Abstract
Background:
The coronavirus disease 2019 is highly infectious and patients with coronavirus diseases demonstrated a series of clinical symptoms caused by severe acute respiratory syndrome coronavirus-2. Without a break of the wave’s hematological profile of the patients is still ambiguous and differs from wave to wave.
Objective:
This study aimed to assess the hematological profile among coronavirus disease 2019 patients during the first and the second waves in Ethiopia.
Methods:
A multi-centered facility-based retrospective cohort study design was conducted at six coronavirus disease 2019 treatment centers. A total of 538 study participants were enrolled in the selected coronavirus disease 2019 treatment centers during the first and second waves of the pandemic. The demographic characteristics, underlying diseases, symptoms, and hematological parameters of patients were recorded. Data were entered into Epi-Data Manager 4.6 and analyzed using STATA 14.2. An independent sample t-test was used to assess the mean differences in hematological parameters across waves. Associations between categorical variables across waves were also determined using Chi-square and Fisher exact tests.
Results:
Among the total study participants, 240 (44.6%) and 298 (55.4%) patients were taken from wave-1 and wave-2, respectively. The average age of the study participants was 56.44 ± 16.25 years. The most frequent comorbidities in this study were hypertension, diabetes mellitus, ischemic heart disease, and asthma. The most presenting symptoms of COVID-19 infection across the two waves were presented. In the first wave, dry cough 166 (69.2%), fatigue 153 (63.75%), shortness of breath 148 (61.67%), and fever accounted 116 (48.33%), while during the second wave, dry cough 242 (81.2%), fatigue 244 (81.88%) shortness of breath 204 (68.47%) and fever account 180 (60.40%). White blood cells (WBC), neutrophils, and lymphocytes had shown increment during the first wave. Platelet count and platelet distribution width (p < 0.001) had significant mean differences across the two waves, while the other had no significant mean difference.
Conclusion:
In the present study, dry cough, fatigue, shortness of breath, and fever were found to be the most presenting symptoms of COVID-19 infection across the two waves. Only the platelet count and platelet distribution width had significant mean differences across the two waves, while the other had no significant mean difference across waves.
Introduction
Severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) caused coronavirus disease (COVID-19) is a large family of enveloped positive single-stranded ribonucleic acid (RNA) viruses of both medical and vertebrate importance. 1 Because the virus particle is expiated and shows a characteristic “corona” (crown) of spike proteins around its lipid envelope, the names of the viruses are referred to as coronaviruses. 2 It is a massive circle of relatives of viruses that cause ailments starting from the unusual bloodless to extra extreme illnesses along with the Middle East Respiratory Syndrome and the Severe Acute Respiratory Syndrome. 3
The 2019 novel coronavirus (nCoV) is a new stress that has now no longer been formerly recognized in human beings; it was first detected in Wuhan, China, in December 2019 and has swiftly unfolded to the United States and across the world. 4 It was declared a public health emergency on 30 January 2020 by the World Health Organization (WHO). 5 On 11 March 2020, WHO declared it a pandemic disease. 6 It has been spread very quickly; according to the WHO epidemiological report as of 30 July 2023, over 768 million confirmed cases and over 6.9 million deaths have been reported globally. 7
The disease is highly infectious, and patients with coronavirus diseases demonstrate a series of clinical symptoms, including raised body temperature, dry cough, diarrhea, headaches, myalgia, vomiting, anorexia, multiple organ dysfunction are all symptoms of multiple organ dysfunction, 8 fever, fatigue, myalgia, and dyspnea. 4 The clinical manifestations of novel COVID-19 vary from asymptomatic to acute respiratory distress, depending on the virus route of entry, virus load, host immunity, age, and comorbidity, 9 like cardiovascular disease, diabetes, chronic respiratory disease, and cancer. The severe stage of the pandemic may be characterized by acute respiratory distress syndrome, difficult-to-tackle, septic shock, metabolic acidosis, and bleeding and coagulation dysfunction.10,11
The current existing report on laboratory-confirmed coronavirus cases reported changes in the patient’s hematological parameters, including WBC, lymphocytes, monocytes, neutrophils, Red blood cells (RBC), hemoglobin, and platelets. 12 As a result of their revealed changes, hematological measures (parameters) can be used as predictive markers; this finding is correlated with the study reported in the study done in Iran. 13 Many types of research have proven that comorbidities, age, and abnormalities of numerous scientific biomarkers may be essential to apprehending the sickness severity.14,15 Even though the clinical characteristics of the COVID-19 pandemic have been widely reported, the summary of changes in the common hematological variables that are observed in patients with various waves of COVID-19 infection is still not well investigated, the published laboratory findings of COVID-19 infection are collected from hospital-admitted patients at a single time point which limits our understanding of the dynamic hematological changes during the disease, clinical symptoms and severity of the disease is differing across a wave. 16 The consistency and difference of the hematological profiles of COVID-19 patients across waves remain largely unknown. Therefore, the present study aimed to assess the hematological profiles of COVID-19 patients during the first and second waves in Ethiopia.
Methods
Study design and period
A multi-centered hospital-based retrospective cohort study design was conducted at six COVID-19 treatment centers (Jimma University Medical Center (JUMC), St. Peter’s Specialized Hospital, Eka Kotebe General Hospital, Bethzatha General Hospital, Halleluyah General Hospital and Bahir Dar University Tibebe Ghion Specialized Hospital) in Ethiopia from January 2022 to June 2022.
Population
Source population: All individuals who were tested for COVID-19 at the selected treatment centers.
Study population: COVID-19-positive individuals who were confirmed by polymerase chain reaction (PCR) in the selected treatment centers.
Eligibility criterion
COVID-19 patients who were confirmed by PCR and had common hematological parameters were included; however, patients who had no confirmed COVID-19 viruses by PCR and had no common Hematological parameters were excluded.
Sample size determination
The sample size (n) required for the study was calculated using a single population proportion formula by considering the following assumptions. A 95% confidence level with
A total of 538 study participants (COVID-19 suspected and tested patients were selected from the selected COVID-19 treatment centers. Finally, the study participants were proportionally allocated and selected using a simple random sampling technique from each treatment center accordingly.
Variables
Dependent variables
Hematological profiles of COVID-19 patients across waves
Independent variables
Sociodemographic factors: Sex, age, residency, educational status, marital status, Comorbidities: Chronic liver disease, on anticoagulant therapy before admission to the center, hypertension, ischemic heart disease, diabetes mellitus, asthma, viral hepatitis,
Clinical and related variables: WHO clinical stage, hospital stay, physical examinations, signs and symptoms.
Hematological parameters: WBC count, neutrophil, lymphocyte, neutrophil/lymphocyte ratio, monocyte, basophil, hemoglobin, mean corpuscular volume, mean corpuscular hemoglobin, mean corpuscular hemoglobin concentration, red cell volume distribution width-coefficient of variation, platelet count and activated partial thromboplastin time.
Data collection tool and procedure
Data collection tool: The data were collected by using a structured and pre-tested checklist. The checklist was adopted from the WHO tool for data extraction and developed after an extensive review of the literature and similar study tools.17,18
Data collection procedure: Twelve data collectors and six supervisors (Laboratory Technologists) were selected and trained for data collection by the principal and co-investigators.
They were allowed to exercise utilization of the tool with the principal investigator for one day before starting normal data extraction. Afterward, data were extracted from suspected COVID-19 and tested patients’ medical records (electronic or paper-based). Basic information such as age, gender, educational status, ethnicity, nationality, marital status, medical history, comorbidity, clinical signs and symptoms, physical examinations, COVID-19 status, and hospital stay variables were collected from the admission records. Hematological parameters were also collected from the medical records of the patients.
Operational definition
Moderate cases of COVID-19: Moderate cases had clinical signs of pneumonia (fever, cough) but no signs of severe pneumonia (oxygen saturation 90%). 19
Severe cases of COVID-19: Severe cases were admitted to the intensive care unit due to severe hypoxemia (oxygen saturation <90%). 20
Critical cases of COVID-19: Acute respiratory distress syndrome (ARDS) within 1 week of known clinical insult or new or worsening respiratory symptoms, chest imaging indicating bilateral opacities not fully explained by volume overload, labor or lung collapse, respiratory failure not fully explained by cardiac failure or fluid overload, oxygenation impairment in adult and children, acute life-threatening organ dysfunction, evidence of septic shock with characteristics of persistent hypotension despite volume resuscitation in adults and children. 20
Reference interval of hematological parameters: was determined by considering the Lower limit of 95% confidence interval (CI) and Upper limit of 95% CI of the hematological parameters reported in Ethiopia. 21
Chronic lung disease: Defined as a group of lung diseases that block airflow and make it difficult to breathe. It includes asthma, chronic obstructive pulmonary disease (COPD), pulmonary fibrosis, asbestosis, pneumonitis, and other lung conditions.
Missing data: The values of the variables of interest were not recorded in the patient’s record book.
Quality control
To ensure the data quality high emphasis was given to designing a data collection checklist. The effectiveness and clarity of the checklists were tested on 5% of the total sample before data collection from adjacent COVID-19 treatment centers to ensure that the prepared checklists needed to make necessary adjustments and the required corrections were made accordingly. Two days of training were provided to data collectors and supervisors for the survey, the method of data collection, the checklists, and other related issues. During data collection, each data collector’s issues and directions were monitored, and necessary corrections were made. The integrity of the collected data was checked, and both the data collector and the investigators took corrective action in the field. Daily meetings were held between data collectors, supervisors, and chief authors to discuss potential issues and assess the progress of data collection. Facilitators and supervisors were assigned to support and guide the data collection and so doing it was increased the chances of consistency and reliability in the collected data.
Statistical analysis
The collected data was checked for completeness and consistency. The data was entered into the Epi-Data Manager version 4.6 and exported to STATA version 14.2 for statistical analysis. Necessary data processing, explorations, and data management like recoding, categorizing, merging, computing, and counting were done before the actual data analysis. Categorical variables were presented as frequencies with percentages, whereas mean, standard deviation, median, and range were used to describe continuous variables and displayed in graphs, charts, and tables. Evaluation of data normality was performed using the Shapiro-Wilk test. Non-normally distributed continuous variables were analyzed using non-parametric tests. 22 Independent sample t-tests were applied to compare the hematological profiles’ across the wave of COVID-19 patients and COVID centers respectively. Categorical variables were compared using the Chi-square test or Fisher’s exact test. p ≤ 0.05 was considered as a statistically significant.
Results
Sociodemographic characteristics and medical history of COVID-19 patients
In this study, 538 COVID-19 patients were included to identify the hematological profiles across waves. In the first (13 March 2020–18 January 2021) and the second wave (19 January 2021–28 June 2021), 240 (44.6%) and 298 (55.4%) patients were recruited, respectively. The minimum age was 1 and the maximum age was 97 years, with an average age of 56.44 ± 16.25. Most of the patients in the first and second waves were males, 162 (67.5%) and 183 (61.4%), respectively.
Twenty-six (10.83%) of the patients at wave-1 and 43 (14.43%) of the patients at wave-2 were illiterate. Of the 240 patients, 93 (38.75%) had hypertension, 83 (34.58%) had diabetes mellitus, 44 (18.33%) had chronic liver disease, and 31 (12.92%) had ischemic heart disease medical history during the first COVID-19 waves. During the second wave, out of 298 patients, 116 (38.93%) had hypertension, 98 (32.89%) had diabetes mellitus, and 37 (12.41%) had a medical history of ischemic heart disease (Table 1).
The distribution and association between sociodemographic characteristics and medical history of COVID-19 patients during the first and second waves of COVID-19 in Ethiopia (N = 538).

p-values correspond with the Chi-squared test values, and bold with asterisk (*) indicates significant associations; Unknown: is for unseen/undetected or missing results.
COVID-19, coronavirus disease 2019; AIDS/HIV, acquired immune deficiency syndrome/human immune virus; WHO, World Health Organization.
Distribution of clinical-related and hematological parameters among the first and second waves of COVID-19 patients
More than half of the study participants, 132 (55.23%) patients at the first wave and 173 (58.05%) patients at the second wave were severe. Whereas 72 (30.13%) and 35 (14.64%) were in moderate and critical stages during the first wave, respectively. And 81 (27.18%) in moderate and the remaining 44 (14.77%) were in the critical stage during the second wave (Figure 1).
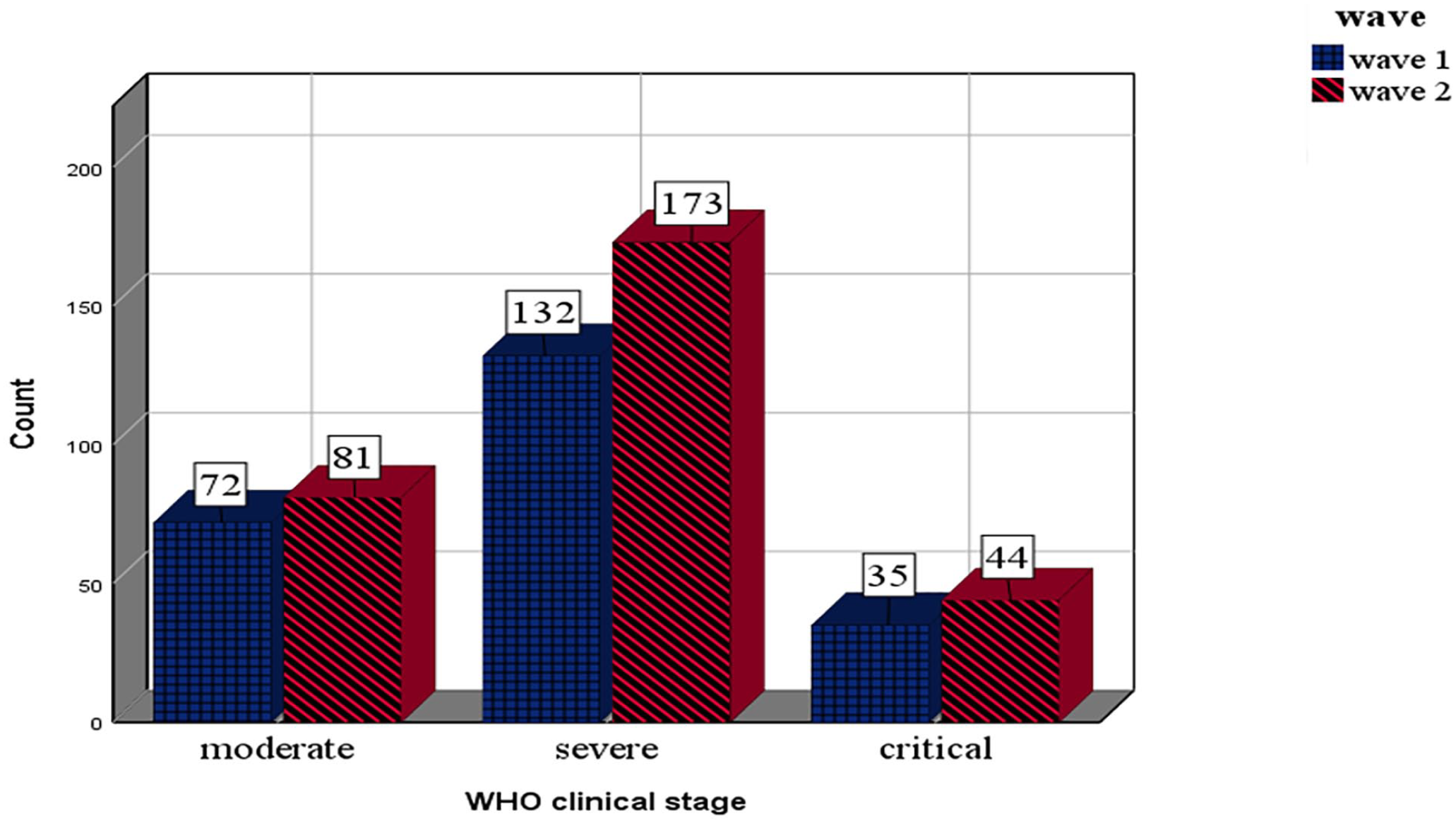
WHO clinical stages of COVID-19 disease across waves.
The most relevant clinical symptoms during the first and second waves of symptomatic COVID-19 patients were recorded. Out of 240 patients in the first wave, 166 (69.2%) had a dry cough, 153 (63.75%) had fatigue, 148 (61.67%) had shortness of breath, and 116 (48.33%) had a fever. In the second wave, 244 (81.88%) had fatigue, 242 (81.2%) had a dry cough, 204 (68.47%) had shortness of breath, and 180 (60.40%) had a fever. The Chi-square test of association indicated that there was a statistically significant association between headache, myalgia, chest pain, dry cough, fatigue, fever, joint pain, enlarged glands, rash, shortness of breath, dark or bloody urine, yellow skin/eyes, stiff neck, difficulty swallowing, wheezing, diarrhea, and chills/rigors across the waves of COVID-19 (Table 2).
The prevalence of COVID-19 symptoms among COVID-19 patients during the first and second waves of COVID-19 pandemic in Ethiopia (N = 538).
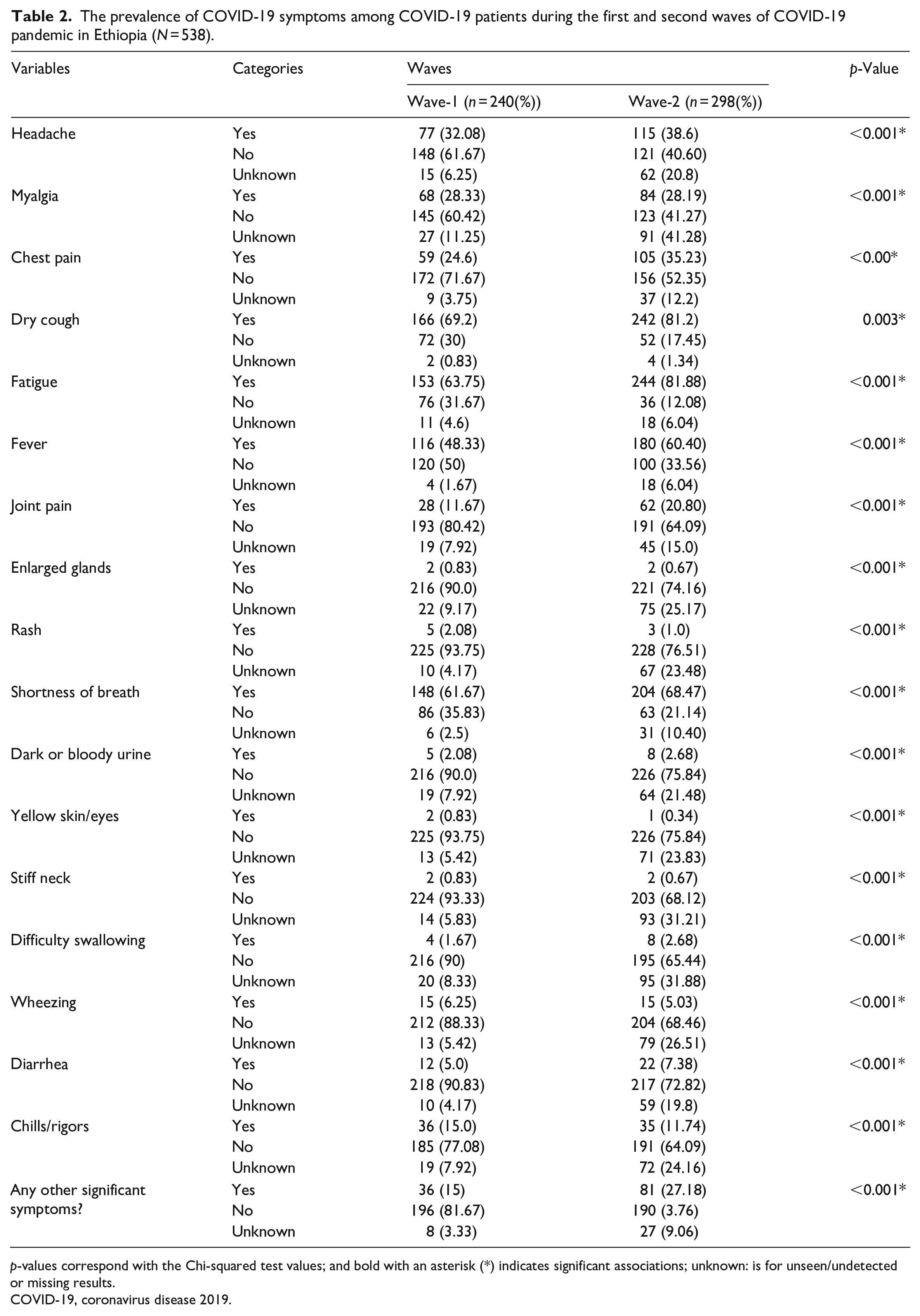
p-values correspond with the Chi-squared test values; and bold with an asterisk (*) indicates significant associations; unknown: is for unseen/undetected or missing results.
COVID-19, coronavirus disease 2019.
Changes in hematological profiles of COVID-19 patients
The findings of these studies assessed the hematological parameters of COVID-19 patients at each wave from its normal range values. Out of 240 COVID-19 patients at the first wave, most of them, 161 (67.6%), had WBC value ranges from 3 to 11.2, 179 (76.5%) patients had neutrophil values above the normal range (1.18–6.73), 104 (43.3%) of patients’ lymphocytes had the hematological parameter values above the normal range value (>4.5). From the total of 298 patients at the second wave, 210 (70.7%) of patients had WBC values in the normal range, 186 (72.4%) Neutrophils were above the normal range of 6.7, and 122 (40.9%) lymphocyte values were above the normal range (Table 3).
The distributions of hematological parameters range in value among COVID-19 patients during the first and second waves of the COVID-19 pandemic in Ethiopia (N = 538).

COVID-19, coronavirus disease 2019; WBC, white blood cell count; fl, femtoliter.
The independent sample t-test result indicates that only the platelet count (<0.001) and platelet distribution width (<0.001) had significant mean differences across the two waves, while the other had no significant mean difference (Table 4).
The independent sample t-test for hematological parameters of COVID-19 patients across waves in Ethiopia (N = 538).
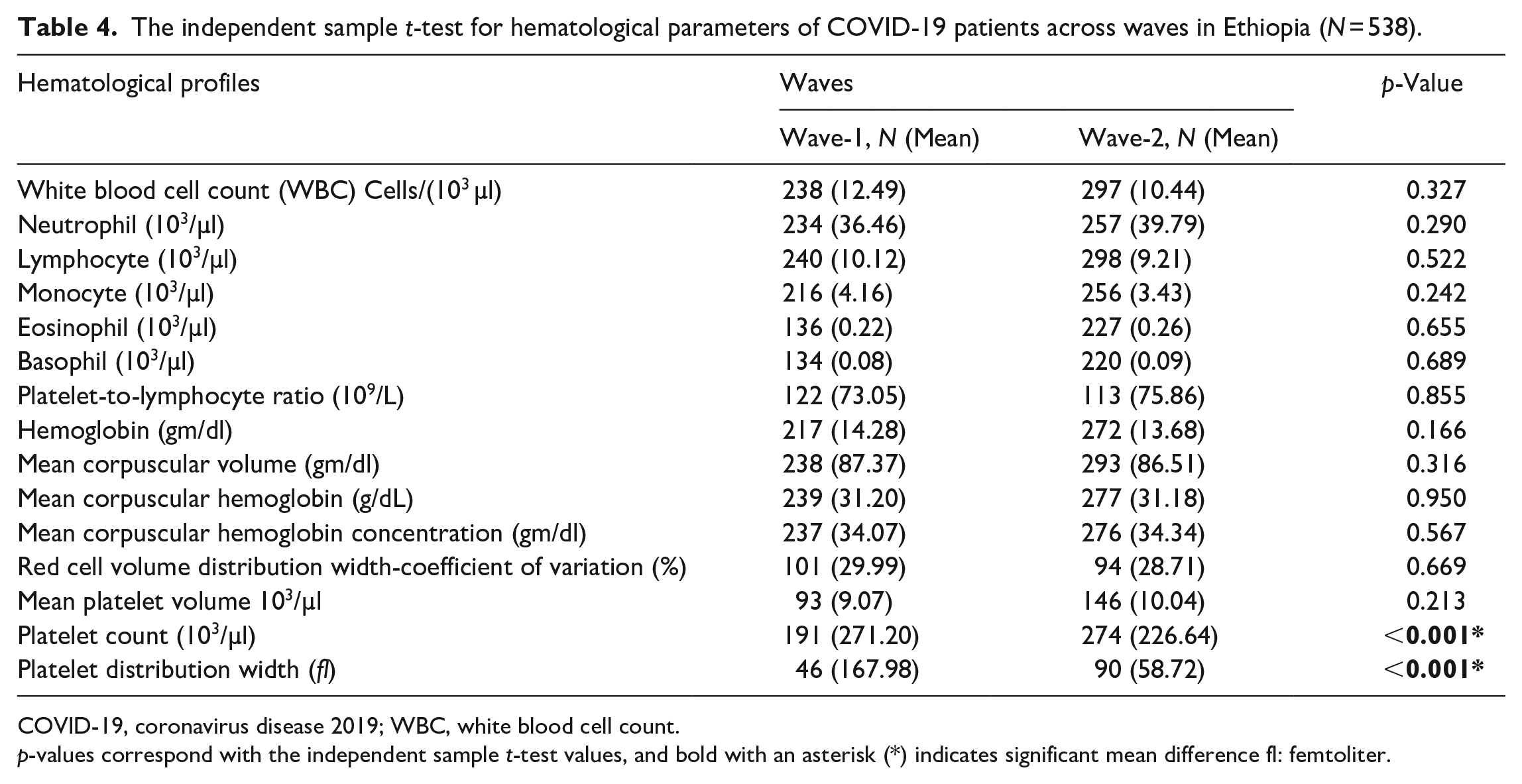
COVID-19, coronavirus disease 2019; WBC, white blood cell count.
p-values correspond with the independent sample t-test values, and bold with an asterisk (*) indicates significant mean difference fl: femtoliter.
Discussion
COVID-19 pandemic cases worldwide and different speculations of its effect on the human body are also flashing every day. We still have a lot to know about the effect of COVID-19 on different hematological profiles in patients who were infected with COVID-19 across each wave. Along with the molecular test of RT-PCR, exploring the change of hematological parameters obtained through complete blood count (CBC) is very important to understand the clinical status and prognosis of COVID-19 patients. 23 Therefore, we assessed the hematological parameters of 538 COVID-19 patients during wave-1 and wave-2 in Ethiopia. In this study, in addition to hematological parameters, we assessed demographic characteristics (age, gender, educational status, and marital status), medical history with COVID-19, clinical stages, and symptoms of patients COVID-19 patients. The majority of the study’s participants were enrolled in the second (55.39%) and in the first (44.6%) wave. This finding is similar to study conducted in Portugal. 24 This can be transposed to the general situation of the country since the majority of the cases were diagnosed during the second wave.
In this study, males are more infected than females by COVID-19 during both waves; this is supported by the study reported in.25–28 Many hormonal, genetic, immunologic, lifestyle, and behavioral factors might increase the risk of the infection. In this study, the most affected age range was 53–69 years, with 86 (35.83%) and 109 (36.58%) during wave-1 and wave-2, respectively. This study is in line with the study conducted in Lebanon. 25 This finding contradicts the study reported in India. 29 Education status and marital status were significant with the COVID-19 infection in both waves.
The most relevant clinical symptoms during the first and second waves of the symptomatic COVID-19 patients were recorded, which was supported by the study reported by Leulseged et al. 27 Out of 240 patients in the first wave, the patients had; dry cough, fatigue, shortness of breath, and a fever. In the second wave had fatigue, dry cough, shortness of breath, and 180 (60.40%) had a fever. There was a statistically significant association between headache, myalgia, chest pain, dry cough, fatigue, fever, joint pain, enlarged glands, rash, shortness of breath, dark or bloody urine, yellow skin/eyes, stiff neck, difficulty swallowing, wheezing, diarrhea and chills/rigors and waves of COVID-19, this study is comparable with the study done by Yang et al. 30 In this finding, more than half, 132 (55.23%) patients at the first wave and 173 (58.05%) at the second wave were in severe clinical stages; this finding is similar to the study reported.19,31 The most frequent commodities in this study were hypertension, diabetes mellitus, ischemic heart disease, and asthma; this study is similar to the study conducted by Mollinedo-Gajate et al.32,33 In this study, white blood cell count (WBC), neutrophil, lymphocyte, basophil, hemoglobin, mean corpuscular volume, mean corpuscular hemoglobin and platelet count were increased from wave one to wave two. However, the neutrophil to lymphocyte ratio has decreased; this study is contradicted in Madrid, Spain. 32 In this study, among 179 (76.5%) neutrophilia and 104 (43.4%) with presented lymphocytosis during wave-1 and wave-2, these results are in line with the study done in Singapore and Addis Ababa, Ethiopia.20,34 But contradicted with the study done in. 35
The independent sample t-test result indicates that only the platelet count (<0.001) and platelet distribution width (<0.001) had a significant mean difference across the two waves; this finding is in line with the study reported by Shankaralingappa et al., 36 while the other had no significant mean difference.
Strengths and limitations of the study
As a strength, this study provides new insight into the substantial role of other Hematological profiles in patients with COVID-19 across waves. This will further help to formulate a robust plan to control and mitigate the pandemic at large. This study also had limitations; first, it is known that a retrospective cross-sectional study is limited to a multicenter and provides no information on a cause-and-effect relationship. Secondly, only a limited number of hematological profiles are studied, limiting the findings’ implications. There were poor medical records and documentation in the selected COVID-19 treatment centers. These profiles and clinical characteristics need further investigations and should be used for risk stratification in patients with COVID-19 as well as the strain investigation of SARS-CoV2.
Conclusion
This study finding presented the first study on multicenter study design across the first and second waves of COVID-19 infected with SARS-CoV2 in Ethiopia. Dry cough, fatigue, shortness of breath, fever, and fatigue, dry cough, shortness of breath, and fever were the most presenting symptoms of COVID-19 infection in Ethiopia during the first and second wave of the pandemic. The most frequent comorbidities in this study were hypertension, diabetes mellitus, ischemic heart disease, and asthma. Only the platelet count and platelet distribution width had significant mean differences across the two waves, while the other had no significant mean difference across waves.
Supplemental Material
sj-docx-1-smo-10.1177_20503121241253522 – Supplemental material for Assessment of the hematological profiles among COVID-19 patients during the first and second waves in Ethiopia: A multicenter retrospective cohort study
Supplemental material, sj-docx-1-smo-10.1177_20503121241253522 for Assessment of the hematological profiles among COVID-19 patients during the first and second waves in Ethiopia: A multicenter retrospective cohort study by Minale Fekadie Baye, Diliab Desta, Samuel Hunegnaw, Ahmed Johar, Israel Bekele, Kebenesa Angasu, Nebiyou Simegnew Bayleyegn, Habtamu Abebe Getahun and Assefa Legesse Sisay in SAGE Open Medicine
Supplemental Material
sj-docx-2-smo-10.1177_20503121241253522 – Supplemental material for Assessment of the hematological profiles among COVID-19 patients during the first and second waves in Ethiopia: A multicenter retrospective cohort study
Supplemental material, sj-docx-2-smo-10.1177_20503121241253522 for Assessment of the hematological profiles among COVID-19 patients during the first and second waves in Ethiopia: A multicenter retrospective cohort study by Minale Fekadie Baye, Diliab Desta, Samuel Hunegnaw, Ahmed Johar, Israel Bekele, Kebenesa Angasu, Nebiyou Simegnew Bayleyegn, Habtamu Abebe Getahun and Assefa Legesse Sisay in SAGE Open Medicine
Footnotes
Acknowledgements
The authors are thankful for the financial support provided by the JUIH. We would like to thank the Medical directors and staff of JUMC, St. Peter’s Specialized Hospital, EKA Kotebe General Hospital, Bethzatha General Hospital, Hallelujah General Hospital, and Bahir Dar University Tibebe Ghion Specialized Hospital) Ethiopia for their positive and heartfelt collaboration and assistance during the study.
Author contributions
Conceptualizations: Assefa Legesse Sisay, Nebiyou Simegnew Bayleyegn, Kebenessa Angasu, Habtamu Abebe Getahun. Data curation: Assefa Legesseg, Minale Fekadie Baye, Samuel Hunegnew, Diliab Desta. Formal analysis: Assefa Legesse Sisay, Habtamu Abebe Getahun, Minale Fekadie Baye, Kebenessa Angasu, Israel Bekele. Investigations: Minale Fekadie Baye, Assefa Lgesse Sisay, Kebenessa Angasu, Samuel Hunegnew, Diliab Desta. Methodology: Assefa Legesse Sisay, Minale Fekadie Baye, Habtamu Abebe, Nibiyou Simegnew Getahun, Israel Bekele. Supervision: Nibiyou Simegnew Bayleyegn, Kebenessa Angasu, Habtamu Abebe Getahun, Assefa Legesse Sisay, Samuel Hunegnew, Israel Bekele. Software: Assefa Legesse Sisay, Habtamu Abebe Getahun, Minale Fekadie Baye, Kebenessa Angasu, Israel Bekele and Diliab Desta. Validation: Minale Fekadie Baye, Habtamu Abebe Getahun, Diliab Desta, Assefa Legesse Sisay. Visualization: Minale Fekadie Baye, Assefa Lgesse Sisay, Samuel Hunegnew, Diliab Desta. Writing original Draft: Assefa Legesse Sisay, Habtamu Abeb Abebe Getahun, Minale Fekadie Baye. Writing Review and Editing: Assefa Legesse Sisay, Habtamu Abebe Getahun, Diliab Desta, Samuel, Hunegnew, Nibiyou Simegnew Bayleyegn, Minale Fekadie Baye, Israel Bekele.
Availability of data and material
The datasets used and analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the JUIH.
Ethical considerations and consent to participate
Ethical clearance and approval for the research were obtained from the Institutional Review Board (IRB) of Jimma University with Ref. No. JUIRB43/22. The director of the selected COVID-19 treatment centers was communicated with a formal letter written by Jimma University Institute Health (JUIH), Research, and Innovation director office. A support letter was secured from each treatment unit. When reviewing patient records, patient medical record numbers were used to differentiate study participants during data collection, and the collected data were kept confidential. We used secondary data retrieved from the patient cards/databases that had already been collected for the patient’s care in the selected treatment center. Due to the reason that inconvenience and difficulty in reaching those COVID-19 patients, we did not seek individual-level written informed consent. Instead, the Ethical Review Board (IRB) of the university waived written consent from the Health facilities offices. The treatment center database management department had consented to each patient and caregiver to use the data for research purposes in a confidential way at the time of enrollment.
Informed consent
The director of the selected COVID-19 treatment centers was communicated with a formal letter written by Jimma University Institute Health (JUIH), Research, and Innovation director office. A support letter was secured from each treatment unit. When reviewing patient records, patient medical record numbers were used to differentiate study participants during data collection, and the collected data were kept confidential. We used secondary data retrieved from the patient cards/databases that had already been collected for the patients’ care in the selected treatment center. Due to the reason that inconvenient and difficulty in reaching those COVID-19 patients, we did not seek individual-level written informed consent. Instead, the Ethical Review Board (IRB) of the university waived written consent from the Health facilities offices. The treatment center database management department had consented to each patient and caregiver to use the data for research purposes in a confidential way at the time of enrollment.
Trial registration
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
