Abstract
Objectives:
Post-COVID-19 vaccine-associated vasculitis stands as one of the most serious side effects attributed to COVID-19 vaccines. This complication encompasses diverse manifestations which vary in presentation and severity. Moreover, it can impact patients across all age groups, with a notably elevated incidence in the elderly. This systematic review seeks to review and evaluate the spectrum of vasculitis manifestations linked to COVID-19 vaccination.
Methods:
A systematic review of the literature was done by searching through PubMed, Google Scholar, and Scopus up to October 2022. Articles including data about sex, age at diagnosis, vasculitis clinical manifestations, type of vaccination, most commonly used investigations, comorbid medical conditions, treatments, and clinical outcomes were included in the final analysis. Furthermore, vasculitis flare-ups post-vaccination were considered part of this review.
Results:
A total number of 117 studies describing 158 patients developing vasculitis following COVID-19 vaccination were included in the final analysis. Among the patients who developed vasculitis, the most administered type of vaccination was the mRNA vaccine subtype (n = 103), followed by the viral vector vaccines (n = 42) and inactivated viral vaccines (n = 10). On the other hand, about 38% of vasculitis-related symptoms occurred after the administration of the first dose of the vaccine and 37% occurred after taking the second dose. The skin (60.7%) and the kidneys (27.8%) were the most affected organs and complete remission was achieved in 111 patients (70%), while partial remission occurred in 11% of the patient population.
Conclusion:
COVID-19 vaccine-induced vasculitis is a rare occurrence associated with COVID-19 vaccines. It generally presents a favorable prognosis and outcomes for the vast majority of patients, ultimately leading to full remission within days. This review emphasizes the notion that the advantages of COVID-19 vaccines outweigh the potential risks, particularly for individuals with compromised immune systems.
Introduction
Vaccination represents a safe and effective strategy for preventing infectious diseases. Typically, vaccine-related adverse events are mild and localized, manifesting as discomfort, swelling, or redness at the injection site due to non-specific inflammation and irritation. 1 Occasionally, systemic reactions leading to constitutional symptoms could occur. 1 The pathophysiology of this condition stems from a type 3 or delayed hypersensitivity reaction causing immune-complex deposition and T-cell activation. 2 Although severe adverse events are infrequently reported, a few vaccine-related vasculitis cases following influenza vaccination have been reported since 1974. 2 This has led to the development of theories linking vaccinations with the development of vasculitis that can range from mild to life-threatening. 2 Interestingly, these adverse events have become more prevalent in the wake of the COVID-19 pandemic. For instance, numerous cases of vasculitis, including giant cell arteritis (GCA), Takayasu arteritis (TAK), polyarteritis nodosa (PAN), and small vessel vasculitis presenting with purpura and vesiculo-bullous lesions, have all been reported following the administration of COVID-19 vaccines.3,4 Furthermore, isolated cutaneous vasculitis and vasculopathy have been associated with COVID-19 vaccines, although the mechanism of pathophysiology is still under investigation. 5 While these conditions exhibit distinct clinical features, they are unified by a shared pathological process characterized by immuno-inflammatory reactions. These reactions predominantly target the tunica media of vascular walls, although they may also affect the tunica adventitia and tunica intima in specific types of vasculitis. 3
While the administration of COVID-19 vaccines has been linked to exacerbating autoimmune conditions in individuals who were already diagnosed, there have been numerous emerging case reports detailing the emergence of new-onset autoimmune disorders following vaccination. 6 Multiple factors, including molecular mimicry, autoantibody production, and vaccine adjuvants, appear to contribute to the development of autoimmune disease following COVID-19 immunization. 6 However, it remains unclear whether these associations represent coincidental findings with a temporal relationship or if they play a causative role in triggering autoimmune reactions. 6
This review article systematically examined and analyzed numerous cases of vasculitis associated with COVID-19 vaccination, with the aim of elucidating the pathogenesis and underlying mechanisms of COVID-19 vaccine-induced vasculitis. It provides a comprehensive assessment of clinical presentations, histological and immunopathological characteristics, specific COVID-19 vaccines administered, diagnostic investigations, coexisting medical conditions, and treatment modalities utilized in each case. Additionally, the review explores instances of preexisting vasculitis exacerbated following COVID-19 vaccination.
Methods
Protocol registration
We followed the guidelines outlined in the Cochrane Handbook of Systematic Reviews and Meta-Analysis and adhered to the recommendations provided in the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. 7 In addition, we followed the guidelines outlined in the summarizing systematic reviews: methodological development, conduct, and reporting of an Umbrella review approach. 8 Our study was registered in the International Prospective Register of Systematic Reviews (PROSPERO) and holds the unique identifying number (UIN): CRD42022361595.
Search strategy
We searched the following databases from inception until October 2022: PubMed, Google Scholar, and Scopus for cases of vasculitis post-COVID-19 vaccine, including all studies published in English. Search terms included subject keywords relevant to the COVID-19 vaccine (e.g., SARS-CoV-2 OR Coronavirus OR COVID-19 OR severe acute respiratory syndrome coronavirus 2) and (vaccination OR vaccine or booster or immunization) and vasculitis (e.g., arteritis OR Vasculitis OR Vasculitides OR Angiitis). The references of the selected studies were checked to ensure the completeness of the search. Articles in languages other than English were excluded from the systematic review.
Eligibility criteria
Our inclusion criteria encompassed case reports, case series, and observational studies. Additionally, reviews and commentaries were only included if they reported original data. All cases with vasculitis post-COVID-19 vaccination including GCA, urticarial vasculitis, TAK, cryoglobulinemic vasculitis, cutaneous PAN, central nervous system (CNS) vasculitis, microscopic polyangiitis, leukocytoclastic vasculitis, Behçet’s disease, Henoch-Schönlein’s purpura, and cutaneous vasculitis were included. We also investigated the exacerbation of vasculitis in individuals who had been previously diagnosed with the condition after receiving the COVID-19 vaccination. Conversely, our exclusion criteria encompassed all studies that involved patients who did not receive the COVID-19 vaccination or cases with strong risk factors for vasculitis besides the vaccine itself. Moreover, articles that were not reported in English or those without confirmed vasculitis using labs, biopsy, or clinical criteria were excluded. All cutaneous vasculitis cases that were not confirmed using skin biopsy were also excluded. Furthermore, it is noteworthy to mention that our inclusion-exclusion criteria had no age, gender, ethnicity, or comorbidity restrictions.
Study selection
Following the database search, titles and abstracts were independently screened by reviewers in Covidence (https://www.covidence.org/), a systematic review production tool. Two reviewers screened each record, and each report was retrieved. Duplicates were removed. Forward and reverse reference searches followed the same procedure. Conflicts were resolved by a third reviewer (senior author).
Data extraction
Data were extracted by six authors using a Microsoft Excel template spreadsheet and reviewed by one author of the team. Each reviewer collected data independently from each report. Conflicts were resolved after a thorough discussion and agreement by reviewers and, when needed, a senior reviewer was consulted to resolve the conflict.
Data extraction included publication details (author names and publication dates), patient demographics (age and gender), vaccine information (type, dosage, timing), symptom onset post-vaccination, vasculitis manifestations, evaluation methods (labs, imaging, biopsy), comorbidities, treatment details (steroids, immunosuppressive drugs), vasculitis type categorization (large vessel, medium vessel, small vessel), investigations (biopsy, direct immunofluorescence, antinuclear antibodies (ANA), ANCA, C-reactive protein, erythrocyte sedimentation rate, rheumatoid factor, kidney function tests, imaging, complete blood counts), and treatment outcomes (recovery status).
Strategy for data synthesis
This was mainly a narrative review; thus, it is not anticipated that sufficient comparable quantitative studies were available to enable a meta-analysis or any other quantitative statistical assessment. If that was the case, this data was extracted to permit synthesis regarding the association between COVID-19 vaccination, vasculitis type, and the outcome of the disease. On the other hand, individual cases were grouped as a cohort in order to perform pilot bivariate and multivariate regression analyses so as to identify the potential association of these vaccinations with the occurrence of vasculitis. Descriptive analysis (range and median) of the extracted data was implemented whenever applicable.
Assessment of risk of bias
Two reviewers independently assessed the risk of bias for observational studies and randomized control trials (RCTs) using the Joanna Briggs Institute’s critical appraisal tools. In any case of disagreement, consensus was reached with a third reviewer. 8
Results
We have identified more than 1382 potentially relevant studies from PubMed, Scopus, and Google Scholar. At first, we removed duplicate studies (n = 196). We screened 1186 articles by title and abstract and excluded 1043 articles. We then reviewed the full text of 143 and found 117 articles that are eligible for the inclusion criteria (Figure 1). The 117 articles reported 158 patients with vasculitis in association with COVID-19 vaccination.1,9–121 These articles included case reports, case series, observational studies, images in medicine, and letters to the editor, commentaries, and editorials that included the presentation of original data. Among the 158 patients, 139 developed vasculitis for the first time, and 19 had relapsing symptoms after a COVID-19 vaccination.
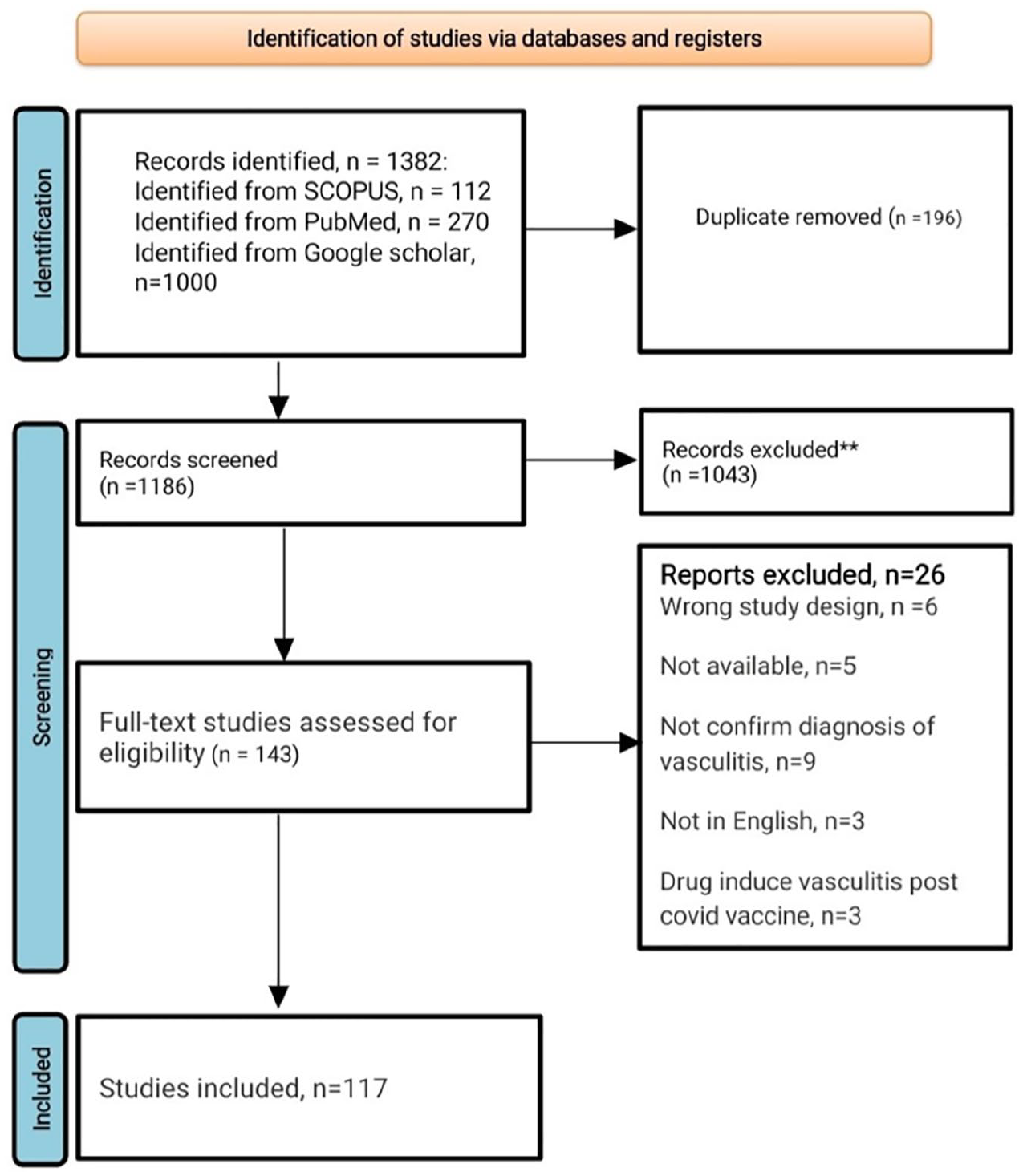
The PRISMA flow diagram of literature screening for patients with vasculitis post-COVID-19 vaccination.
Demographic and clinical characteristics of patients with vasculitis following COVID-19 vaccination
The mean age of the patients with vasculitis following COVID-19 vaccines was 56.7 years, the median age was 60 ± 19.38 years. Females constituted 53.1% of patients while 46.8% were males. Hypertension (was the most common comorbidity affecting 21 patients out of 158 (13.2%), followed by diabetes (6.9%)). Dyslipidemia, hypothyroidism, and asthma were also reported in (6), (5), and (5) of the patients, respectively (Table 1).
Characteristics of patients with vasculitis associated with COVID-19 vaccination in the literature.

Two psoriasis, rheumatoid arthritis, polymyalgia rheumatic, Hashimoto’s thyroiditis, celiac.
bCOVID-19 history, positive COVID-19 PCR or serology.
HTN: hypertension; DM: diabetes mellitus; AAV: (ANCA)-associated vasculitides; LCV: leukocytoclastic vasculitis; HSP: Henoch-Schonlein purpura; GPA: granulomatosis with polyangiitis; MPA: microscopic polyangiitis.
Furthermore, 19 patients (12%) were diagnosed with vasculitis prior to vaccination and had reported flare-ups of their underlying disease after the vaccination. Interestingly, most of this subgroup had cryoglobulinemic vasculitis (8). Other patients had ANCA-associated vasculitis (2), leukocytoclastic vasculitis (1), Behcet’s (2), Henoch-Schonlein purpura (2), granulomatosis with polyangiitis (1), and microscopic polyangiitis (3). Other autoimmune diseases were identified prior to the vaccination in eight patients including Sjogren’s syndrome, psoriasis, rheumatoid arthritis, polymyalgia rheumatica, Hashimoto’s thyroiditis, and celiac disease (Table 1).
Isolated cutaneous vasculitis was the most common type of vasculitis that affected the patients following COVID-19 vaccination (36%), while the second most common type was ANCA-associated vasculitis (21.5%) (Figure 2). Other types of vasculitis, including cryoglobulinemic vasculitis, GCA, urticarial vasculitis, and IgA vasculitis, had an incidence rate of 6.3%, 9.4%, 3.7%, and 15% of the cases, respectively (Table 2).

A column chart demonstrating the number of vasculitis cases related to COVID-19 vaccinations, with categorization of the cases by the type of vasculitis developed and the specific vaccine administered.
vasculitis manifestations following COVID-19 vaccination (n = number of cases).

Unclassified vasculitis.
Renal arteritis, immune-complex vasculitis.
IgAV: IgA vasculitis; HSP: Henoch-Schonlein purpura; AAV: (ANCA)-associated vasculitides; LCV: leukocytoclastic vasculitis; LVV: large vessel vasculitis; SVV: small vessel vasculitis; MVV: medium-vessel vasculitis; CV: cryoglobulinemic vasculitis; CNS: Central nervous system; GPA: granulomatosis with polyangiitis; MPA: microscopic polyangiitis.
An overview of the vaccine types and associated symptomology and vasculitis
Among the 57 cases of skin vasculitis including 6 cases of urticarial vasculitis, 29 occurred after the mRNA vaccine, 19 after the viral vector vaccine, and 8 after inactivated vaccines (Table 2). IgA vasculitis was seen in 17 cases after mRNA vaccines and 7 after viral vector vaccines. ANCA-associated vasculitis, which includes granulomatosis with polyangiitis and microscopic polyangiitis, appeared in 34 cases, with 24 linked to mRNA vaccines, 8 to viral vector vaccines, and 2 to inactivated vaccines. GCA was reported in 15 patients, primarily after the administration of mRNA vaccines (9 patients). Cryoglobulinemic vasculitis, Behcet’s disease, TAK, and Kawasaki disease occurred after mRNA vaccines in (7/10), (3/3), (1/2), and (1/1) of patients, respectively (Table 2).
Among the different COVID-19 vaccines, the Pfizer-BioNTech mRNA vaccine (BNT162b2) had the highest association with vasculitis, accounting for 70.8% of cases. The Moderna mRNA vaccine (mRNA1273) was linked to vasculitis in 25.2% of cases. Viral vector vaccines, specifically Oxford-AstraZeneca and Johnson & Johnson, were responsible for 26.5% of vasculitis cases (Figure 3). Inactivated viral vaccines like Sinovac, Sinopharm, and COVAXIN-BBV152 were the least associated with vasculitis, with only a few cases reported as shown in Table 3.

A column chart demonstrating the percentage of COVID-19 vaccine-related vasculitis as it relates to different vaccine types.
Vasculitis manifestations following COVID-19 vaccination (n = number of cases).

Vasculitis symptoms occurred after the first vaccine dose in 38.6% of cases, after the second dose in 37.3%, after both doses in 2.5%, and after a third dose in 4.4%. On average, symptoms appeared about 7 days after the vaccine. Skin manifestations were the most commonly reported symptoms (60.7%), followed by renal dysfunction (27.8%), joint pain (12.9%), respiratory dysfunction (10.1%), musculoskeletal pain (9.4%), and gastrointestinal issues (8.8%). General symptoms such as fever (19.6%), fatigue (13.2%), and headache (9.4%) were also reported. Investigations used for the diagnosis of vasculitis included laboratory tests, imaging tests, and biopsy of the involved organ. Elevated inflammatory markers were found in 43.6% of cases, while positive ANCA tests were observed in 18.9%. Imaging studies showed signs of vasculitis in 27.2% of patients, and biopsy confirmed vasculitis in 67.7% of cases (Table 4). A summary of the recorded clinical manifestations, demographics, and characteristics of patients with vasculitis after COVID-19 vaccination is described in detail in Table 4.
The features of all patients with vasculitis associated with COVID-19 vaccination in the literature.

GIT: gastrointestinal tract; IVIG: intravenous immunoglobulin; PR: partial response; CR: complete response; NSAID: non-steroidal anti-inflammatory drugs.
Treatment approaches and outcomes in vasculitis following COVID-19 vaccination
The most common therapeutic modality used for vasculitis after COVID-19 vaccination was corticosteroids, which were utilized in 78.4% of cases. Other immunosuppressive therapies such as rituximab and cyclophosphamide were used in 9.4% of cases each, azathioprine in 3.7%, and intravenous immunoglobulin in 12.6%. Plasmapheresis on the other hand was performed in nine patients, and non-steroidal anti-inflammatory drugs (NSAIDs) were administered in 5.6% of cases. The outcomes varied, with 70.2% experiencing a complete response to treatment, 11.3% having a partial response, and 3.7% demonstrating no significant clinical response. On average, the duration of remission was about 14 days (Table 4). Most patients (56 cases) had remission lasting less than 2 weeks, while only 4 patients experienced remission lasting more than 3 months (Figure 4).

A column chart demonstrating the number of patients and the duration of remission in cases with COVID-19 vaccine-related vasculitis.
Discussion
Vasculitis encompasses a diverse group of disorders, characterized by inflammation in blood vessels. It can be triggered by various factors such as infections, drugs, connective tissue disorders, malignancy, and systemic inflammatory diseases. Nevertheless, in certain cases, vasculitis may exhibit an idiopathic nature, where despite thorough investigation, no precise underlying cause can be identified. 122 Recent studies have proposed an association between COVID-19 vaccination and various types of vasculitis. 122 Due to the widespread administration of COVID-19 vaccines throughout the pandemic, several side effects have been reported, including autoimmune conditions like inflammatory arthritis, skin rashes, and alopecia. This necessitates the need to further investigate the association between COVID-19 vaccines and autoimmunity. 19
The correlation between vaccinations and vasculitis extends beyond COVID-19 vaccines. In fact, documented cases of vasculitis associated with vaccinations can be found in the medical literature for a range of vaccines, including Bacillus Calmette-Guerin (BCG), diphtheria, pertussis, and tetanus (DPT) vaccination, hepatitis A and hepatitis B vaccination, meningococcal vaccination, as well as measles, mumps, and rubella (MMR) vaccination. 123
There have been reports of vasculitis following COVID-19 vaccination with various cutaneous manifestations. 54 Interestingly, some vaccines were associated with urticarial vasculitis eruptions, which lack a true leukocytoclastic vascular inflammation in a proportion of these cases. 124 According to a recent study, the percentage of cutaneous vasculitis out of the total of cutaneous reactions after the Pfizer and Moderna COVID-19 vaccines were 2.9% and 0.7% respectively, occurring within approximately 2 weeks after the initial doses of the Moderna (mRNA-1273) and Pfizer (New York, NY, USA)-BioNTech (Mainz, Germany) (BNT162b2) vaccines. 125 While there have been hypotheses suggesting an autoimmune mechanism triggered by vaccine proteins, the exact pathophysiology of vasculitis following vaccination remains unknown. 126 Some theories propose that both SARS-CoV-2 infection and mRNA vaccines might potentially induce autoimmunity, possibly through cross-reactivity between vaccine components and self-antigens. 123
We found that most patients with vasculitis following COVID-19 vaccines were elderly, with a median age of 57 years. Both male and female patients were affected similarly, with a female-to-male ratio of 1.1:1. The predominant type of vasculitis observed was cutaneous vasculitis, with over half of the patients presenting with skin manifestations. Additionally, 27% of patients had renal disorders. While other organs, such as the lungs, joints, muscles, and gastrointestinal tract, were also involved, they were affected to a lesser extent. Many patients experienced various constitutional symptoms, including fever, fatigue, and weakness. Notably, our patients also had other autoimmune disorders, such as diabetes mellitus, hypothyroidism, asthma, and Sjogren’s syndrome. This underscores the genetic susceptibility of these individuals to autoimmune conditions, which could potentially be triggered by vaccinations.
Interestingly, our findings revealed that most vasculitis cases occurred following exposure to mRNA vaccines, accounting for 65% of the cases. Specifically, Pfizer and Moderna vaccines were predominantly associated with these incidents. This could be attributed to factors related to the composition of these vaccines or, alternatively, the high prevalence of their use in the population. The development of vasculitis after SARS-CoV-2 vaccine administration occurred, on average, around 7 days following vaccination. In over 40% of the cases, patients exhibited elevated inflammatory markers, while around 30% of the patients tested positive for ANCA.
The management of COVID-19 vaccine-associated vasculitis aligned with established practices for vasculitis management in general. The outcomes were favorable, with over 70% of patients achieving a complete response, and a minimal 3% treatment failure rate. In our cohort of 158 patients with vasculitis, roughly 80% of individuals received corticosteroid therapy. The majority of these patients experienced a successful resolution of symptoms, typically within a remission period lasting around 14 days (Table 3). Multiple treatment approaches were utilized, including NSAIDs, rituximab, cyclophosphamide, antihistamines, and plasmapheresis. Notably, corticosteroids proved to be the most effective among these options. Similar to other cases of vasculitis, the choice of treatment is contingent upon the specific disease pattern and requires a thorough evaluation of patients exhibiting active vasculitis.
COVID-19 has resulted in a global mortality toll exceeding 6 million fatalities, often attributed to an aberrant or overactive host immune reaction. 127 Consequently, the benefits of vaccination outweigh the associated risks, albeit occasionally complicated by rare immune-mediated adverse events. These events may arise as a result of proinflammatory stimulation, revealing an individual’s susceptibility to developing maladaptive immune responses. 128 Given the rising incidence of autoimmune-related disorders following COVID-19 vaccination, there is an urgent need for the prompt publication of post-marketing surveillance findings for these vaccines.
This article carries certain limitations that warrant consideration. Firstly, most case reports lacked critical information pertaining to the patient’s medical histories and serological tests related to prior COVID-19 infection. Additionally, our study’s sample size remains relatively small when juxtaposed with the total number of individuals who have received COVID-19 vaccinations. Furthermore, it is important to acknowledge that some vasculitis cases may have gone unreported. Therefore, further investigations are imperative to comprehensively explore the diverse manifestations and prevalence of this disease post-COVID-19 vaccination. Lastly, we lacked the capacity to conduct comprehensive assessments of vasculitis prevalence following different vaccines on a large scale.
Conclusion
In summary, vasculitis cases post-COVID-19 vaccination manifest in various forms and presentations. Awareness of this issue is crucial for both early detection and effective management in individuals with both new-onset and preexisting vasculitis. Fortunately, most patients experience a positive prognosis. Nevertheless, additional studies are warranted to gain a deeper understanding of the specific risk factors associated with vasculitis development or exacerbation following COVID-19 vaccinations.
Footnotes
Acknowledgements
None.
Author contributions
A.A., M.A., and M.A. contributed to design of the study, data analysis, data interpretation, and drafting of the manuscript. E.R., B.Q., S.H., Y.A.T., Z.S., O.D., and A.W.M.J. contributed to design of the study, data collection data extraction, and data interpretation. L.A. contributed to design of the study, data interpretation, and supervision of the work. All authors have read and approved the final manuscript. Each author has participated sufficiently in the work to take public responsibility for the content.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
This review article did not require any ethical approval from any research committee.
Informed consent
This review article did not require informed consent since it did not involve human or animal subjects.
Guarantor
All authors have read and approved the manuscript; on behalf of all the contributors, I will act as guarantor and will correspond with the journal from this point onward.
