Abstract
Background:
Platelet count and bleeding risk are frequently used in clinical assessments of the severity of immune thrombocytopenia. On the other hand, immune thrombocytopenia also influences patients’ health-related quality of life. The immune thrombocytopenia life quality index is a new tool for evaluating health-related quality of life in immune thrombocytopenia patients.
Objective:
To assess the psychometric properties of the Amharic version of the immune thrombocytopenia life quality index tool for immune thrombocytopenia patients in Ethiopia.
Methods:
The facility-based cross-sectional study was carried out from 15 September to 30 October 2022. The school of pharmacy institutional ethics review board provided ethical clearance and approval of the study protocol (approval number: ERB/SOP/487/14/2022). Both written informed consent and family or legal guardian consent were obtained for participants aged 14–18 years and informed consent for participants aged >18 years. The tool was forward-backward translated before being pretested on five immune thrombocytopenia patients. Descriptive statistics were used to report sociodemographic and clinical data. The psychometric properties of the immune thrombocytopenia life quality index in Amharic were evaluated for acceptability, reliability, and construct validity.
Results:
This study included 100 participants, at the age of diagnosis of immune thrombocytopenia, the median with inter-quartile range of platelet count (×109/L) was 15.00 (18.00). The majority of study participants (79, 79%) received prednisolone alone as first-line therapy. The tool psychometric test was reliable; the correlation coefficient between items ranged from 0.147 to 0.956, and the overall Cronbach’s alpha coefficient was 0.956. Furthermore, the tool was construct-valid, with factor analysis between components revealing that Item-1 had an eigenvalue greater than one and explained 72.532% of the total variance among all 10 immune thrombocytopenia life quality index domains. The immune thrombocytopenia life quality index total (alternative score) had a strong (r = 0.9) correlation with Item-8 and a moderate (r = 0.317) correlation with Item-5.
Conclusion:
The Amharic version of the immune thrombocytopenia life quality index tool is a valid and reliable tool that can be used to assess the health-related quality of life of immune thrombocytopenia patients in Ethiopia.
Introduction
Immune thrombocytopenia (ITP) is a bleeding disorder caused by autoantibody-mediated and cell-mediated destruction of platelets, leading to accelerated platelet clearance and impaired thrombopoiesis.1 –3 It is described by a transient or persistent reduction in platelet count of 100 × 109/L and an increased risk of bleeding.4,5 According to the ITP world impact survey, the most common clinical presentations at diagnosis were petechiae (64%) and bruising of unknown origin (65%). Anxiety about maintaining a stable platelet count (34%), as well as fatigue, were common signs and symptoms that patients wanted to be resolved. 6 These signs and symptoms, such as fatigue, headache, anxiety, and fear of bleeding had an impact on health-related quality of life (HRQoL). 7
HRQoL is a patient-reported outcome that is typically measured using tools that have been carefully developed and validated. 8 Patient-reported outcome measures allow researchers to identify and quantify the impact of a disease or its treatments on different HRQoL domains, as well as capture patients’ perspectives on treatment effectiveness by taking a holistic approach. Platelet count and bleeding risk are frequently used in clinical assessments of ITP severity. On the other hand, ITP is known to have an impact on patients’ HRQoL and fatigue.6,9,10 Clinicians and researchers are increasingly recognizing HRQoL as an important tool for assessing the efficacy of medical interventions, and they have discovered morbidity in ITP patients, such as fatigue, fear of bleeding, and a negative impact on the role, social, and occupational activities. 11
HRQoL impairment in ITP patients is influenced by actual bleeding, fatigue, decreased energy, depression, and treatment side effects, which may lead to limitations in patients’ lifestyles.12,13 Patient-reported outcomes in ITP are important for understanding and guiding treatment by changing treatment modalities such as medications, medical care, patient education, or support services to improve patient outcomes. 14 Several studies have used various tools to assess the HRQoL of ITP patients,7,13,15 –17 of which the ITP life quality index (ILQI) is the most recently developed tool for assessing HRQoL in ITP patients. The ILQI is one of the valid and reliable tools for assessing disease-specific outcomes in adult ITP patients who have been diagnosed and treated. It is a 10-item measure of patient-reported outcomes that was developed to support patient–physician discussions and check treatment guidelines. It is used in clinical practice as a tool to assess changes in HRQoL over time. The psychometric validity of the ILQI was evaluated in 1507 adult ITP patients and 472 healthcare providers from 13 countries using ITP world impact survey data indicating patients’ and physicians’ perceptions of ITP diagnosis, signs and symptoms, and treatment. 18 Evidence from the United Kingdom supports the ILQI’s content validity and confirms that the ILQI concepts are relevant to patients, as well as that the ILQI items were consistently understood by patients. 19 For the Japanese and French populations, the tool has also been translated and linguistically validated. 20
However, there is no translated and linguistically validated ITP-specific assessment tool in Amharic (the national language of Ethiopia) to assess the HRQoL of ITP patients in Ethiopia. Variations in interpretations of quality of life (QoL) in patient-reported outcomes may result from cultural differences between countries. As a result, it is critical to ensure that the tool is psychometrically sound before using it in Ethiopia. To adapt the inventory for use in Ethiopian ITP patients, it is important to evaluate its acceptability, reliability, and validity, as well as its suitability for Ethiopian ITP patients. Hence, the purpose of the current study was to determine the acceptability, reliability, and validity of the ITP disease-specific ILQI tool for Ethiopian ITP patients.
Materials and methods
Study setting
This study was conducted at Tikur Anbessa Specialized Hospital (TASH), which is the largest teaching hospital affiliated with the College of Health Sciences at Addis Ababa University and serves as a training center for medical, pharmacy, and other health sciences students. It is also a facility where specialized comprehensive and clinical services are provided. According to the health management information system data of the hospital, the hematology clinic had an average of 110 ITP patients visiting the hematology clinic per month.
Source and study populations
All patients treated for ITP at the hematology clinic of TASH were the source of the population whereas the study population was all patients who fulfil the eligibility criteria.
Study design and period
The facility-based cross-sectional study was carried out from 15 September to 30 October 2022.
Eligibility criteria
Eligible patients include (i) all patients attending a hospital during the study period who have a confirmed diagnosis of ITP according to the guidelines of the American Society of Hematology of 2019, 21 and the standardization of terminology, definitions, and outcome criteria in ITP of adults and children (primary, secondary, newly diagnosed, persistent, chronic, and severe ITP). 4 (ii) Age ⩾14 years. (iii) Patients who were willing to participate in the study were included. Patients who had incomplete medical records were excluded from the study.
Sample size determination and sampling technique
The sample size was determined using the item-to-subject ratio based on the statistical aspect of psychometric tool validation. Because the ILQI contains ten items, and we used a 1:10 ratio, the sample size for our study was 100. The literature recommends minimum sample sizes between 3 and 20 times the number of variables, with absolute values ranging from 100 to over 1000.22 –24 A consecutive sampling technique was used to recruit study participants from the TASH hematology clinic.
Data collection and management
Data abstraction format
The data collection format was developed and used to collect information from the medical record or directly from the patient, such as sociodemographic, clinical and pathologic, treatment-related, and follow-up-related characteristics.
ITP life quality index
The ILQI was created by clinical experts in the field of ITP and was originally thought to be unidimensional, producing a single score. 18 This is a 10-item patient questionnaire with a recall period of “last month.” Responses range from “never” to “always.” Three questions (questions 1, 2, and 5) have additional response options that allow the patient to indicate that the question is irrelevant to them or that they do not wish to answer. For the additional responses, “I am not currently working/studying due to ITP” the value 4 applies, and for the responses “I am not currently working/studying due to other reasons or does not apply/prefer not to say” the value 0 applies. A total sum score between 7 and 40 was originally proposed, and the content and psychometric validity of the ILQI was assessed in 13 countries. 18
Data quality assurance
This ILQI questionnaire was first translated from the original English version into Amharic by a bilingual person fluent in Amharic and English. A second bilingual translator, who did not know the original English version, back-translated the tool from the Amharic version into English. The two English translation versions were then compared for equivalence. Any discrepancies in the translation were resolved in a discussion. A pre-test was then administered to 5% of ITP patients at St. Poulos Hospital Millennium Medical College. The purpose of the pre-test was to ensure that respondents understand the questions and can review the wording, logic, and skip order in a way that makes sense to respondents. Based on the result of the pre-test, all necessary changes and adjustments were made before the actual study is conducted. To ensure the completeness and consistency of data, the study director reviews all data daily.
Statistical analysis
SPSS version 26 was used to enter and analyze data. To report demographic and clinical characteristics, descriptive statistics (frequency, percentage, median with inter-quartile range (IQR)) were used. Because the data were not normally distributed, the median with IQR was used to describe patients’ platelet count, white blood cell count (WBC), hemoglobin, age at diagnosis, durations of ITP since diagnosis, and time of ITP relapse. Acceptability, reliability (internal correlation coefficient, average overall item correlation, and Cronbach’s alpha), and construct validity (factor loading, factor analysis, Kaiser-Meyer-Olkin (KMO), Bartlett’s test for sphericity, and scree plot) of the Amharic version of the ILQI were evaluated.
Translation of the English version of the ILQI tool into Amharic for use in Ethiopian ITP patients
Forward translation of the ILQI into Amharic language and its back translation was performed by a bilingual person fluent in Amharic and English, followed by a pre-test on ITP patients. The workflow of the translation process is shown in Table 1 below.
The workflow of the translation process of the ILQI.

The ILQI was separated into individual items and response options for translation into the Amharic version. Most of the items in the ILQI were reconciled with no revision in the forward and backward translations. A pre-test was done on five ITP-diagnosed patients following forward and backward translations.
Acceptability: Acceptability of the ILQI was determined by response rate, percentage of missing data, time spent completing the questionnaire, and data on questions considered troubling, confusing, or difficult to answer.
Reliability: Cronbach’s alpha coefficient was calculated to assess the reliability of the ILQI tool, which is a measure of the internal consistency of responses and the degree to which the results of individual items are correlated with the results of all other items. This coefficient has a value between 0 and 1, with higher values indicating higher reliability. A correlation between items of 0.45 to 0.98 is considered acceptable, and 0.7 indicates adequate scale reliability of the tool. 25 The inter-item correlation coefficient was also used to investigate the extent to which scores on one item are related to scores on all other items on a scale. 26
Validity: Construct validity refers to the extent to which evidence and theories support interpretations of test results that arise from the proposed uses of tests. Exploratory factor analysis (EFA) and Pearson correlation tests were conducted to test construct validity.27 –29 Factor loading, factor analysis, the KMO measure of sampling adequacy, 30 Bartlett’s test for sphericity, 31 and the scree plot were used to assess the validity of the components of the Amharic version of the ILQI.
Results
Tests of normality for of ITP tool QoL index
The normality test of the ILQI score was performed by using the Kolmogorov-Smirnov test. 32 The tests indicated that the data were not normally distributed (p < 0.0001), Table 2. The overall ILQI tool responses distribution skewness value was 0.708 and the kurtosis was −0.589, with the skewness and kurtosis to their standard errors being 0.241 and 0.478, respectively. To satisfy the normality of the data different transformation methods like log 10 and square root transformation was used and the data were also not normally distributed.
Tests of normality of the ILQI in patients with ITP attending the hematology clinic of TASH, Addis Ababa, Ethiopia 2022 (n = 100).

Lilliefors significance correction.
Socio-demographic characteristics of ITP patients
This study included 100 participants, with the majority (80, 80%) of them female. In terms of age distribution, the average age of study participants was 30 years old, ranging from 15 to 88 years old, with the majority (34, 34%) of participants being between the ages of 25 and 34. Married (71, 71%) and Orthodox Christians (57, 57%) made up the largest proportion. Most participants (95, 95%) lived with their families, and half (51, 51%) resided in Addis Ababa. In addition, half of the healthcare costs of the study participants (50, 50%) were covered by the patients themselves or by their relatives (Table 3).
Sociodemographic data in patients with immune thrombocytopenia attending the hematology clinic at TASH in Addis Ababa, Ethiopia, 2022 (n = 100).

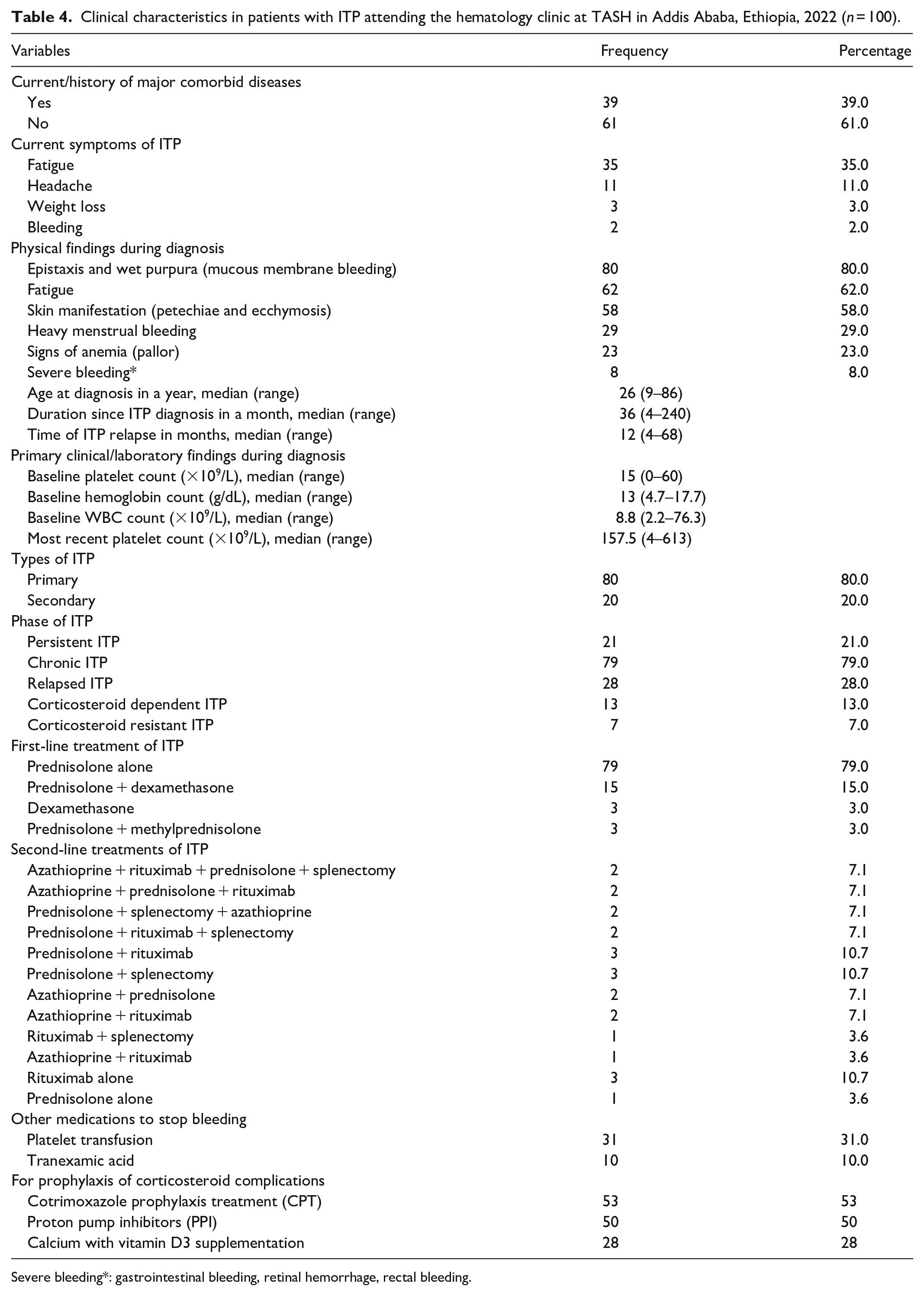
Clinical characteristics of ITP patients during diagnosis
Clinical characteristics and treatment history are shown in Table 4. Of the 100 study participants, only (39, 39%) had previous or current comorbidities (Figure 1). They had fatigue during the study (39, 39%) and during the diagnosis of ITP (62, 62%). Clinical presentations of study participants during diagnosis were only (80, 80%) had epistaxis and wet purpura (mucosal bleeding).
Clinical characteristics in patients with ITP attending the hematology clinic at TASH in Addis Ababa, Ethiopia, 2022 (n = 100).
Severe bleeding*: gastrointestinal bleeding, retinal hemorrhage, rectal bleeding.

Comorbidities in patients with ITP attending the hematology clinic at TASH in Addis Ababa, Ethiopia, 2022 (n = 100). Others*: Chronis disease, Stroke, Lupus nephritis, Heart failure, Dyslipidemia, Epilepsy, Gout arteritis.
The median value of primary clinical/laboratory findings at diagnoses, such as platelet count, hemoglobin, and WBC count, was 15 × 109/L ranging from 0 × 109/L to 60 × 109/L, 13 g/dL ranging from 4.7 to 17.7 g/dL, and 8.8 × 109/L ranging from 2.2 × 109/L to 76.3 × 109/L respectively. The median age of study participants at ITP diagnosis was 26 years, ranging from 9 to 86 years; the median duration of ITP since diagnosis was 36 months, ranging from 4 to 240 months; and the median duration of ITP relapse was 12 months, ranging from 2 to 68 months.
The majority of study participants (79, 79%) received prednisolone alone as first-line therapy, and azathioprine, rituximab, and prednisolone were the most commonly prescribed drugs for second-line therapy; in addition, 14 patients underwent splenectomy, and two other patients were awaiting surgery for ITP treatment.
About (31, 31%) of the study participants received platelet transfusions to prevent bleeding and (10, 10%) took tranexamic acid to stop bleeding. In addition, approximately (53, 53%), (50, 50%), and (28, 28%) of study participants took cotrimoxazole prophylaxis, proton pump inhibitors, and calcium with vitamin D3 as prophylaxis to prevent immunodeficiency-related infections, peptic ulcers, and osteoporosis, respectively.
Acceptability of ITP tool QoL index
Acceptability was tested in terms of overall compliance for each of the items of the ILQI tool. Overall, 100 patients participated and all of the 10 items of the questionnaire were completed within 5–10 min.
Reliability analysis for the ITP tool QoL index
The internal correlation coefficient, average item overall correlation, and Cronbach’s alpha were evaluated to determine whether the ILQI tool is reliable.
Internal consistency of the ILQI
The inter-item correlation coefficient between items ranged from 0.147 to 0.956, with 0.552 being the average (Table 5). This value was greater than the ideal range of average inter-item correlation of 0.15–0.50, 26 indicating that the items are highly correlated and measure the same construct or idea.
Inter-item correlation coefficient between components of ILQI in patients with ITP attending the hematology clinic at TASH in Addis Ababa, Ethiopia, 2022 (n = 100).

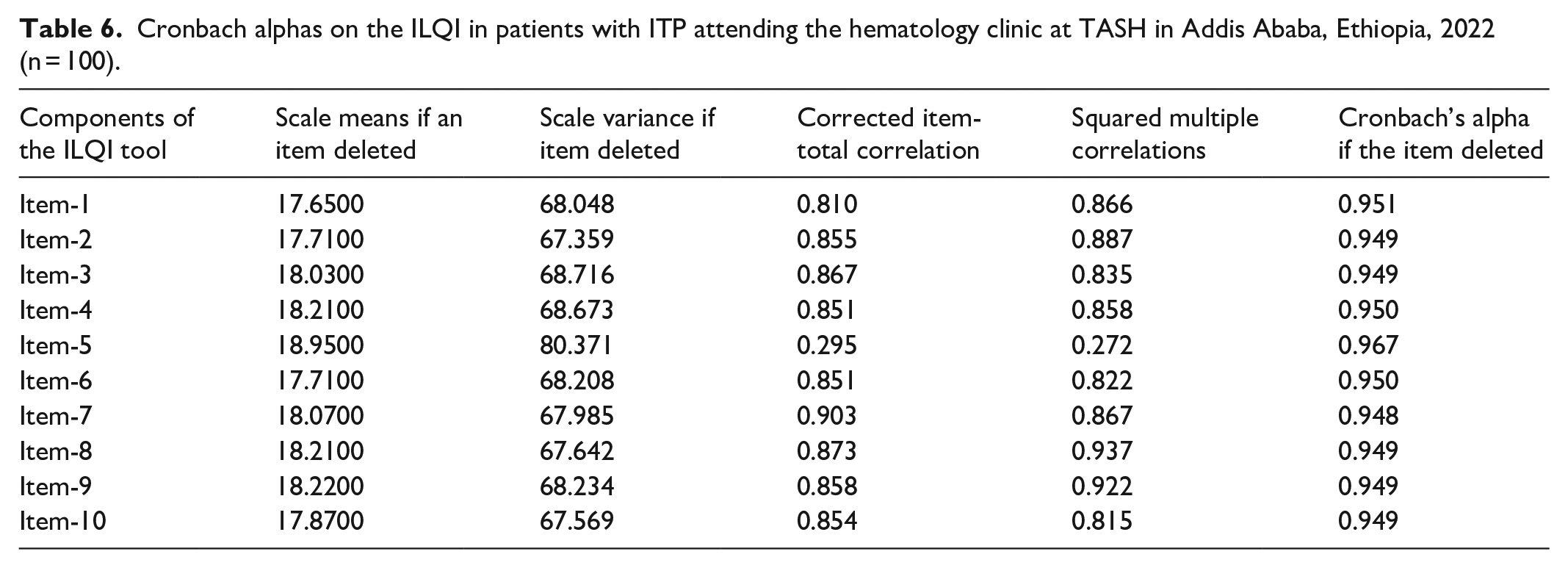
Cronbach’s alpha and item-total correlation of the ITP tool QoL index
The results of the reliability analysis revealed that the Amharic version of the ILQI had excellent internal consistency. Cronbach’s alpha was 0.956 overall, and 0.953 for standardized items. These values were greater than the traditional cut-off point of 0.7,25,33 indicating that the ILQI is trustworthy in this context (Table 6).
Cronbach alphas on the ILQI in patients with ITP attending the hematology clinic at TASH in Addis Ababa, Ethiopia, 2022 (n = 100).
Validity analysis for ITP tool QoL index
In this study, factor loading, factor analysis, the KMO measure of sampling adequacy, 30 Bartlett’s test for sphericity, 31 and the scree plot were used to assess the construct validity of the components of the Amharic version of the ILQI.
Factor analysis of the ITP tool QoL index
The KMO measure for sampling adequacy and Bartlett’s test for sphericity were used to determine whether the sample was suitable for factor analysis.30,31 The KMO value was 0.875, and the results of Bartlett’s test for sphericity were significant (X 2 = 1244.824, Degrees of freedom (DF) = 45, p < 0.001). As a result, principal factor analysis was possible. 34
When the results of the factor analysis were compared between the individual components of the ITP patients, it was discovered that Item-1 had an eigenvalue greater than 1. Item-1 with eigenvalues greater than 1 described 72.532% of the total variance of all 10 ILQI domains, according to this interpretation. This indicates that Item-1 accounted for the most substantial portion of the ILQI values in this setting (Table 7). The scree plot analysis also shows that Item-1 was crucial in explaining the observed variability (Figure 2).
Principal factor analysis of the 10 items of the ILQI in patients with ITP attending the hematology clinic at TASH in Addis Ababa, Ethiopia, 2022 (n = 100).

Extraction method: principal component analysis.

Scree plot of the 10 items of the ILQI in patients with ITP attending the hematology clinic at TASH in Addis Ababa, Ethiopia, 2022 (n = 100).
Factor loading of ITP tool QoL index
To test the construct validity of the Amharic version of the ILQI tool, factor loading was conducted. 34 The results of the factor loading showed that the influence of ITP on the completion of daily tasks (0.928), the influence of ITP on the ability to assist people (0.903), and the influence of ITP on the ability to focus on daily tasks (0.901) had high positive loading (Table 8).
Factor loading construct validity of the 10 items of the ILQI in patients with ITP attending the hematology clinic at TASH in Addis Ababa, Ethiopia, 2022 (n = 100).

Extraction method: principal component analysis.
Convergent validity correlations of the ILQI
Convergent validity correlations were used to examine the relationship between the ILQI total score (alternative score), 18 and each ILQI item, as well as the relationship between individual ILQI items. Table 9 shows the assumed strong correlations (r > 0.50) and moderate correlations (r > 0.30 but 0.50). 35 The ILQI total (alternative score) had a strong (r = 0.9) correlation with Item-8 (impact of ITP on the ability to support people close to you) and a moderate (r = 0.317) correlation with Item-5 (impact of ITP on their sex life).
Convergent validity correlations of the total ILQI score (alternative score) and each ILQI item in patients with ITP attending the hematology clinic at TASH in Addis Ababa, Ethiopia, 2022 (n = 100).

Correlation is significant at the 0.01 level (2-tailed).
Correlation is significant at the 0.05 level (2-tailed).
EFA of the ILQI
The factor loadings of the EFA show that the ILQI has a unidimensional structure, which supports the formation of an overall score that includes all ten items.27,29 Table 10 shows the structural equation model for the EFA sample. The model fit indices showed acceptable model fit (root mean squared error of approximation (RMSEA) = 0.282, probability RMSEA = 0.05 (p-close) 0.0001, comparative fit index (CFI) = 0.782, Tucker-Lewis index (TLI) = 0.720, and standardized root mean square residual (SRMR) = 0.061). The explained common variance is 0.904, indicating that the ILQI is unidimensional and a total score can be used, as shown in Figure 3.
Structural equation model for the EFA of total ILQI score (alternative score) and each ILQI item in patients with ITP attending the hematology clinic at TASH in Addis Ababa, Ethiopia, 2022 (n = 100).


Confirmatory factor analysis solution of the standardized unidimensional factor of total ILQI score (alternative score) and each ILQI item in patients with ITP attending the hematology clinic at TASH in Addis Ababa, Ethiopia, 2022 (n = 100).
Mean scores of ILQI for ITP patients
Mean scores for the ILQI tool ranged from 1.12 to 2.42, with the highest mean score (2.42 ± 1.17) for the assessment of the impact of ITP on work life or study and the lowest mean score (1.12 ± 0.78) for the assessment of ITP on sex life. Whereas the overall mean score for ILQI was 2.01 ± 0.92 (Table 11).
Mean scores of ILQI in patients with ITP attending the hematology clinic at TASH in Addis Ababa, Ethiopia, 2022 (n = 100).

Discussions
The purpose of this study was to evaluate the psychometric properties of the ILQI in Amharic for Ethiopia. This study is the first to investigate whether the Amharic version of the ILQI tool is a good, reliable, and valid tool for assessing HRQoL in Ethiopian ITP patients. Platelet count and bleeding risk are often used in the clinical evaluation of ITP. It was found that there is an unmet need for a valid and reliable patient-based outcome assessment that focuses on the understudied HRQoL influences associated with ITP. 20 The interpretation of the results of psychometric tools used in clinical practice and research is referred to as validity and reliability. Psychometrically sound assessment methods are necessary for optimal clinical research and educational practice. 36 The results confirmed that the Amharic version of the ILQI is a psychometrically sound tool to assess HRQoL in Ethiopian ITP patients.
Cronbach’s alpha and the correlation coefficient between items were used to determine the ILQI’s reliability. Cronbach’s alpha was 0.956 overall, and all reported Cronbach’s alpha values for individual items were greater than 0.7. The Cronbach’s alpha of the individual ILQI items was greater than 0.90, indicating that the items within the unidimensional score are related and measure the same underlying construct.25,33 When Item-5 (impact of ITP on sex life) was deleted, the Cronbach’s alpha increased from 0.956 to 0.967; this could be because 15% of study participants responded “does not apply to my life” or “would not like to say” when asked how ITP has affected their sexual life. The inter-item correlation coefficient ranged from 0.147 to 0.956, corresponding to an average inter-item correlation of 0.15 to 0.50 and demonstrating that when compared to items on another scale, each item is highly correlated with the other. In this reliability analysis, Cronbach’s alpha (0.933) and the correlation coefficient between items (0.2205–0.8001) were consistent with the first psychometric assessment of the ILQI in a global survey of patients with ITP. 18 According to this reliability analysis, the Amharic version of the tool was a reliable patient-reported outcome measure of HRQoL in Ethiopian ITP patients.
The ILQI tool’s validity was investigated using factor loading and factor analysis. 27 The individual components of the ITP patients were subjected to factor analysis, and it was discovered that Item-1 (impact of ITP on their work life or studies) had an eigenvalue greater than 1. With this interpretation, Item-1 with eigenvalues greater than 1 explained 72.532% of the total variance of all 10 ILQI domains. This indicates that Item-1 (impact of ITP on their work life or studies) explained most of the items in the Amharic version of the ILQI in Ethiopian ITP patients. In addition to factor analysis, factor loading results revealed that ITP had a high positive loading on the ability to complete daily tasks (Item-7) (0.928), ability to help people (Item-8) (0.903), and ability to focus on daily tasks (Item-3) (0.901). Similarly, Item-7 (0.899) followed by Item-1 (0.874) and Item-3 (0.864) also had higher factor loading in the first psychometric assessment of the ILQI in a global survey of patients with ITP. 18 Furthermore, during the validity analysis, it was found that Item-5 had the lowest positive factor loading at 0.337. This result is consistent with the first psychometric assessment of the ILQI in a global survey of patients with ITP, where Item-5 had the lowest positive factor loading at 0.783 compared to the other items. 18
Convergent validity correlations were used to examine the relationship between the ILQI total score (alternative score) 18 and each ILQI item and the relationship between individual ILQI items. Strong correlations (r > 0.50) and moderate correlations (r > 0.30 but 0.50) were assumed.26,35 The ILQI total (alternative score) had a strong (r = 0.9) correlation with Item-8 (impact of ITP on the ability to support people close to you) and a moderate (r = 0.317) correlation with Item-5 (impact of ITP on sex life). In this analysis, there is a strong correlation between Item-8 (impact of ITP on the ability to support close people) and Item-9 (negative impact of ITP on their hobbies) with (r = 0.956), followed by Item-1 (impact of ITP on working life and studying) and Item-2 (absence from work or education due to ITP) with (r = 0.891). On the other hand, the first psychometric assessment of the ILQI in a global survey of patients with ITP found a strong correlation between Item-1 (impact of ITP on work life or study) and Item-2 (absence from work or education due to ITP with (r = 0.804)). 18 In addition, a weak correlation was found between Item-5 (impact of ITP on their sex life) and other ILQI items, ranging from r = 0.147 to 0.371, with the lowest correlation (r = 0.147) with Item-5 (impact of ITP on their sex life) and Item-6 (ITP affects your energy level). This weak correlation may be because Item-5 (impact of ITP on sex life) included the additional response option “not applicable/prefer not to say” and 15% of study participants responded “not applicable/prefer not to say,” which may have contributed to the poor correlations. The first psychometric assessment of the ILQI in a global survey of ITP patients found a weak correlation (r = 0.2205) between Item-5 (impact of ITP on their sex life) and Item-1 (impact of ITP on their work life or studies). 18
The overall mean score for the ILQI was 2.01 ± 0.92 and ranged from 1.12 to 2.42. ITP had the greatest impact on their work life or studies with a mean score of (2.42 ± 1.17), followed by their energy level (2.36 ± 1.11). On the other hand, the impact of ITP on their sex life was the least, followed by the negative impact of ITP on their hobbies with a mean of (1.12 ± 0.78) and (1.85 ± 1.10), respectively. According to the literature, ITP patients experience negative effects on their sexual activities, such as decreased libido and bruising and bleeding during sexual intercourse. 11 In this study, however, 15% of study participants responded “does not apply to my life” or “would not like to say” when asked about the impact of ITP on their sexual life. A qualitative study conducted in the UK discovered that the impact of ITP on sexual life was less significant. 19 In addition, the effects of ITP on the completion of daily tasks (2.00 ± 1.07) and the ability to concentrate on daily tasks (2.42 ± 1.17) were average.
All factor loadings in this output are statistically significant (p < 0.001). The output subset with the standardized factor loadings. All indicators are interpreted as correlations between each indicator and the latent factors because they all load on a single factor. Both the CFI and the TLI are incremental fit indices. These indices have values greater than 0.95, indicating a very good fit. Values of 0.90 or higher are considered evidence of an acceptable fit. 37 In this analysis, the values of CFI (0.782) and TLI (0.720) are low (both < 0.90), indicating a poor fit model. SRMR values up to 0.05 are considered indicative of a good-fitting model. Values between 0.05 and 0.10 indicate an acceptable fit. 37 The SRMR (0.061) was within the “acceptable range”(<0.1). The smallest possible RMSEA is 0. Values <0.05 are considered indicative of values up to 0.08 are considered acceptable, and p-close is a test of whether the model deviates significantly from a model that fits the data well (RMSEA ⩽ 0.05).37,38 The RMSEA (0.282) of this analysis was and the p-close test is significant (p < 0.001). Both findings indicate a poor model fit to the data. On the other hand, the model fit indices of the first psychometric evaluation of the ILQI in a global survey of ITP patients showed acceptable model fit (RMSEA = 0.06, CFI = 0.99, and SRMR = 0.02). 18 Finally, this validation and reliability study had certain limitations. The study did not measure test-retest reliability and the responsiveness of the ILQI.
Conclusions
The Amharic version of the ILQI tool is a valid and reliable tool that could be used in clinical assessments to study the HRQoL of ITP patients in Ethiopia. Psychometric evaluation of the Amharic version of the ILQI shows that it is suitable for measuring the impact of ITP on HRQoL in clinical practice in the Ethiopian population, which may improve the consistency of patient-centered decision making and lead to better outcomes for those patients whose HRQoL has been negatively affected by their ITP. In addition, cross-cultural implementation of the ILQI will lead to more consistent care in clinical practice, better outcomes for patients with ITP, and an improvement in overall HRQoL that comprehensively reflects the experiences of demographically diverse patients with ITP.
Supplemental Material
sj-docx-1-smo-10.1177_20503121231199869 – Supplemental material for Reliability and validity of the Amharic version of immune thrombocytopenia life quality index tool for assessment of the health-related quality of life in Ethiopian patients of immune thrombocytopenia: Cross-sectional study
Supplemental material, sj-docx-1-smo-10.1177_20503121231199869 for Reliability and validity of the Amharic version of immune thrombocytopenia life quality index tool for assessment of the health-related quality of life in Ethiopian patients of immune thrombocytopenia: Cross-sectional study by Dessale Abate Beyene, Eskinder Ayalew Sisay, Atalay Mulu Fentie and Amha Gebremedhin in SAGE Open Medicine
Supplemental Material
sj-docx-2-smo-10.1177_20503121231199869 – Supplemental material for Reliability and validity of the Amharic version of immune thrombocytopenia life quality index tool for assessment of the health-related quality of life in Ethiopian patients of immune thrombocytopenia: Cross-sectional study
Supplemental material, sj-docx-2-smo-10.1177_20503121231199869 for Reliability and validity of the Amharic version of immune thrombocytopenia life quality index tool for assessment of the health-related quality of life in Ethiopian patients of immune thrombocytopenia: Cross-sectional study by Dessale Abate Beyene, Eskinder Ayalew Sisay, Atalay Mulu Fentie and Amha Gebremedhin in SAGE Open Medicine
Footnotes
Acknowledgements
The authors would like to thank all the study participants for their time and willingness to participate in the study. We would also like to express our sincere gratitude to the outpatient hematology clinic TASH, Department of Pharmacology and Clinical Pharmacy, School of Pharmacy, College of Health Science, Addis Ababa University, and the data collectors for their support throughout the study period.
Authors’ contributions
DAB contributed to the study design, performed the statistical analysis; wrote, reviewed, and edited both the original draft and final manuscript. EAS, AMF, and AG conceptualized and participated in the study design, critically reviewing, modifying, and analyzing the draft of the manuscript. All the authors have read and approved the final version of the manuscript.
Data sharing statement
The datasets used during the current study are available from the corresponding author at a reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical consideration and consent statement
The ethical review board of Addis Ababa University, College of Health Sciences, and School of Pharmacy (ERB/SOP/487/14/2022) approved. A formal approval letter was obtained from the Department of Internal Medicine (TASH), Division of Hematology, before data collection. The objectives of the study were clearly explained to the participants. Data were collected after each participant provided written informed consent. Both written informed consent and family or legal guardian consent were obtained for participants aged 14–18 years, and written informed consent was obtained for participants aged >18 years. Participants in the study were given the option to decline or discontinue participation at any time, and they were also allowed to ask questions about the study. To maintain confidentiality, the participant’s name was not used during data collection, and all other personal information was completely obscured throughout the study period.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
