Abstract
Objectives:
The spread of severe acute respiratory syndrome coronavirus 2 in Ethiopia is below par understood and to date has been poorly characterized by a lower number of confirmed cases and deaths to other regions of the sub-Sahara African including Ethiopia. Timely and effective predictors for inpatient mortality rate were crucial for improving the management of hospitalized cases. This study aimed to assessed predictors for inpatient mortality of COVID-19 hospitalized adult patients in two diagnosed and treatment centers, North West Ethiopia.
Methods:
A facility-based retrospective cohort study was conducted among COVID-19 adult admitted cases in two treatment centers, Northwest Ethiopia, from 1 October 2020 to 30 December 2020. Data from the records of children were extracted using a standardized checklist. Epi-Data version 3.2 was used for data entry, and Stata version 14 was used for analysis. Bi-variable and multivariable Cox regression analyses were conducted to identify predictors of mortality. Finally, variables with P < 0.05 were a significant predictor of inpatient mortality.
Result:
The mean (±standard deviation) age of participant cases was 48.6 (±18.8) years. The median (±interquartile range) time for death reported after was 13 (±6) days. The overall incidence rate inpatient mortality rate was determined as 1.8 (95% confidence interval: 1.72, 2.15) per100 person per days of observation. Cases at baseline age ⩾ 61 years (adjusted hazard ratio = 1.56; 95% confidence interval: 1.3, 2.4), being male gender (adjusted hazard ratio = 1.9; 95% CI: 2.1, 8.6), admission with comorbidity (adjusted hazard ratio: 4.4, 95% confidence interval: 2.3, 8.4), and decreased neutrophil count ⩽ 65 103/uL at (P < 0.03) were independent predictors for inpatient mortality.
Conclusion:
In general, 72.4% of COVID-19 inpatient deaths were occurred within 2 weeks after admission. The mortality risk factors for severe patients identified in this study using a multivariate Cox regression model included elderly age (⩾60 years), being male, baseline comorbidity, and neutrophil count ⩽65 103/uL were associated with inpatient mortality.
Introduction
The coronavirus disease 2019 (COVID-19) pandemic, caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2),1,2 poses huge health and societal burden globally. SARS-CoV-2 is easily transmitted from person to person 3 via droplets from the respiratory tract of infected people, including asymptomatic individuals. 4 The spectrum of the COVID-19 severity varies broadly, from asymptomatic infection to sever complication like organ failure to death. 5 A spectrum of presentation has been reported, ranging from asymptomatic infection to severe lower respiratory tract illness presenting with fever, cough, chest pain, anosmia, and progress to acute respiratory distress syndrome (ARDS) for final death outcome.6,7 There is unprecedented urgency to understand who is at most risk of severe outcome, and timely analysis were needed to identification risk individuals. 8 The precise mortality rate of the most severe forms of SARS-CoV-2 infections that are admitted to the intensive care unit (ICU) varies among studies, ranging from 8.1% to 30% for hospitalized for SARS-CoV-2 pneumonia, and up to 16% to 78% for critical (ICU) care patients.6,9–11 Within 1 year, the SARS-CoV-2 pandemic has affected more than 125 million people worldwide. During the first wave in spring 2020, hospitalization rates were high, reaching up to 70% in France, 55% in Spain, 50% in the United Kingdom, and 20% in Germany until the end of April.12,13
Most of the published studies conducted on the SARS-CoV-2 pneumonia are well narrated in their distribution at China and Europe countries;14,15 however, new species derivation, variation, and magnitude was not well stipulated, so far in Ethiopia.16,17 Clinicians should to a deep need for mortality predictors that allow to quickly triaging patients with severe coronavirus disease 2019 (COVID-19). 18 Age and gender are well-established risk factors for severe treatment outcome and 90% associated of inpatient-related mortality.8,16 Laboratory markers including levels of C-reactive protein, ferritin, D-dimer, fibrinogen, and (PaO2/FiO2, PaCO2 mmHg)19–21 are attributed for effectual treatment outcome.5,10,20 Early identification of mortality-related factors for critical cases is crucial on clinical support and formulating preventive measures to optimizing treatment options. 17 Therefore, this study was aimed to assess predictors for inpatient mortality of COVID-19 hospitalized adult patients in two diagnosed and treatment centers, North West Ethiopia.
Methods
Study design and setting
A facility-based retrospective cohort study was conducted among 288 admitted cases in two COVID-19 diagnosed, and treatment centers from 1 October to 30 December 2020. These two centers are the first hospitals designed to manage positive adult cases of SARS-CoV-2 infections in Benishangul Gumuz regions, North-Western Ethiopia. 22 The diagnosis was confirmed by polymerase chain reaction (PCR). Individually, these two centers had a capacity of admitting more than 250 cases per month.22,23 According to the 2019 national population projection this region had an estimated 1.7 million population were survived, and the area is more prominent for infection transmission due to center for national project (site for GERD) and trade corridor with south Sudan. 24
Study population
The study participants were all COVID-19 adults ⩾18 years admitted in Assosa and Pawe general hospitals from 1 October to 30 December 2020 were included. Children aged <18 years were excluded, where the Ethiopia national COVID-19 inpatient’s treatment criteria for children <18 years were different set up and setting. 1 The clinical follow-up was immediately initiated by taking of chest computed tomography scan compatible with confirmed diagnosis of COVID-19 pneumonia followed by routine blood workup (including complete blood count, inflammatory markers, liver, renal function test, and arterial blood gas analysis) were required before hospitalization of a given cases for admission. 25
Sample size determination
We determined the minimum sample size for this study using survival analysis formula including the following assumption in STATA(SE) version-14 by considering, -sided significance level (α = 5%), Za1/2 =

HR = hazard ratio (AHR) 0.56 from Tolossa et al. 16 By inserting all parameters into the STATA (SE)\14 gives 288. Therefore, the final sample size was calculated and selected as to be 288, and therefore, we included all admitted cases aged > 18 years without sampling procedure from 1 October to 30 December 2020.
Outcome ascertainment
The primary outcome was in-hospital mortality of adult patients at any phases of treatment and declared by physicians as ceased. Cured/discharge was analyzed as a censored event in the Cox regression of this survival analysis. The Cox regression model proposed by Moore D and Dirk F was applied in 2015. 26
Independent variables
We identified independent variable in three categories (1) demographic data for all patients, including age, sex, number of family, and resident; (2) clinical variables also included comorbidities, smoking status, hospital admission time, presence of fever (defined as axillary temperature of at least 37.5°C), dyspnea, cough, diarrhea, and all hematological profiles of patients were selected as independent variables.1,27
Laboratory and treatment procedures
Laboratory confirmation of the SARS COV-2 infection was defined as positive real-time reverse transcriptase polymerase chain reaction (RT-PCR) from nasal and pharyngeal swab; samples were prospectively collected and analyzed at the Molecular Virology Units of each center according to the WHO guidelines. 16 Treatments included the use of antiviral therapy (lopinavir/ritonavir or darunavir/ritonavir), hydroxychloroquine, enoxaparin, and immunomodulatory/immunosuppressive therapy such as corticosteroids, tocilizumab, and sarilumab. Lopinavir/ritonavir 400/100 mg was administered orally twice daily for 14 days, while darunavir/ritonavir 800/100 mg was administered once daily for 14 days. Corticosteroid treatment consisted in dexamethasone 20 mg daily for 5 days or methylprednisolone 1 mg/kg intravenously daily for 5 days. Oxygen support consisted of low (cannula and simple masks) and high flow (Venturi and reservoir masks, Nasal High Flow), helmet continuous positive airway pressure (CPAP) or noninvasive ventilation (NIV). The choice for the oxygen support was determined by rapid deterioration of P/F ratio and upgraded if a further worsening after 2 h of treatment was detected.1,7
Data collection instruments and quality assurance
Data abstraction tool was prepared from COVID-19 individual input log sheet federal ministry of health of COVID-19 clinical management book 1 with previously published articles in Ethiopia.16,17 The data abstraction tool was prepared from COVID-19 individual input log sheet federal ministry of health of COVID-19 clinical management book with previously published articles in Ethiopia. However, the checklists was pilot tested before data collection among 15 COVID-19 admitted patients in Jawi Hospitals, and then we incorporated on the checklists economic status, and number of family in the house incorporated for final data collections. Two diploma nurses and two BSc nurses were recruited for data collection & supervision. One day of training was given for data collection and supervision. Before the actual data collection, the prepared checklist was pretested on 5% 14 of the original sample size files were tested in Jawi COVID-19 treatment centered. Variables of COVID-19 contact history and levels of cholesterol were included and incorporated after the pilot tested.
Statistical analyses
EPI-DATA version 3.2 and STATA/14 software were used for data entry and analysis, respectively. The Pearson’s chi-square test was used to compare categorical variables. A variable with P value < 0.1 at bi-variable Cox regression analysis were candidate transferee for multivariable Cox proportional model. Multivariable Cox regression model with 95% confidence interval (CI) and adjusted hazard ratio (AHR) was used to identify a significant predictor of inpatient mortality at P value < 0.05. The overall model adequacy of the proportional hazard model was assessed using a cox-Snell residual graph.
Result
Socio-demographic and clinical profiles of adult COVID-19 patients
From 1 October 2020 through to 30 December 2020, totally 289 cases admitted in two general and referal hospitals. Majority of, 207 (71.8%) vs 81 (28.2%) of participants were from Assosa and Pawe hospitals, respectively. More than half-165 (54.17%) vs 123 (45.8%) of cases was male and females in gender. The mean (±SD) age of the patient was 49.5 (±18.88) years. Briefly, among total cases, majority 147 (51.5%) of patients age were ⩾61 years, and 165 (57.5%) of them were urban dwellers. Moreover, 38 (65.5%) died cases were hospitalized for ⩾10 days. The most commonly self-reported symptoms at onset of illness were fever (n = 232 (80.5%) shortness of breathing (n = 227 (78.8%) Myalgia (n = 177 (61.46%) Vomiting (n = 97 (33.68%) cough (n = 171, (59.4%) and global headache (n = 175 (60.8%)). Of the total 288 admitted cases, 162 (56.3%) had comorbidity, the commonest are (CLD = 44, HTN = 49, DM = 24, HIV = 20), whereas the remaining cases were TB and other renal complications. Out of 288 radiology result, 10/288 (3.5%) and 23/288 (7.9%) had unilateral and multilateral opacity of lunge, respectively. More than half (62.1%) and 37 (63.7%) of admitted cases had smoking and index cases contact history, respectively (Table 1).
Socio-demographic and clinical findings of COVID-19 admitted patients in two treatment centers North West Ethiopia 2021.
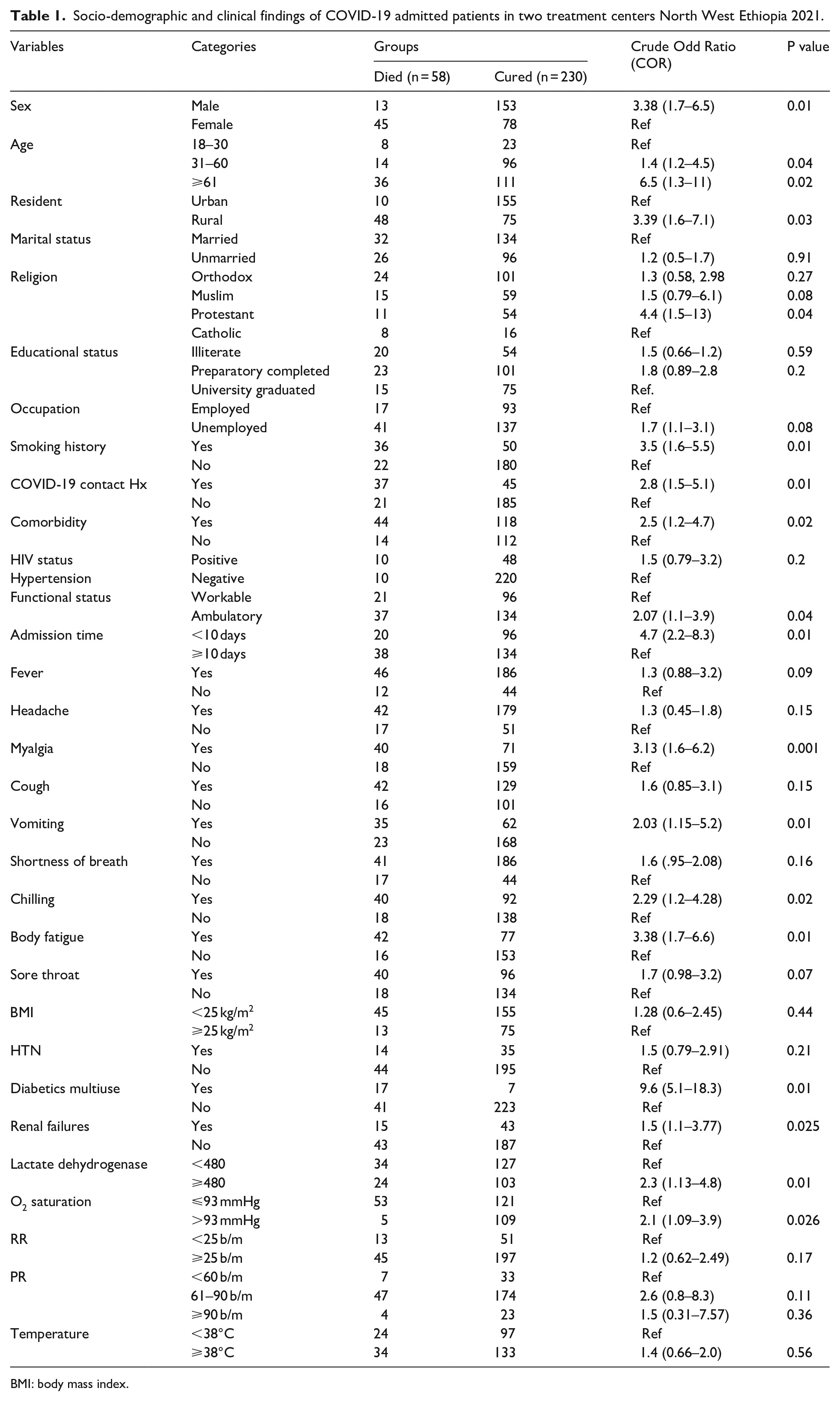
BMI: body mass index.
The laboratory features at admission are presented in (Table 2), and the inflammatory measures were a little bit high. Moreover, the absolute decrement of neutrophil count ⩽ 65 103/uL for cured cases (mean ± SD; 54.83 ± 10.67) versus died cases (mean ± SD: 0.96 ± 0.38) had a significant risk of variation for mortality exactly at (P < 0.03) of the two groups (Table 2).
Baseline laboratory indicators COVID-19 hospitalized adult patients in two general hospitals in North West Ethiopia 2021.

SD: standard deviation, mg: milligram, PO2: partial oxgen saturation in the blood, mg/dl: milligram per decilitres.
Mortality Rate and hazard risk
This retrospective cohort contributed a total follow-up time of 3127 persons per day of risk observation. Of the total 288 cases, 58 (20.2%) were died, whereas the remaining 230 (78. 8%) cases were cured. The number of deaths within the first 5, 10, 15, 20, and above 25 days of risk observation were found 10 (17.2%), 39.6%, 18.6%, 13.8%, 6.1%, and 3.4% respectively. The median (±interquartile range (IQR)) time for death reported after admissions were found 13 (±6) days with a cumulative survival probability of 82.6%; 95% CI (77.1: 86.8). The overall incidence density rate (IDR) of inpatient mortality was found 1.8 (95% CI; 1.72, 2.15) per100 person-days of observation.
Predictors for inpatient mortality
In the final model, the following three factors (i.e. sex, age, and comorbidity of patients) were significantly associated with COVID-19 inpatients mortality. We observed that the hazard of death among admitted COVID-19 patients presenting with age ⩾61 years for were nearly 1.5 (AHR = 1.56; 95% CI: 1.3, 2.4, P < 0.03) times increased as compared with age <30 years. Likewise, death hazards for patients having previous medical comorbidity were 4.1 (AHR = 4.4; 95% CI: 2.3, 8.4, P < 0.02) times higher than those without any comorbidity. Moreover, the hazard of death for males admitted COVID-19 patients was 1.9 (AHR = 1.9; 95% CI: 2.1; 8.6, P < 0.001) times higher than females admitted COVID-19 participants.
Discussion
Our study illustrates significant associations of overall adult inpatient COVID-19 mortality rate with clinical, laboratory, and demographic factors. The results demonstrated that advanced age > 61 years, male gender, baseline medical comorbidity, and absolute decrement in neutrophil count ⩽ 65 103/uL among admitted COVID-19 patients are disproportionately prevalent for expired (ceased) groups. Moreover, at the end of the follow-up, 58 (20.2%) participants have died, and whereas the remaining 230 (78.8%) were cured within 2671 persons per day risk of observation. Regarding to patients presenting symptoms; fever was the most commonly self-reported chief complained of illness (n = 232, 80.5%), while dyspnea (n = 227, 78.8%), myalgia (n = 177, 61.46%), cough (n = 171, 59.4%), headache (n = 175, 60.8%) were also frequent presenting symptoms for admitted cases. Furthermore, shortness of breathing (41/58 vs 17/58) and vomiting (35/58 vs 23/58) had been reported considerably more in died or expired group than in cured cases. Moreover, the overall IDR of inpatient mortality was found 1.8 (95% CI; 1.72, 2.15) per100 person-days of observation. Likewise, the absolute decrement of neutrophil count ⩽ 65 103/uL (mean ± SD: (0.96 ± 0.38)) had a significant variation for risk of mortality as compared with declared cure cases (mean ± SD; (54.83 ± 10.67) exactly at (P < 0.03) of the two groups. This is consistent with finding in Mexico, 9 provinces in Shandong China, 27 Beijing china, 28 and Wuhan china. 29 This might be associated with having neutrophils had powers of hyperactive for inflammatory initiation of epithelial tissue and demoted the viral progression. The percentage of (62%) of older age ⩾ 61 years inpatients death was much higher than death group on age < 30 years (8/58, 13.8%). This report has the significant association of elderly age within increased hazards of death of COVID-19 patients, and consistent findings in Wuhan china, 29 Irina, 6 Tel Aviv, 30 and Riyadh, Saudi Arabia. 31 This might be when age increased SARS-COV-2 infection had poor clinical outcomes; physiologically, the T-cell will degenerate and functions will be compromised for control of viral replication. Furthermore, we found a significant association between the sex of the study participants and the hazards of inpatient mortality. The risks of death for admitted cases were increased two-fold for male than females’ cases. Consistent with study finding in Addis Ababa, 17 Nekemte, 16 Iran, 6 China, 32 and Israel. 30 According to Shi et al., 32 the reasons for the sex-related difference in SARS-CoV-2 infection are not clear. Some studies have indicated that sex-related differences might be confounded by comorbidity and females being more immune-privileged than males that might be related to sex hormones with an immune-enhancing effect like estrogen in females.28,30,32,33 This is consistent with finding in Addis Ababa, 17 Nekemte; 16 the risks of mortality for COVID-19 cases admitted with comorbidity; were four-fold higher as compared without at least one comorbidity. Several studies also reported that comorbidity was an independent risk factor that can delay viral clearances and is significantly associated with death hazard10,16,17 while other studies reported no significant association with comorbidity.28,34
Study limitations
This study had paramount limitations; mainly focused on adult admitted cases and did not include non-hospitalized all patients caused restriction differences in demographic and clinical variables. In addition, the absence of a validated data abstraction tool/questionnaire for all indices may create biases on reliability of results.
Conclusion
More than 72.4% of inpatient deaths occurred within 2 weeks after admission. The cured and expired cases had distinct hematologic and clinical features, which were analyzed using principal components analysis. The mortality risk factors for COVID-19 admitted cases were recognized in this report on Multivariate Cox regression including being male, age (⩾61) years, baseline comorbidity, and neutrophil count ⩽ 65 103/uL.
Footnotes
Acknowledgements
The Pawe Woreda Health Bureau, administrative staff members have been supported during the data collection for this research. The authors thank the senior researcher Mr. Tamirate Shewano of an epidemiologist and senior researcher for his unreserved editing and proof reading of last version on English language of the manuscript.
Author contributions
F.K., T.K., and T.G. conceived the study desgin. F.K. and T.K. supervised the data collection, did the analysis, and wrote the manuscript. Finally, all authors commented, edited the draft, and approved the final version of this manuscript.
Availability of data and materials
All relevant data are within this manuscript as supporting information
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
We obtained ethical clearance from our institution committee by looking at the research aims and methodologies of researchers to make sure that the research will be conducted in a way that protects the dignity, rights, and safety of the admitted COVID-19 participants. The ethical and research board (IRB) of Pawe Woreda Health Bureau approved this proposal for data collection with protocol refill no. PWOH/84/2013. Letter of data collection was obtained from the board and submitted for the two COVID-19 treatment centers with a meeting number 012/2013 for data accessed from the patient.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: For this study, material and financical support was from the Pawe Woreda Health Bureau administrative staffs. However, the funders had no role except giving support questionairy, wage data collectores and other financial aids during the data collections.
Informed consent
Since the data was collected on secondary file of recorded review the Woreda Health Burea waived informed consent from each hospitalized adult ⩾18 patient. But confidentially of data was maintained secured at end levels.
