Abstract
We describe the case of a 61-year-old male patient with a history of hemophilia A and previous hepatitis C virus infection with sustained virological response and no previous documentation of cirrhosis, who was admitted for variceal bleeding. He was taken for endoscopic evaluation with evidence of active variceal hemorrhage requiring rubber band ligation. Patients with congenital coagulation disorders, such as hemophilia A, are excluded from international guidelines for gastrointestinal bleeding, making their management and counseling challenging. In this article, we describe the specific interventions to be performed in patients with hemophilia A and upper gastrointestinal tract bleeding, specifically variceal bleeding, focusing on pre-endoscopic and endoscopic management.
Introduction
Upper gastrointestinal tract bleeding (UGB) is defined as bleeding of gastrointestinal origin involving the esophagus, stomach, or duodenum; commonly delimited to that originating above the angle of Treitz. 1 The clinical manifestations are very diverse, including visible forms of bleeding such as hematemesis, coffee-ground emesis, melena, or in some cases hematochezia. 2
Upper gastrointestinal bleeding remains the leading cause of gastroenterology hospitalization, accounting for more than 500,000 admissions annually in the United States. 3 In Colombia, the annual incidence has been reported to be between 50 and 150 cases per 100,000 inhabitants. 4
The etiology of non-variceal UGB has been extensively studied, the most common being secondary to peptic ulcer (30%–50%), Mallory-Weiss tear (15%–20%), erosive gastritis or duodenitis (10%–15%), esophagitis (5%–10%), gastric neoplasia (5%), gastrointestinal angiodysplasia (5%), and other miscellaneous causes (5%).1,5 Variceal hemorrhage is commonly differentiated from non-variceal hemorrhage by its pathophysiology linked to portal hypertension. Mortality due to upper gastrointestinal bleeding has remained stable between 5% and 10% in the last 20 years3,6 which has been a cause of high social and economic repercussions.
Congenital hemophilia A is an inherited genetic disorder caused by a decrease in the amount or activity of clotting factor VIII, which globally predisposes to bleeding manifestations. 7 Clinical practice guidelines for the management of UGB (Japanese, European, and North American1,7–9 among others) do not include patients with hemophilia, and information on the management of this entity in patients with coagulation disorders of this type is limited. This poses a therapeutic challenge for multiple medical specialties and can cause great anxiety for both the medical staff and the patient.
This article presents the case of a patient with a history of congenital hemophilia A and hepatitis C virus (HCV) infection who presented with variceal UGB as the first manifestation of previously undiagnosed cirrhosis.
Case report
A 61-year-old male from Bogotá D.C., Colombia, with a history of mild congenital hemophilia A, systemic arterial hypertension, and type 2 diabetes managed with enalapril 20 mg once a day (QD), amlodipine 20 mg QD, and insulin glargine 22 IU QD, presented with three episodes of hematemesis. Diagnosed with HCV infection in 1998, he underwent successful treatment with pegylated interferon and ribavirin.
The patient was drowsy upon admission. Admitted for life-threatening bleeding, the patient responded well to crystalloid management, maintaining a mean arterial pressure of >65 mmHg. Initial complete blood count (CBC) showed hemoglobin at 8.2 g/dL (baseline hemoglobin 11.3 g/dL), platelets at 149.6 × 103/uL, creatinine at 0.71 mg/dL, and factor VIII activity at 28.2%. Albumin value was 2.6 g/dL, PT and PTT were 14.7/11.6 s and 43.7/28.8 s, and total bilirubin was 1.65 mg/dL. Hematology administered 4500 IU of recombinant factor VIII before endoscopy, revealing four variceal bundles larger than 5 mm. Elastic band ligation achieved bleeding control (Figure 1(a) and 1(b)). The patient received terlipressin and ceftriaxone prophylaxis before endoscopic procedures. Esomeprazole was also administered at an infusion rate of 8 mg/h.
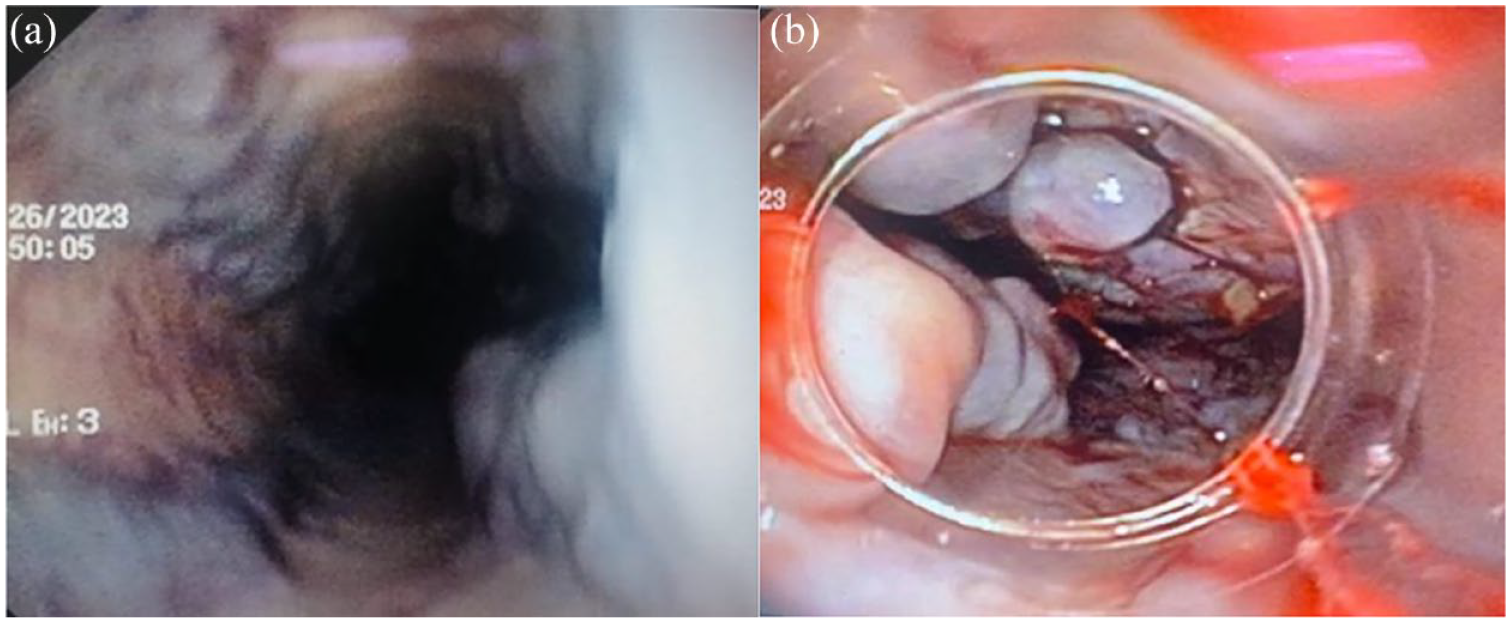
(a) Four large variceal bundles (diameter greater than 5 mm) with red signs. (b) Large variceal bundles with stigmata of recent bleeding. View through the cap of the ligating equipment used on the gastroscope.
Extension studies confirmed previously undocumented Child-Pugh B cirrhosis attributed to past HCV infection. The patient’s clinical condition improved, and he was discharged with outpatient propranolol management, along with hepatology and hematology follow-up.
The patient had variceal UGB recurrence despite secondary prevention, so he became a transjugular intrahepatic portosystemic shunt (TIPS) candidate and was listed for liver transplant.
Discussion
UGB is a significant reason for hospital admissions, with variable presentations that can range from self-limiting to potentially life-threatening. Mortality associated with severe cases is concerning. Initial management involves risk assessment using tools like the Glasgow-Blatchford scale (GBS). 10 A GBS score of 0–1 suggests low clinical risk,1,11 while a score ⩾2 necessitates hospital admission. Treatment focuses on stabilizing the patient’s cardiovascular status, transfusing red blood cells if necessary, and performing endoscopy promptly, within 24 h for non-variceal bleeds and 6–12 h for variceal bleeds post-stabilization. 8
The above clinical practice guideline recommendations arise from studies in which patients with a diagnosis of hemophilia were not included, making the approach challenging due to the lack of available evidence. The risk of gastrointestinal bleeding has been related to the severity of hemophilia, being higher in patients with severe hemophilia A (serum factor VIII levels <1%), compared to moderate (levels 1%–5%) and mild (levels >5%). 7 There are no recent studies evaluating the etiology of non-variceal UGB on a large scale, but the available information confirms that peptic ulcer and acute erosive gastroduodenitis are the most frequent causes of bleeding in this group of patients.7,12
Variceal bleeding is of high relevance considering the relationship of hepatotropic virus infection and blood product transfusions prior to the establishment of screening programs and the development of factor VIII replacement therapies, which left patients with hemophilia at increased risk of hepatitis B and C virus infection, as well as the development of cirrhosis, portal hypertension, and subsequent variceal bleeding. 13 It is important to note that congenital hemophilia can cause spontaneous bleeding; however, UGB in patients with portal hypertension is likely from esophageal varices. Case management is challenging as congenital hemophilia A can worsen the bleeding, making it more difficult to treat.
Helicobacter pylori infection and the use of nonsteroidal anti-inflammatory drugs (NSAIDs) are independent risk factors for the development of upper gastrointestinal bleeding in patients with hemophilia. 14 The use of COX-2 selective NSAIDs is recommended if their use is absolutely necessary. The occurrence of spontaneous posterior peritoneal hematoma has been identified as a risk factor associated with UGB in patients with hemophilia. 13 Gastrointestinal bleeding in patients with hemophilia A is considered life-threatening bleeding, so its suspicion necessarily requires in-hospital evaluation.
GBS does not include hemophilia as a variable, so it should not be used to define admission in a patient with hemophilia and UGB. There is no specific information on pre-endoscopic management in this type of patient, so it is suggested to follow the same recommendations as in other patients.
In patients with hemophilia, additional considerations should be taken into account regarding the safety of endoscopic procedures since they could lacerate the mucosa and predispose to increased bleeding. In patients with mild hemophilia A, the use of 0.3 mcg/kg IV or SC desmopressin over a period of 15–30 min, 30 min prior to endoscopy is indicated. 7 Its use is recommended in patients who already have a documented favorable response to this drug. Prophylaxis for patients with moderate and severe hemophilia A consists of administration of recombinant factor VIII 1–3 h prior to endoscopy, with a target factor level of 0.8–1 IU/mL. 15 This is based on expert recommendation and studies have suggested that the risk of bleeding in elective endoscopic procedures in patients with congenital coagulation disorders is not significantly higher. Concern about the safety of endoscopic procedures and risk of continuous bleeding from mucosal damage site in band ligation or injury site caused by sclerotherapy needle has been addressed, and findings are summarized in Table 1.
Studies addressing the safety and outcomes of endoscopic hemostasis methods for variceal bleeding in patients with hemophilia.

There is no information to suggest that endoscopic treatment is different in patients with hemophilia A. Endoscopic treatment should be implemented when high-risk findings are found: ulcers with active bleeding and visible vessels. Regarding variceal bleeding, endoscopic treatment should follow the same guidelines as in patients without hemophilia A, with banding ligation as the first-line therapy. 8 Endoscopic sclerotherapy has also been reported to be a safe and effective procedure in patients with variceal bleeding due to cirrhosis and hemophilia A. 16 Rebleeding rates can be as high as 22% in patients with hemophilia, despite endoscopic management. 13
In the literature, there are descriptions of the use of TIPS as rescue therapy generating an adequate decrease in the portal hypertensive gradient. 17 In Figure 2, we propose an algorithm for the approach and management of patients with suspected variceal UGB and a history of hemophilia.

Algorithm for the approach and treatment of patients with hemophilia and UGB. ICU: intensive care unit; PPI: proton pump inhibitor; TIPS: transjugular intrahepatic portosystemic shunt; UGB: Upper gastrointestinal tract bleeding; UGIE: upper gastrointestinal endoscopy.
Conclusion
Hemophilia A is a congenital coagulation disorder that predisposes to gastrointestinal bleeding, which is considered life-threatening bleeding. The guidelines do not contemplate this type of patient within their recommendations, but the available evidence suggests that pre-endoscopic and endoscopic treatment is the same as in patients without hemophilia, both in non-variceal and variceal bleeding. Experts recommend that patients should receive prophylaxis with desmopressin or recombinant factor VIII, depending on the severity of hemophilia A, prior to endoscopy. 19
Footnotes
Acknowledgements
None.
Author contributions
The authors contributed equally to the elaboration of this article.
Data availability
Additional data can be requested from the authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval to report this case was obtained from the Ethics Committee of the HUSI and the Pontificia Universidad Javeriana (032-2023).
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
