 13 (19.1%)), myelodysplastic syndromes (n = 8 (11.8%)), chronic leukemias and myeloproliferative neoplasms (n = 7 (10.3%)), and Hodgkin lymphoma (n = 3 (4.4%)).
In terms of performance status, 51.6% of patients with solid organ neoplasia and 36.8% of patients with hematologic neoplasms had ECOG of 0–2. Data on body mass index was available for 20% of patients of which 10.0%, 40.0%, 27.6%, and 21.3% were underweight, within normal range, overweight, and obese, respectively. The most frequent comorbidities were arterial hypertension (n = 114 (44.9%)), CVD (n = 55 (21.7%)) including coronary heart disease, vascular disease, heart failure of any cause, cerebrovascular disease; diabetes mellitus (n = 41 (16.1%)), and chronic lung disease (CLD, n = 30 (11.8%)) including asthma, chronic obstructive pulmonary disease, and interstitial lung disease.
Regarding the allocation of resources in the context of the COVID-19 pandemic, 12.9% and 30.8% of patients with solid and hematologic neoplasms, respectively, were admitted to the ICU. However, 34.3% of patients had reorientation of therapeutic effort because of their disease stage, performance status, and oncologic prognosis, and thus did not receive intensive care despite being critically ill. This was more frequent in solid than hematologic cancer patients (40.3% versus 17.3%, p > 0.001, Table 1). Admission to the ICU was also limited by scarcity of ICU resources in 2.8% of cases. In patients admitted to the ICU, those with hematologic neoplasms had a greater requirement for invasive mechanical ventilation (IMV), vasopressors, and antibiotics, compared to those with solid tumors (Table 1).
Most complications during hospital stay were similar between the two groups, except for bacterial infection/bacteremia which was significantly higher in patients with hematologic malignancy (36.8% versus 22.6%, p = 0.023). The most common complications were acute respiratory distress syndrome, venous thromboembolic disease, and acute kidney injury Kidney Disease Improving Global Outcome scale 24 3 (KDIGO 3) for both groups.
Table 2 shows clinical and laboratory characteristics measured at admission as part of the severity assessment recommended by the Colombian Consensus for the management of SARS-COV-2 infection at that time 25 stratified by status of oncologic treatment (active treatment versus no active treatment).
Characteristics of cancer patients according to whether they received active oncological treatment prior to COVID 19 diagnosis.

IQR: interquartile range; CRP: C-reactive protein; LDH: lactate dehydrogenase; QSOFA: Quick Sequential Organ Failure Assessment. Bold values are statistically significant.
Active treatment was defined as chemotherapy, immunotherapy, radiotherapy, target therapy, and/or surgery within 8 weeks prior to admission.
p Value (Student’s t-tests, chi-square, or Mann-Whitney U tests), differences were considered statistically significant if p < 0.05.
Thirty-four (18.3%) patients with solid tumors on active treatment received chemotherapy or chemotherapy plus immunotherapy and 38 (20.4%) received other treatments such as immunotherapy alone, radiotherapy, hormonal therapy, targeted therapy, or surgery. Twenty-six (38.2%) hematologic patients on active treatment were receiving chemotherapy, chemotherapy plus proteasome inhibitors, or proteasome inhibitors alone, 5 (7.3%) had received radiotherapy, and 2 (2.9%) other targeted therapies. No significant differences were found in baseline characteristics or outcomes, except for hospital stay, which was longer in patients with active treatment (median 11 days versus 8 days, p = 0.047, Table 3).
Univariate and multivariate analysis of factors associated with mortality.

CVD: cardiovascular disease; CRP: C-reactive protein on admission; LDH: lactate dehydrogenase; HR: hazard ratio; ECOG: Eastern Cooperative Oncology Group.
p Value (Log Rank test).
p Value (Cox regression model) differences were considered statistically significant if p < 0.05.
For each unit increase.
Patients with hematologic neoplasms had higher overall survival than patients with solid tumors (23 days; 95% CI 16–39 days versus 17 days; 95% CI 14–22; log rank, p = 0.024) (Figure 1(a)). Survival was also analyzed excluding less aggressive tumors, such as chronic leukemias, myeloproliferative neoplasms, differentiated thyroid cancer, and nonmelanoma skin cancer, and this difference was maintained (Figure 1B).
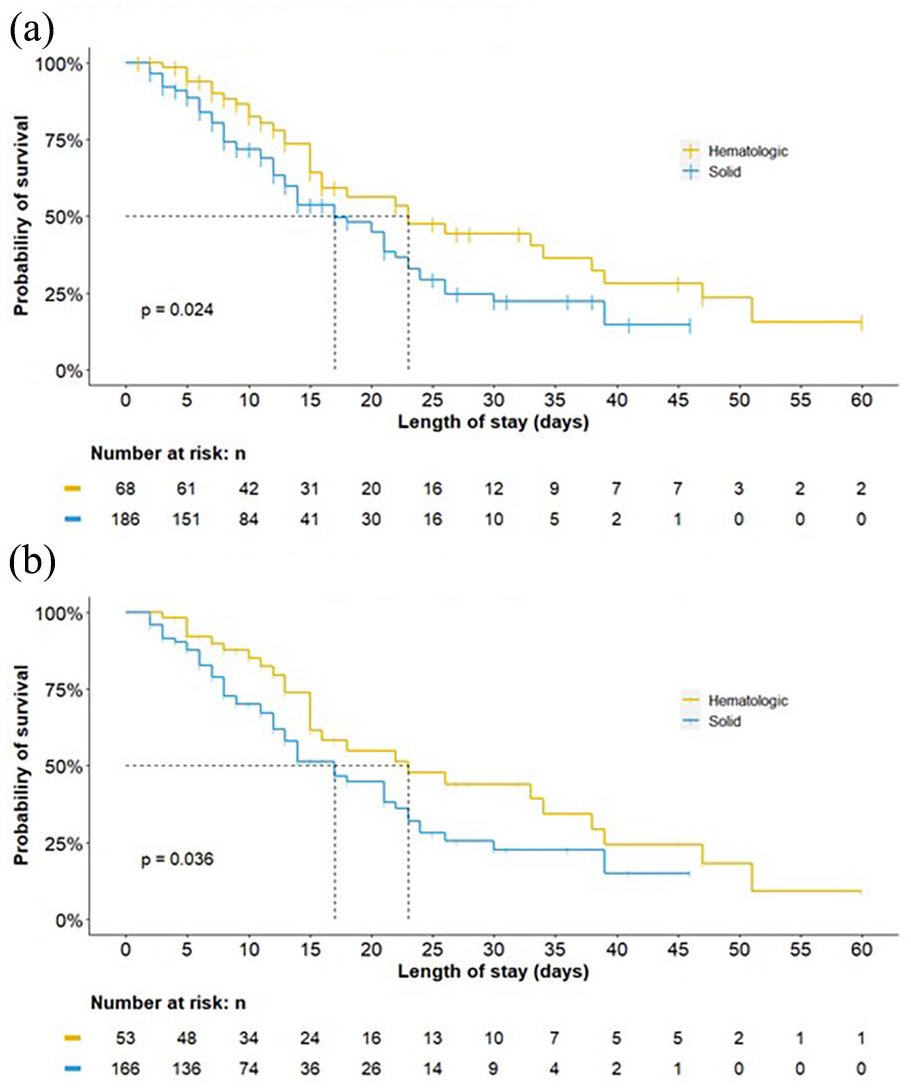
(a) Survival by type of neoplasm. (b) Survival includying only aggressive neoplasms.
In patients with solid tumors not in remission, there was no survival difference between patients with active oncologic treatment compared to patients without active treatment (p = 0.99, Figure 2(a)). Patients with solid tumors and metastatic disease showed a tendency toward inferior survival. However, this difference was not statistically significant (p = 0.081, Figure 2(b)). Conversely, among patients with active hematologic malignancy, those with active treatment had higher overall survival than those without active treatment (median overall survival 33 days; 95% CI 23 to not estimable (NE) versus 15 days; 95% CI 10 to NE; p < 0.01; Figure 2(c)).
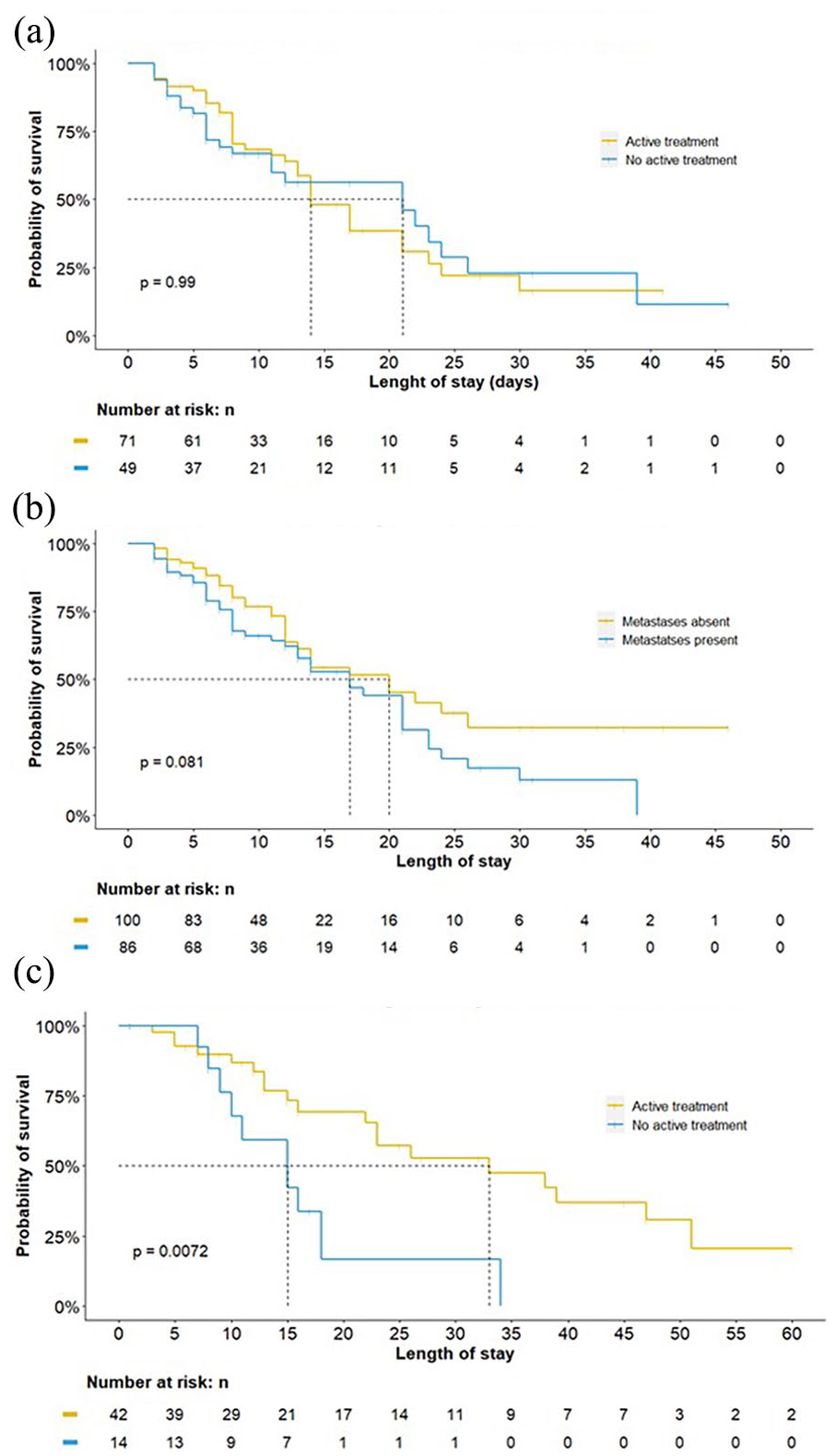
Survival for solid and hematological neoplasms according to status. (a) Survival in solid neoplasms not in remission. (b) Survival in solid neoplasms not in remission. (c) Survival in hematologic neoplasms not in remission.
The univariate analysis (Table 3) showed that older age, having a solid neoplasm, active untreated hematologic neoplasm, diabetes mellitus, CVD, CLD, C-reactive protein (CRP), lactate dehydrogenase, elevated D-dimer, ECOG scale 3–4, respiratory failure on admission, and a higher Charlson’s comorbidity index were associated with higher mortality. The data met the Shoenfeld’s proportional hazards assumption (p = 0.445). In multivariate analysis, only the Charlson’s comorbidity index (HR 1.15; 95% CI 1.06–1.26; p = 0.001), increased CRP (HR 1.02; 95% CI 1.00–1.03; p = 0.036), and respiratory failure (HR 5.38; 95% CI 2.79–10.37; p < 0.001) remained significantly associated with higher mortality.
Discussion
COVID-19 disease continues to be a factor that correlates with worse outcomes in cancer patients for multiple reasons. Contrary to what has been reported in other studies,3,6,8,26 this cohort showed worse survival in patients with solid organ neoplasms compared to hematologic neoplasms. We also observed longer in-hospital stay, but not worse survival in those who were receiving active anticancer treatment (< 8 weeks) compared to patients without active treatment.
Possible explanations for these discrepancies with previous literature may be related to a lower admission rate to the ICU (12.9% versus 30.8%), lower delivery of mechanical ventilation (9.7% versus 29.4%), and vasopressors (8.1% versus 29.4%) in patients with solid neoplasms with respect to those with hematologic neoplasms. Almost half of the patients with solid neoplasms had metastatic involvement (46.2%), which may have influenced decision-making on ICU resources allocation in the setting of scarcity of resources caused by the pandemic. Additionally, we found that patients with active treatment for hematologic malignancies had better survival than those with active neoplasms and no treatment. Furthermore, the proportion of patients with active treatment was higher for hematologic patients compared with solid tumor patients. Patients with active neoplasm under active treatment may have been more fit and with better oncologic prognosis than patients with active neoplasms without treatment. Therefore, the higher proportion of those patients in the hematologic group may have contributed in part to the observed difference in survival. However, in multivariate analysis, neither having a solid neoplasm nor being in active treatment were significantly associated with survival. All the above suggests that the observed higher mortality in solid tumor patients might be better explained by older age, worse ECOG scores, and less ICU resources allocation.
Data on the implications of anticancer therapies in outcomes of patients with COVID-19 have been contradictory. In theory, cytotoxic chemotherapy, radiotherapy, and surgery may suppress the host immune response against COVID-19, whereas immunotherapy may paradoxically exacerbate immune-mediated end-organ damage by increasing inflammatory response. 27 The multicenter on Covid study evidenced that receipt of chemotherapy, targeted therapy, or immunotherapy did not worsen mortality in European patients with cancer. 17 Brar et al. found similar results. 28 Conversely, Yekedüz et al. reported that use of cytotoxic chemotherapy, but not other therapies (i.e., targeted therapy, surgery, immunotherapy or radiotherapy) was associated with higher mortality. 19 Chavez-MacGregor et. al also reported that patients with recent anticancer treatment fared worse than patients with cancer and no recent anticancer treatment. 10 In our study, we found no significant differences in baseline severity indices or risk factors between patients who were under active anticancer treatment versus those who were not. The only significant difference was a longer hospital stay for the former. However, mortality was similar and high for both groups. This is in agreement with aforementioned research from Pinato et al. 17 and Brar et al. 28 The discrepancies with other research may be explained by the heterogeneity of included patients in all studies (e.g., patients with immunotherapy mixed with patients with targeted therapy and radiotherapy), since different treatments may have different impacts on immunity. All this suggests that further studies on the topic need to be designed to analyze a more homogeneous group of patients.
Various studies in the general population and in some cancer cohorts,2,5,9,18,29,30 have reported that age and cardiovascular comorbidity are important risk factors for unfavorable outcomes and mortality in COVID-19. In contrast, and even though cardiovascular comorbidities were frequent in our study, we found that these factors were associated with mortality in univariate analysis but not in multivariate analysis. Diabetes, ECOG ⩾3, increased CRP, and respiratory failure were also associated with higher mortality in univariate analysis, but only elevated CRP greater than 10, respiratory failure and higher Charlson’s index remained significantly associated in multivariate analysis. It has also been recognized that both active and previous use of tobacco is a negative prognostic factor in patients with cancer and COVID-19. 31 The prevalence of smoking in our study was low and we did not find an association with outcomes.
There has been discussion regarding possible markers of disease severity, and CRP has been related to adverse outcomes along with other markers in different cohorts of cancer patients.12,29,32,33 Our study suggests that CRP holds its prognostic value in this set of patients, with higher values progressively associated with worse outcomes.
The overall mortality found in this population is similar to that documented in other studies.34–36 However, it is expected to decrease following advances in knowledge of the disease, vaccination, and increased availability of resources for the care of patients with severe COVID-19 infection and cancer.37,38 Vaccination in Colombia began on February 20, 2021, 39 and the patients in our study had a low probability of being vaccinated. Several studies have shown that cancer is a risk factor for mortality due to COVID-19 40,41 and that vaccination is safe and effective. 42 However, studies of vaccination for Covid-19 in cancer patients suggest a lower rate of seroconversion and a lower intensity of immune response, 43 which may require more frequent boosters. 44 Therefore, although it is currently expected that vaccinated cancer patients with COVID-19 infection will have lower mortality, studies are needed to evaluate the best vaccination strategy in this population.
An important advantage of our study is the high proportion of patients with hematological malignancies, in contrast to other studies, which has led to the need for meta-analyses. 5 Another strength is that data were gathered in the context of a medical registry, which increases accuracy and fidelity.
The main limitation of our study is its retrospective nature and single institution setting. Furthermore, some of the observed differences were not statistically significant, probably due to sample size, which was limited to a single center. Heterogeneity in the type of neoplasms, comorbidities, and disease stages may have also contributed to these results. However, the use of multivariate analysis takes into account this heterogeneity, which increases validity of the study and allowed to find significant associations with mortality. Additionally, the associations found in this study are consistent throughout multiple reports in the literature5,35 with biologic plausibility. However, it is necessary to expand these analyzes in larger populations, representative of various oncological pathologies and stages of the disease, ideally in a multicentric setting.
Conclusion
COVID-19 disease continues to be a factor that correlates with worse outcomes in cancer patients. Our findings contrast with some literature in that patients with solid tumors fared worse than patients with hematologic tumors. Additionally, we found no association between recent anticancer treatment and higher mortality and found that CRP levels, Charlson Comorbidity Index and respiratory failure at admission due to COVID-19 infection were associated with higher in-hospital mortality in cancer patients. Further studies evaluating the impact of vaccination and antiviral therapy on these outcomes are needed.
Footnotes
Acknowledgements
We would like to express our gratitude to all the personnel of the research office and department of internal medicine for the administration of the COVID-19 registry database.
Author contributions
Sandra Brigitte Amado-Garzón contributed to supervision and leadership, study conceptualization and design, data collection and analysis, drafting of the manuscript. Luisana Molina-Pimienta and Juan Manuel Vásquez-Jiménez contributed to study conceptualization and design, data collection and analysis, drafting of the manuscript. Karen Lizeth Álvarez-Raigoza, Mauricio Manrique-Samer, Carlos E. Lombo-Moreno contributed to data collection and analysis, drafting of the manuscript. Alejandra Cañas-Arboleda contributed to study conceptualization and design, verification of data analysis, proofreading of the manuscript.
Data availability statement
Data is available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received no external funding or grants of any kind for the conduction of this study. The study was entirely funded by Hospital Universitario San Ignacio and Pontificia Universidad Javeriana.
Ethical considerations
The study was approved by Comité de Investigaciones y Ética institucional (CIEI) of Pontificia Universidad Javeriana–Hospital Universitario San Ignacio (approval number 2021/155).
Informed consent
Informed consent waiver was granted by CIEI, since this research was considered “without risk (sin riesgo)” according to Colombian law (Artículo 11. Resolución número 8430 de 1993–Ministerio de Salud).
Consent to participate
An informed consent waiver was granted considering that data was retrospectively collected with no direct intervention to patients, and precautions were taken to preserve confidentiality of data, in accordance with Colombian law and international regulations.
Consent for publication
Not applicable.
Trial registration
Not applicable.
