Abstract
Introduction:
Arteriovenous fistula (AVF) is the preferred vascular access option due to its lower risk of complications and better long-term outcomes. However, AVF maturation failure is still quite high. Achieving an adequate blood flow rate (BFR) through the AVF is essential for maintaining hemodialysis adequacy. This study aims to investigate brachial artery intraoperative BFR and peak systolic velocity (PSV) increase as a predictor of brachiocephalic AVF maturation.
Methods:
A multicenter retrospective cohort study was conducted on patients with end stage renal disease undergoing brachiocephalic AVF creation from July 2019 to February 2022 from five hospitals. Doppler ultrasound examinations of BFR and PSV were collected. BFR and PSV increases were calculated by comparing pre-operative and intraoperative results. Maturity was determined at 6 weeks postoperatively.
Results:
This study included 83 patients, with 50 patients (60.24%) achieving maturity at 6 weeks. Brachial artery BFR difference has an excellent diagnostic value to predict brachiocephalic AVF maturation with an area under the curve (AUC) of 97%. BFR increase of 184.58 ml/min predicts brachiocephalic AVF maturity with a sensitivity of 100%, specificity of 84.8%, and accuracy of 93.98%. Meanwhile, brachial artery PSV increase has a low diagnostic value to predict brachiocephalic AVF with an AUC of 71.2%. PSV increase of 8.97 cm/s predicts brachiocephalic AVF maturity with a sensitivity of 82%, specificity of 51.5%, and accuracy of 69.9%.
Conclusion:
The increase in intraoperative brachial artery BFR and PSV in brachiocephalic AVF can be used as a useful parameter to predict AVF maturation.
Introduction
Kidney transplantation is the best renal replacement therapy choice for end stage renal disease (ESRD) patients. However, hemodialysis is the most common renal replacement therapy management globally, especially in developing countries such as Indonesia.1,2 In 2010, there were 2.62 million people worldwide undergoing hemodialysis therapy which is expected to double by 2030, while in Indonesia, there were 132,142 chronic kidney disease patients undergoing chronic hemodialysis.1–3
Arteriovenous fistula (AVF) is one of the best options recommended for hemodialysis access due to lower complications and better patency rate compared to other vascular accesses. 4 The choice of AVF as vascular access for hemodialysis in developing countries is the first choice because it is more accessible. In general, there are three AVF surgery types, namely radiocephalic, brachiocephalic, and brachiobasilic. Among the three types of AVF, brachiocephalic AVF is one of most widely used as hemodialysis access. AVF maturation failure is still quite high. In general, the failure rate of AVF maturation ranges from 20% to 60%. Good surgical technique and proper early treatment are expected to increase the percentage of maturation and patency of AVF.2,4,5 The ideal AVF has matured and can be used as hemodialysis access after 4–8 weeks postoperatively.2,4-6 In the Clinical Management Guidelines issued by the Kidney Disease Outcomes Quality Initiative in 2019, an AVF is said to be mature if the access meets the rule of six criteria of blood flow at a rate of >600 ml/min; distance from the skin <0.6 cm; and has a diameter >0.6 cm at the sixth week postoperative evaluation of AVF.4,7–11 Diabetes mellitus (DM), prior vascular procedures, and atherosclerotic disease may impair vascular quality in ESRD patients. Therefore, ultrasound examination of blood vessels before AVF surgery is critical to reduce the risk of maturation failure. 10
AVF is a vital lifeline for patients with renal failure with hemodialysis replacement therapy. AVF that fails to mature in time causes a burden for patients, health workers, and facilities as well as the state because it requires special interventions, site selection, and other hemodialysis access methods.1,2 The causes of hemodialysis maturity failure should be anticipated so that the risk can be reduced as little as possible. AVF maturity failure can be reduced by controlling the patient’s comorbid factors affecting vascular quality and performing a good perioperative examination and correct surgical techniques.9–12 Early detection of the risk of AVF maturity failure can be done as a preventive measure to reduce morbidity and is expected to reduce the burden on personnel and health facilities.13,14 One essential factor in maintaining hemodialysis adequacy is the achievement of blood flow rate (BFR) at the hemodialysis access, in this case, AVF. Intraoperative evaluation of BFR values shortly after AVF creation can improve the failure rate of the AVF creation maturation process.14–17
BFR measurement in the feeding-artery is currently considered to have an advantage over the draining vein, especially immediately after surgery, because the vein wall is thinner and is strongly influenced by pressure by the operator during ultrasound, which results in high variation in BFR and peak systolic velocity (PSV) measurements in the vein. In addition, arteries have a larger diameter, laminar blood flow, and less turbulence.8–10,14,16–18
Pratama et al. 19 reported that there was a relationship between PSV values and an increase in PSV with the level of maturation in radiocephalic AVF with an accuracy of 92.9%. However, there are no studies on the relationship between an increase in BFR or PSV with the incidence of AVF maturation, especially in the feeding-artery of brachiocephalic AVF. Therefore, we investigated the relationship between the increase in BFR and PSV of the feeding-artery (brachial artery) intraoperatively and whether it can be a predictor of brachiocephalic AVF ultrasonographic maturation.
Methods
A Multicenter retrospective cohort study was conducted on patients with ESRD undergoing end-to-side brachiocephalic AVF creation from July 2019 to February 2022 from five hospitals in four cities. Prior ethical approval from the ethics committee of the Faculty of Medicine University of Indonesia—Cipto Mangunkusumo Hospital Number KET-362/UN2.F1/ETIK/PPM.00.02/2022 was in accordance with the Declaration of Helsinki. Written informed consent for publication of the participants’ data was obtained from the participants during the patient visits to the hospital.
Patients included in the study are ESRD patients (estimated glomerular filtration rate (eGFR) < 15 ml/min/1.73 m2) with a pre-operative cephalic vein and brachial artery diameters of ⩾2 mm undergoing brachiocephalic AVF operation. Critically ill patients, patients with previous AVF operation, patients with complications before 6 weeks postoperatively, and patients who did not visit the center after 6 weeks are excluded. Pre-operative data extracted includes age, sex, blood pressure, hypertension, diabetes, and smoking history. Doppler ultrasound examinations of BFR and PSV were performed on the brachial artery pre-operatively and intraoperatively. Ultrasound was measured by placing the probe linearly on the brachial artery 5 cm proximal to the anastomosis and measuring the BFR (ml/min) and the PSV (cm/s). BFR and PSV differences were calculated by comparing pre-operative and intraoperative results. Pre-operative ultrasounds were performed before the operation in the operating room. Intraoperative ultrasounds were performed immediately after anastomosis between the cephalic vein and the brachialis artery was achieved. Postoperative ultrasounds were conducted 6 weeks after the operation during the patient’s visits to the hospital. All ultrasound was performed by a consultant vascular surgeon with more than 10 years of experience. AVF maturity was checked 6 weeks postoperative using the rule of six criteria of blood flow at a rate of >600 ml/min, vein depth <0.6 cm, and a diameter >0.6 cm.
Statistical analyses
Categorical data were analyzed using chi-square analysis. Numerical data were analyzed using Independent T-test or Mann–Whitney test. Results were shown as mean ± standard deviation (SD) if normally distributed or else median (min; max) was used. Receiver operator curve (ROC) diagnostic tests were used to determine the cut-off value of BFR and PSV difference to predict AVF maturation. Multivariate analyses were conducted on factors with p < 0.25 on bivariate analysis.
Results
A total of 83 patients from five hospitals were included in this study. Subject characteristics are presented in Table 1.
Subjects baseline characteristics.

A total of 50 patients (60.24%) achieves AVF maturity 6 weeks after brachiocephalic AVF creation. There was no significant difference between age, sex, DM, hypertension, smoking history, and pre-operative artery and vein diameter (Table 2). There was a statistically significant difference between brachiocephalic AVF maturation and brachial artery BFR intraoperative increase (p < 0.001) and PSV intraoperative increase (p = 0.001) (Table 3).
Subjects baseline characteristics between mature and immature group 6 week post-brachiocephalic AVF creation.

OR: odds ratio; SD: standard deviation; IQR: interquartile range; CI: confidence interval.
Mann–Whitney.
Chi square.
Independent T-test.
BFR and peak systolic value difference between mature and immature group 6 week post-brachiocephalic AVF creation.

Mann–Whitney. BFR: blood flow rate; PSV: peak systolic value; IQR: interquartile range.
ROC measurement for brachial artery BFR difference has an excellent diagnostic value to predict brachiocephalic AVF maturation at the sixth week with an area under the curve (AUC) of 97% (Figure 1). The highest sensitivity and specificity were obtained when the brachial artery BFR increase was 184.58 ml/min. This cut-off value predicts brachiocephalic AVF maturity at the sixth week with a sensitivity of 100%, specificity of 84.8%, a positive predictive value (PPV) of 90.5%, a negative predictive value (NPV) of 100%, and an accuracy of 93.98%.

ROC curve depicting the sensitivity and specificity of brachial artery BFR difference to predict brachiocephalic AVF maturation at the sixth week.
On the other hand, the ROC measurement for brachial artery PSV increase has a low diagnostic value to predict brachiocephalic AVF maturation at the sixth week, with an AUC of 71.2% (Figure 2). The best sensitivity and specificity were obtained at the 8.97 cm/s cut-off. This value predicts brachiocephalic AVF maturity at the sixth week with a sensitivity of 82%, specificity of 51.5%, a PPV of 71.9%, an NPV of 65.4%, and an accuracy of 69.9%.

ROC curve depicting the sensitivity and specificity of brachial artery peak systolic value difference to predict brachiocephalic AVF maturation at the sixth week.
Multivariate was subsequently done to analyze factors affecting maturation (Table 4). Brachial artery BFR increase can predict 87.5% of maturation.
Multivariate analysis of factor affecting maturation.
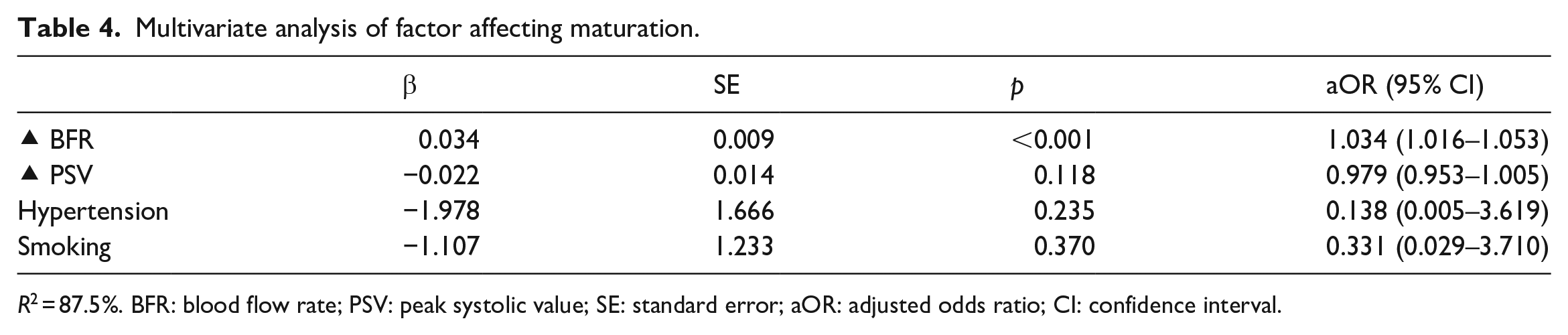
R2 = 87.5%. BFR: blood flow rate; PSV: peak systolic value; SE: standard error; aOR: adjusted odds ratio; CI: confidence interval.
Discussion
Currently, hemodialysis as renal replacement therapy is the most common choice in developed and developing countries such as Indonesia. AVF is a vascular access for hemodialysis recommended by KDOQI due to its relatively low complication rate and good patency. However, the failure rate of maturation is still relatively high, reaching 60%.1,4
Based on the characteristics of the subjects, it is known that the number of ESRD patients who underwent brachiocephalic AVF surgery was slightly more predominantly male (50.6%), with a mean age of 53.2 years. This is in accordance with data from USDRS in 2021 20 and the 2018 Indonesia Renal Registry report. 3 However, we found no relationship between sex and brachiocephalic AVF maturation consistent with studies by Won et al. and Joannides et al.2,20,21,22 We also found no relationship between age and level of brachiocephalic AVF maturation consistent with studies by Chan et al. and Siddiqui et al.1,2,5,23
Smoking is one of the factors that have an association with the incidence of atherosclerosis in arterial blood vessels. The quality of blood vessels can determine the maturation and patency rate of an AVF.4,15,23 However, in this study, we found no significant relationship between smoking and brachiocephalic AVF maturation. This is consistent with a study conducted by Arhuidese et al. 24 However, it was mentioned that smoking caused a decrease in the patency rate of AVF. This difference may be due to the data in this study did not differentiate between active and inactive smokers.
Hypertension and DM are the most common comorbidities found in patients with ESRD. However, we found that the maturation of the brachiocephalic AVF was not associated with DM or hypertension. This is consistent with research conducted by Santosa et al. in Padang and by Siddiqui et al. in Canada. The results of studies and publications linking DM and hypertension with AVF maturation are often contradictory and inconsistent. This may be because hypertension and DM are chronic degenerative diseases and take time to affect the quality of hemodynamics and vascular anatomy, which will affect the quality and maturation of AVF. In addition, it is also influenced by each individual long-term control of blood sugar and blood pressure levels.6,25
The mean pre-operative brachial artery diameter was similar between maturing and failing AVF. There was no significant difference between the pre-operative brachial artery diameter and the maturation of brachiocephalic AVF in this study. Various literature shows that both venous and arterial diameters are associated with AVF maturation. Based on the publication by Allon et al., AVF with an arterial diameter >2 mm has a good maturation rate and primary patency. However, in this study, the subjects whose data were statistically processed met the inclusion criteria, namely those with an arterial diameter of 2 mm, so they had a better maturation rate. This further strengthens the existing theoretical basis that good mapping using ultrasound of the size and anatomy of blood vessels improves the success rate of maturation of an AVF.6,8–10,16
The pre-operative cephalic vein diameter of brachiocephalic AVF did not significantly differ from the AVF maturation rate at week six postoperatively. This study’s median cephalic vein diameter was 2.65 mm for those who matured and 2.69 mm for those who failed to mature. As with the diameter of the brachial artery, subjects with a cephalic vein diameter of 2 mm were used as subjects in this study. Research conducted by Masengu and Hanko reported that the pre-operative vein size above 2.5 mm has a higher AVF maturation success rate compared to smaller sizes. 26
Researchers chose the brachial artery as the location for measuring BFR and PSV because the artery has several advantages, including because it has a thicker wall compared to the vein wall. This reduces the errors made by the ultrasound operator when pressing the probe. In addition, the shape of the artery is straighter, and the resulting flow is also a linear flow, so deviations or differences in measurements made by the ultrasound operator can be avoided. 27
This study aims to find new predictors that can represent the inflow and outflow conditions in AVF, which are expected to be predictors of maturation. From the subject data obtained by ultrasound examination, it was found that the median increase in intraoperative BFR of brachiocephalic AVF was higher in subjects who matured, which was 342.90 ml/min, compared to subjects who failed to mature, which was 83.94 ml/min. There was a significant difference in the increase of intraoperative BFR between maturing and failing to mature brachiocephalic AVFs, where a high increase in BFR allows an AVF to mature. This is in accordance with the theoretical basis of shear stress that has been previously disclosed. 28 The increase in BFR occurs due to the response of the brachial artery as the feeding artery of the brachiocephalic AVF to increased shear stress after anastomosis of the brachial artery to the cephalic vein. In this study, the cut-off value of the intraoperative brachial artery BFR increase was 184.58 ml/min with a sensitivity level of 100% and specificity of 84.8%, with an accuracy value of 93.98%. This is in line with research conducted by Saucy et al., who proved that BFR is one of the predictors of AVF maturation. Bergman et al. reported that the cut-off value of BFR draining vein post-anastomosis in brachiocephalic AVF of 308 ml/min could predict AVF maturity. Johnson et al. also published a similar report, mentioning post-anastomosis draining vein BFR above 320 ml/min was associated with lower revision rates and a longer AVF patency rate. The brachial artery BFR cut-off by the researchers has a lower number because the researchers calculated the increase between pre-operative and intraoperative. A high increase in BFR value indicates a good quality blood flow, not only at the measurement location but also in the flow that passes through, including the draining vein. In this study, five subjects with increased brachial artery BFR above 184.58 failed to mature at week 6. The possible reasons for this maturation failure are due to the influence of anatomy and anatomical structure and physiology of blood vessels, as well as postoperative conditions. This follows Lin et al., who said that if the intraoperative AVF is considered good, it is necessary to closely monitor postoperatively to prevent maturation failure and improve the patency rate. We could not identify the cause in more detail because this study used retrospective data. A possibility of a low increase in blood rate may be caused by stenosis in the blood vessel, causing AVF immaturity.16,28–32
In this study, it also appears that measuring the increase in PSV in the brachial artery intraoperatively for brachiocephalic AVF can help predict maturation at week 6. The median value of PSV increases in subjects who experienced maturation of brachiocephalic AVF at the sixth postoperative week was higher compared to subjects who failed AVF maturation. A significant difference was found between intraoperative increases in PSV in AVF maturity. A high intraoperative increase in brachial artery PSV has a good impact on AVF maturation. This study found that the cut-off of intraoperative brachial artery PSV increase in brachiocephalic AVF was 8.97 cm/s with a sensitivity value of 82% and specificity of 51.5%. Meanwhile, the accuracy value was obtained at 69.9%. This figure is smaller than the increase in BFR as a predictive factor for the maturation of brachiocephalic AVF. According to Blackwell and Fruhwald, there is a relationship between arterial flow volume and arterial PSV, which can be explained based on the formula for calculating flow volume and the formula for calculating mean flow velocity. Therefore, an increase in PSV will be followed by an increase in BFR. However, high PSV is also used as an indicator of stenosis of a blood vessel related to the location of the PSV measurement, whether in the pre-stenosis, stenosis, or post-stenosis area. A total of 16 subjects had an intraoperative brachial artery PSV increase of more than 8.97 cm/s but did not experience maturation. The possible reason for the high PSV value was due to stenosis of the vessel. Research results and publications relating PSV to successful AVF maturation are often contradictory. Lockhart et al. reported no association between AVF maturation and arterial PSV values. However, Sedlacek et al. reported that an increase in PSV >50 cm/s is associated with increased AVF maturation.33,34
The strength of this study is that it is the first to assess the increase in BFR and PSV of the brachial artery as predictors of brachiocephalic AVF maturation. The results of this study provide promising results, where the increase in BFR and PSV of the brachial artery by ultrasound can be used as a predictor of brachiocephalic AVF maturation with acceptable accuracy values.
The limitation of this study is that it is a retrospective study, so researchers cannot identify and follow subjects who fail to experience AVF maturation. In addition, this study was conducted on subjects with optimal brachial artery and cephalic vein diameters above 2 mm, so further research is needed to assess the relationship between increased arterial BFR and PSV to AVF maturity in subjects with suboptimal blood vessel size. Vasospasm right after manipulation may also play a role in affecting the intraoperative brachialis artery diameter, subsequently their BFR and PSV. Follow-up in this study was up to 6 weeks postoperatively, so further research is needed to see the long-term effects and AVF patency of intraoperative BFR and PSV increase values. This study also did not monitor until hemodialysis was performed, so the clinical maturation rate is unknown. This study only assessed maturation in brachiocephalic AVF, which generally has a better maturation rate when compared to radiocephalic AVF. Therefore, it is necessary to conduct other studies to assess the effect of increasing BFR and AVF intraoperatively with the maturation rate of radiocephalic AVF. Lastly, this study was conducted by different operators in pre-operative and intraoperative ultrasound mapping until the sixth postoperative week and operators who performed AVF surgery. Although the qualifications and techniques used were the same, each operator’s approach and way of treating the tissue were different, which risks introducing bias in this study. Another limitation of the study is there is no power analysis on the sample size.
Conclusion
The increase in brachial artery BFR and PSV in brachiocephalic AVF obtained from examination using Doppler ultrasound can be used as a useful parameter to predict AVF maturation. The cut-off value of intraoperative brachial artery BFR increase of 184.58 ml/min can be used as a predictor of brachiocephalic AVF maturity with a sensitivity level of 100%, specificity of 84.8%, and an accuracy value of 93.98%, whereas brachial artery PSV increase of 8.97 cm/s can predict maturity with a sensitivity of 82%, specificity of 51.5%, and an accuracy of 69.9%. Increasing the intraoperative brachial artery BFR can be used as a consideration to determine the surgical strategy.
Footnotes
Author contributions
D.P. contributed to project administration, methodology, supervision, and resources. A.M. involved in formal analysis, data curation, writing—original draft, writing—review and editing. A.S. performed methodology, formal analysis, supervision, writing—original draft. A.K. involved in resources, conceptualization, writing—review and editing. M.F.F. and B.F.A. performed formal analysis, writing—review and editing. All authors read and approved the final manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Prior ethical approval from the ethics committee of the Faculty of Medicine University of Indonesia—Cipto Mangunkusumo Hospital Number KET-362/UN2.F1/ETIK/PPM.00.02/2022 was in accordance with the Declaration of Helsinki.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent for publication data was obtained from the participants.
