Abstract
In our article, we explore the transformative potential of Artificial Intelligence and Machine Learning in oculo-oncology, focusing on the diagnosis and management of ocular adnexal tumors. Delving into the intricacies of adnexal conditions such as conjunctival melanoma and squamous conjunctival carcinoma, the study emphasizes recent breakthroughs, such as Artificial Intelligence-driven early detection methods. While acknowledging challenges like the scarcity of specialized datasets and issues in standardizing image capture, the research underscores encouraging patient acceptance, as demonstrated in melanoma diagnosis studies. The abstract calls for overcoming obstacles, conducting clinical trials, establishing global regulatory norms and fostering collaboration between ophthalmologists and Artificial Intelligence experts. Overall, the article envisions Artificial Intelligence’s imminent transformative impact on ocular and periocular cancer diagnosis.
Introduction
The human eye, a complex and crucial organ of the body, is most frequently divided into the anterior and posterior segments, while little regard is given to the orbital adnexa. Since most of the conditions involving the adnexa which includes the eyelids, conjunctiva, extra-ocular muscles, lacrimal glands, etc., are similar to other parts of the body and do not carry a significant distinction in head-and-neck pathophysiology, many pathologists generally do not classify these lesions as part of a distinct region. 1
Most legions of the orbit and adnexa present as space-occupying lesions which are more frequently of neoplastic etiology rather than inflammatory or vascular but recently increased incidences of IgG4-related oculopathies and sarcoidosis have also been reported.2,3
Similarly, ocular adnexal lymphoma which occurs due to chronic inflammation and antigenic stimulation leading to the proliferation of mucosa-associated lymphoid tissue has also been reported in literature of which the largest review was conducted by Fernández et al. 4 and colleagues.
Among the neoplastic lesions of the orbit and adnexa primary malignancies are comparatively more common as compared to metastatic extension of neoplasms from other regions of the body including the nasopharyngeal as well as renal cell carcinoma.5,6
Considering the nature of diagnosis of adnexal tumors, which depend highly on visual clinical assessment as well as histology, it provides us with a great arena for the incorporation of AI and ML to develop algorithms to aid in the detection, diagnosis as well as prognosis of adnexal and orbital tumors.
Previously, ML techniques have been applied to fundus images with promising results in detecting ocular diseases such as diabetic retinopathy (DR),7–9 age-related macular degeneration (AMD),10–12 glaucoma,13–15 myopic maculopathy, 16 retinopathy of prematurity, and papilledema, matching the performance of expert ophthalmologists. While these ML systems have demonstrated a high level of diagnostic accuracy work on the anterior segment of the eye evidence regarding their efficacy for detecting adnexal and orbital tumors has been comparatively scarce.
As of 2023, the US Food and Drug Administration (FDA) has approved the use of AI methods in clinical settings, including devices that utilize smartphone applications to detect clinical photographs and suggest a diagnosis. 17
The field of AI is rapidly expanding, with the ongoing evolution of research in this arena. The purpose of this review is to provide an overview of the applications of AI in the diagnosis and management of adnexal and orbital tumors. We have reviewed the studies and breakthroughs in machine learning (ML) that encompasses a variety of subfields related to orbital and adnexal cancers.
Artificial intelligence and its branches
AI can be broadly classified into three types: artificial narrow intelligence, general artificial intelligence, and superintelligence AI. Narrow AI, which can intelligently perform a specific task, is currently the most widely used form of AI. General AI can perform any cognitive task that a human can, while super AI can surpass human capabilities in performing virtually any cognitive task. However, only weak or narrow AI is currently available, such as Apple’s SIRI, which trains machines to execute a specific task. Reactive machines are AI systems that respond intelligently to present circumstances, such as IBM’s Deep Blue system, ChatGPT, and Google’s AlphaGo. AI machines with limited memory, such as self-driving cars, like Tesla can save experiences or data for a finite time period. The ability to comprehend human emotions, beliefs, and social interaction, known as the theory of mind, has yet to be developed in AI machines. 18
Machine learning (ML) is a subfield of AI in which trained algorithms learn from their experiences without requiring explicit programming. This can be achieved through supervised, semisupervised, or unsupervised learning processes. In supervised learning, machines are trained using datasets of problems and their corresponding solutions. Through trial and error, the machines learn to select the correct responses. In unsupervised learning, machines analyze incoming data without any predetermined solution. Semisupervised learning is a hybrid approach that uses both labeled and unlabeled data to train the machine. 19 Deep learning, on the other hand, is a type of ML that uses multiple layered deep neural networks to recognize and learn unique features of a dataset. 20 Artificial neural network (ANN) is a computational model based on the structure and functions of biological neural networks. 21 The most primitive type of ANN is the feedforward neural network, which has an input, hidden, and output layer. Data enters through the input layer, passes through the hidden layer, and exits through the output nodes. Multiple hidden layers are possible.22,23 Convolutional neural networks (CNNs) are a variant of deep, feed-forward ANN primarily used for analyzing visual imagery. They consist of convolutional and pooling layers that allow the network to encode image features. 24
Conjunctival lesions and the role of artificial intelligence
Among benign overgrowth of cells on the surface of the eye a conjunctival nevus is the most frequently found lesion. It presents as a pigmented lesion which leads to the patient often seeking medical evaluation. The biggest concern with any pigmented lesion and nevus is the possibility of it being a melanoma. Conjunctival melanoma is an uncommon and potentially life-threatening cancer that develops in the front part of the eye, and it is linked to a mortality rate of as much as 30%. 25 It is associated with higher mortality among patients with a higher stage which leads to poor prognosis and the treatment involved is often costly. 26
Despite conjunctival melanoma typically manifesting as a pigmented nodular conjunctival lesion, 27 atypical variants can lead to delayed diagnosis. The condition can originate from primary acquired melanosis or nevus but exhibits various malignant characteristics.
Surgery stands as the primary approach for treatment. During the surgical procedure, additional treatments are often applied to the exposed scleral base and adjacent conjunctival margins to eliminate any potential residual tumor cells. These supplementary treatments encompass a variable combination of methods such as the application of absolute alcohol, instillation of Mitomycin C (MMC) or Interferon-alpha-2B, and the utilization of double freeze–thaw cryotherapy. In cases where tumor margins are poorly defined, topical chemotherapy is employed to address the entire ocular surface, enabling the treatment of diffuse or multifocal lesions and hidden areas. 28
Despite the implementation of surgical techniques and intraoperative adjuvant treatments, conjunctival melanoma (CM) exhibits a significant incidence of local recurrence, approximately 50%–60% within a 5-year period. The recurrence rate can be mitigated with the adoption of combined radiotherapy treatments. 29 Adjuvant radiotherapy is gaining widespread acceptance, primarily through the use of brachytherapy. While the application of external-beam conventional X-ray techniques, stereotactic and radiosurgery, or proton-beam radiotherapy is less common, long-term results from these approaches are eagerly anticipated.
Similarly, another encountered malignant change is squamous conjunctival carcinoma. Globally, the incidence of conjunctival squamous cell carcinoma (SCC) varies geographically, ranging from 0.03 to 1.9 per 100,000/year in Caucasians and 3 to 3.4 per 100,000/year in African populations. The primary risk factor for ocular surface squamous neoplasia (OSSN) is chronic UV B radiation exposure, affecting those in direct sunlight and outdoor occupations. It often appears as a unilateral vascularized limbal mass in the inter-palpebral fissure. Management included surgical resection, topical chemotherapy, immunomodulation, antiviral medications, photodynamic therapy, cryotherapy, and radiotherapy (external beam, proton beam, Strontium-90 or Ruthenium-106 plaque brachytherapy). Recently, proton beam radiotherapy (PBRT) has been considered as an alternative to surgical methods and other conservative approaches since it spares neighboring tissues. 30
Given the current situation in many countries, characterized by an aging population and limited healthcare resources, early diagnosis and feasible detection are crucial to reduce conjunctival melanoma and SCC related mortality. The standard clinical examination involves ophthalmologists using a slit lamp to observe the ocular surface, followed by a biopsy for confirmation. However, in regions with restricted healthcare access and a scarcity of trained ophthalmologists, this poses a significant challenge, particularly in low- and middle-income countries (LMICs).
Considering these limitations AI-based diagnostic systems have been developed over the past few decades. 31 Additionally, digital devices, including smartphone cameras, compact digital cameras, slit-lamp cameras, and dermatoscopy, are extensively utilized as disease screening tools due to their accessibility and user-friendly nature. While numerous machine learning studies have concentrated on skin melanoma, the use of recent technology for identifying conjunctival melanomas has not achieved its potential. Training conventional deep learning networks to detect conjunctival melanoma poses challenges due to the lack of extensive datasets containing images of conjunctival disorders.
Since scarcity of data creates a difficult obstacle, a novel technique called low-shot learning is currently being investigated, aimed at addressing the limitations of conventional deep learning models. This approach focuses on improving the performance of models with limited training data, potentially offering a solution for detecting conjunctival melanoma given the scarcity of available datasets. 32
In this regard, Yoo et al. 33 and colleagues conducted a study, employing deep learning models for conjunctival melanoma detection, a recent experiment utilized 3D phantoms to demonstrate the model’s capability to capture ocular surface images of the eye’s anterior segment, specifically for conjunctival melanoma, using a smartphone. Their study gathered publicly accessible ocular and clinical images of patients with conjunctival disorders (Figure 1(a)–(c)). Utilizing Google image search with keywords like “conjunctiva,” “pterygium,” and others, anterior segment images were collected. Two ophthalmologists manually classified the images, resolving disagreements through discussion. Ambiguous images were excluded for clarity. The search produced de-identified images falling into four significant and prevalent categories: conjunctival melanoma (136 images), nevus or melanosis (93 images), pterygium (75 images), and normal conjunctiva (94 images). In this study, new instances of conjunctival images were created through data augmentation using well-known GAN models, such as CycleGAN and progressive growing of GAN (PGGAN).34,35 This study, achieving a high accuracy and ROC-AUC of 97.2% and 0.983, respectively, in an independent test dataset. Additionally, the model demonstrated a 94.0% accuracy using 3D melanoma phantom images generated from ocular surface images captured by digital cameras, including smartphones. This suggests the effective use of AI techniques, specifically smartphone cameras, for conjunctival melanoma detection.
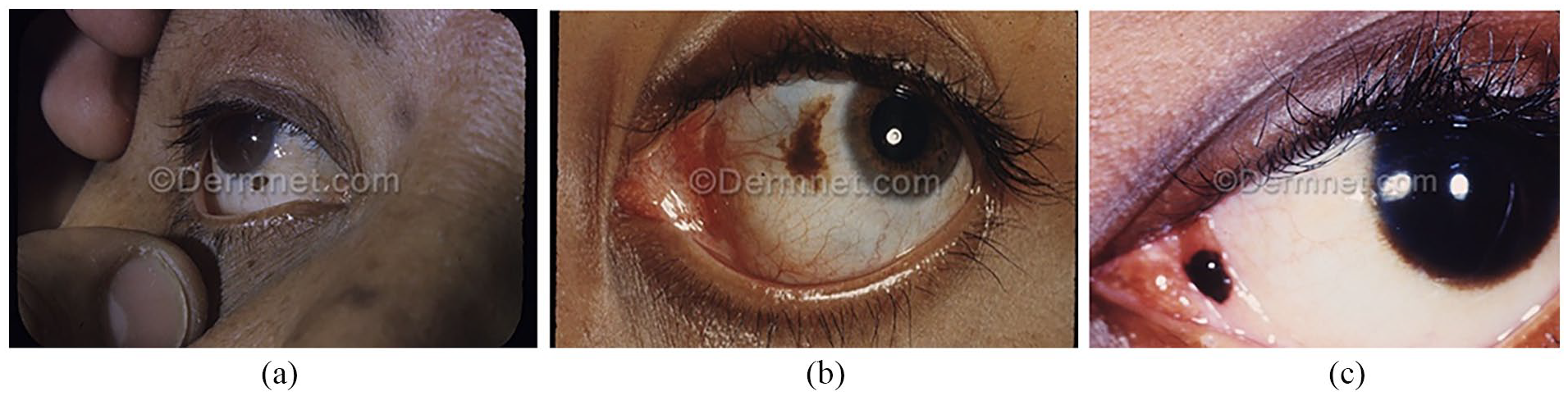
Conjunctival melanocytic nevi. 36
Eyelid lesions and the role of artificial intelligence
Eyelid tumors, both benign and malignant, are common in ophthalmology practice due to the diverse tissues present.36,37 Malignant tumors in eyelids are particularly concerning due to their proximity to the eyes, brain, and sinuses, potentially causing cosmetic issues and serious health problems. Detecting and treating these tumors early is crucial for optimal cosmetic and functional outcomes. While rare, melanoma and sebaceous gland carcinoma are highly dangerous eyelid lesions with significant mortality rates.38,39 However, when identified at an early stage (skin invasion depth ⩽0.75 mm), these tumors can have a higher survival rate. 40 Similarly, lymphomas confined to the orbital and ocular adnexa (OOAL) are an uncommon manifestation, representing around 7%–8% of all non-Hodgkin’s lymphomas (NHLs). Their morphological diversity includes variable germinal centers, plasma cells, and/or monocytic lymphocytes. Approximately 80% of eyelid-involved lymphomas originate from mature B-cells. Treatment options encompass radiotherapy (RT), chemotherapy (CHT), immunomodulating therapy, primary antibiotic treatment, surgical excision, or a combination of these approaches. 41 Thus, like any other malignancy early detection is key to a better prognosis.
As such, using a deep learning algorithm alongside eyelid tumor images could enable early detection of malignant eyelid tumors. This approach offers advantages like improved accessibility and affordability for potential cases. It also allows medical professionals and suspected patients to actively monitor and identify malignant tumors in eyelids at an earlier stage.
Considering this Li et al. 42 and colleagues developed an AI system utilizing a Faster R-CNN and deep learning classification networks. Its purpose was to autonomously detect and differentiate between malignant and benign eyelid tumors in everyday digital camera images. The study also evaluated the system’s performance in detecting common malignancies and benign tumors and compared it with ophthalmologists of varying expertise levels. A dataset comprising 1417 images was employed to create and test a detection system for eyelid tumors, delineating 1533 tumors with precise bounding boxes. Additionally, the deep learning classification system was trained and evaluated using 1533 cropped images, consisting of 1161 benign tumor images and 372 malignant tumor images, generated by the system.
The primary malignant eyelid tumors in their dataset were basal cell carcinoma (BCC) (65.9%), squamous cell carcinoma (SCC) (13.4%), and SGC (17.7%). Other malignancies, such as melanoma and actinic keratosis, constituted 3.0% (Figure 2(a)–(i)). Among benign eyelid tumors, the main types were squamous cell papilloma (SCP) (9.0%), nevus (33.2%), and seborrheic keratosis (9.1%). Additional benign tumors like hemangioma and xanthoma accounted for 47.4%, which was epidemiologically in line with published literature.

Their system’s strong performance (AUC 0.899, 91.5% sensitivity, 79.2% specificity) in external test sets confirmed its generalizability. The system’s outputs aligned well with ground truth, supported by substantial Cohen’s κ coefficients. Compared to ophthalmologists of varying expertise, the system showed higher sensitivity than juniors and seniors, akin to experts; however, its specificity was slightly lower than that of experts. These results suggest the system’s potential for cost-effective early detection of malignant eyelid tumors, sparing unnecessary exams for clearly benign cases. In conclusion, their study highlights the robustness of the deep learning system in discerning malignant from benign eyelid tumors. This AI tool could aid in proactive tumor tracking and early identification, though its performance might vary among different populations.
Datasets and databases
In the context of skin cancer, clinical and dermatoscopic images are frequently used to monitor changes in skin conditions. The emergence of new applications is set to unlock vast amounts of data that have been generated, or will be generated in the future, such as those in hospital settings, and make them available to algorithms, thus leading to an improvement in CNNs. While research-ready datasets such as those found in the ISIC archive gallery, which includes the ISIC Challenges datasets, HAM10000, and BCN20000, already exist, they are primarily focused on general dermatology and do not cater specifically to oculofacial benign or malignant tumors.44–46 Datasets like the Interactive Atlas of Dermoscopy which is a collection of multiple clinical cases, include more than 200 melanomas and basal cell carcinomas. The atlas provides a minimum of two images for each reported case, one epiluminescence microscopy and the other a plain photograph. This resource is available for research purposes at a price of 295$. 47
Another database, Dermofit Image Library 48 contains high-resolution photographs of more than 1000 benign and malignant lesions, similarly, the PH2 Dataset is a free resource that features 200 images, including 40 melanoma and 160 nevi cases. Users can access this dataset after completing an online registration form. 49 These datasets could also be utilized to create exclusive datasets for oculofacial and anterior segment tumors. Such specialized resources could further advance AI research in ophthalmology.
Current challenges in the acceptance of AI among clinicians and patients
A recent survey-based study by Jutzi et al. 50 aimed to understand German patients’ views on the use of AI in diagnosing melanoma. The study involved 298 participants of whom 154 (51.7%) had a diagnosis of melanoma. The results showed that 94% of respondents supported the use of AI in healthcare, which is an encouraging finding for the acceptance of AI in healthcare. Furthermore, 88% of participants were willing to provide their health-related records to develop AI-based applications. Patients with a history of melanoma were more inclined to use AI applications for early diagnosis. A separate qualitative study with 48 patients also reported a positive attitude toward AI for cancer screening, provided that it did not interfere with the doctor–patient relationship. 51 These findings emphasize the importance of involving patients in the development and implementation of AI-based healthcare solutions.
Future of AI in the field of oculo-oncology
The increasing prevalence of general skin lesions, such as melanoma and dermatitis, presents a vast pool of patient data for the training, testing, and validation of AI algorithms for ocular and adnexal lesions. However, it should be noted that while the diagnostic performance of many AI algorithms does appear promising, but their validation in real-world settings is relatively limited, and patient heterogeneity in the real world may result in lower algorithm accuracy. Reporting bias may also exist, where algorithms with negative or unsatisfactory performance are less likely to be reported. To address these issues, it is important to conduct prospective clinical trials to confirm whether AI technology does translate to improved patient outcomes. Once clinically validated and robust AI systems are established, democratizing such technology to stakeholders becomes a challenge, and international, consensus-based reporting guidelines for AI-related interventions, such as CONSORT-AI and SPIRIT-AI, 52 should be implemented.
In healthcare settings, AI has a wide scope for both diagnostic and therapeutic purposes. 53 However, continuous feeding of data is needed to train machine learning approaches, and clinicians and patients should be willing to continuously provide images to improve AI applications. Another major obstacle is the difficulty in standardizing image capture due to variations in lighting environments. Standardization is crucial for developing accurate and reliable AI algorithms, but the diverse and dynamic lighting conditions in clinical settings make it challenging to ensure consistent image quality.
Ocular adnexal tumors are prevalent, yet the complexity of these diseases adds another layer of difficulty for researchers. The diversity in tumor types and their morphological variations makes it challenging to establish a standardized dataset for training AI models. Limited image analysis further compounds these challenges, hindering the development and validation of robust algorithms.
In essence, the intricate nature of ocular adnexal tumors, coupled with the difficulty in standardizing image capture conditions, poses significant barriers to conducting comprehensive research in the field of AI and ML for diagnosis in ophthalmology. Addressing these challenges is crucial to unlocking the potential benefits that AI and ML could bring to the diagnosis and management of ocular adnexal tumors.
Anonymity and privacy should be taken seriously when feeding data into AI systems. To ensure the widespread use of AI systems in healthcare, regulatory norms need to be harmonized globally. Additionally, larger studies among ophthalmologists and oculoplastic surgeons are necessary to provide more insights into their perceptions and acceptance of machine learning in the diagnosis of ocular and periocular cancers.
Limitations
Our study, while shedding light on the promising role of AI in oculo-oncology, has certain limitations. First, the scarcity of specialized datasets for oculofacial tumors, and additionally, the research primarily focuses on a nonstandardized review of published literature on ocular adnexal tumors. The evolving nature of AI in healthcare necessitates continuous data input, presenting practical challenges in real-world settings and requiring ongoing clinician and patient engagement. Furthermore, the study’s scope does not delve into the nuanced perceptions and acceptance of machine learning among ophthalmologists and oculoplastic surgeons. While the study marks a step forward in summarizing the comparatively minimal literature, addressing these limitations is crucial for a comprehensive understanding of AI’s potential in advancing oculo-oncology.
Conclusion
In conclusion, the integration of AI and ML in oculo-oncology holds promise for transforming the diagnosis and management of ocular adnexal tumors. While adnexal conditions such as conjunctival melanoma and squamous conjunctival carcinoma pose challenges, recent studies, such as Yoo et al. 33 and Li et al., 42 demonstrate the potential of AI in early detection. Challenges, including the scarcity of specialized datasets and difficulties in standardizing image capture, must be addressed. Patient acceptance, as shown in melanoma diagnosis studies, is encouraging. The future hinges on overcoming obstacles, conducting clinical trials, establishing global regulatory norms, and fostering collaboration among ophthalmologists and AI experts. With these efforts, AI’s transformative impact on ocular and periocular cancer diagnosis appears imminent.
Footnotes
Acknowledgements
We would like to acknowledge the kind contribution of Dr. Asma Ashraf and Dr. Mufliha Ibrahim for their kind contribution in formatting and editing the manuscript.
Author contribution
Qirat Qurban: Manuscript writing, literature review, and conceptualization. Lorraine Cassidy: Proof reading and conceptualization.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Ethics review is not required since it is a review article.
Informed consent
Not required.
