Abstract
Objective:
Short intravenous antimicrobial therapy for 2–3 days followed by its per oral comparable antimicrobial course is a crucial part of the antimicrobial stewardship program. However, nothing is known about this practice in Ethiopian hospitals. Therefore, this study assessed the proportion, associations, and outcomes of early intravenous to per oral antimicrobial switch for patients who were admitted to the three wards of Ambo University Referral Hospital.
Method:
A hospital-based prospective cohort pilot study was conducted. During the 3-month period, 117 patients met initial inclusion criteria and were followed until Day 3 of intravenous antimicrobial. Of these, 92 (78.6%) subsequently met criteria for early intravenous to per oral switch and are the cohort investigated in this study. Written informed consent was sought from participants and/or parents or guardian for ages 15–17 years. Logistic regression models and independent t-tests were done with a significance of
Results:
Out of 92 study participants, early intravenous to per oral antimicrobial switch was done only for 36 (39.1%). The only independent predictors for lack of early intravenous to per oral antimicrobials switch were poly-pharmacy (adjusted odds ratio = 3.4 at 95% confidence interval, 1.036–11.16;
Conclusion:
The proportion of early intravenous to per oral antimicrobial switch was unsatisfactory. There was a significant difference between the intervention and comparator groups in terms of length of hospital stay, in-hospital complications, and extra cost. Therefore, implementation of interventions that improve the practice of early intravenous to per oral switch is needed urgently.
Introduction
Antimicrobials are among the most commonly prescribed and as well the most misused drugs.1,2 Even though it has been a common practice to initially prescribe empirical intravenous (IV) antimicrobial therapy for seriously ill patients, the ideal route of administration of any antimicrobial is the one that achieves serum concentrations sufficient to produce the desired effect without producing any untoward effects. Thus, IV to per oral (PO) switch is often considered within 3 days after clinical stability.3,4 There are three methods of changing IV to PO antimicrobials: sequential therapy, switch therapy, and step down therapy based on the availability and effectiveness of its PO antimicrobial.3,5
The benefit of early IV to PO switch (within 72 h) has been demonstrated in previous studies conducted among hospitalized patients.6,7 These benefits include a lower risk of IV line-associated infections, decrease the length of hospital stay, reduced nursing workload, and decreased direct and indirect costs.2,5,8–11 It is essential, however, to apply strict evaluation of IV to PO switch criteria, to avoid inappropriate de-escalation.12,13 Thus, patients with meningitis, infective endocarditis, and bone and joint infection were not considered for IV to PO switch within 3 days because it requires a longer or full course of IV treatment.14–16 Thus, the practice of IV to PO switch within an appropriate time is one of the major aspects to improve the rational use of antimicrobials and it is among the major activities of the antimicrobial stewardship program of a given hospital.2,17 In developing countries including Ethiopia, most of the hospitals lack established and or functional antimicrobial stewardship program,8,18,19 because of different factors.20,21
Moreover, only a few studies have been done to identify the practice of early IV to PO switch of antimicrobial among hospitalized patients and almost all of these were done in developed countries; United Kingdom (UK), 22 the united states of America (USA), 23 Netherlands and Germany, 24 Switzerland,9,25 and United Arab emirate. 26
Furthermore, to our knowledge, there was only one published data regarding the practice of early IV to PO switch in Ethiopia. 8 In addition, many hospitals have no antimicrobial stewardship program. 27 Hence, this pilot study could serve as baseline data for identifying and preparing the action plan for Ambo University Referral Hospital (AURH) to promote rational antimicrobial use. Thus, this study was done with the following aims: (1) In patients who are clinically stable, to assess the rate of early IV to PO antimicrobial switch, (2) to determine the factors associated with early IV to PO switch, and (3) to compare the length of stay and cost outcomes between patients who switch early and those who do not.
Methods and materials
Study area and period
The study was conducted from January 01 to March 30, 2019 at AURH in Ambo town, West Shewa Zone, Oromia Region. AURH serves more than 2,586,0076 people of west Shewa Zone (2007 Ethiopian census report). The Hospital has 134 beds and the only referral hospital for the West Shewa zone. There was no antimicrobial stewardship program in AURH prior to this study.
Study design and study population
This is a pilot study done over 3 months. A hospital-based prospective cohort study was conducted. The study populations were all adult patients admitted to medical, surgical, and gynecology and obstetrics wards of AURH during the study period.
Inclusion and exclusion criteria
The inclusion criteria were: any patient with the age of ⩾15 years who was within 3 days of IV antibiotic treatment for an infection where oral antimicrobial could potentially be used for continuation treatment, that is, infection not recommended as IV only, appropriate oral antibiotic available (pharmacological equivalent and effective), and patient able to intake and absorb oral medicine.
Exclusion criteria: Septic shock, meningitis, infected prosthesis, surgical prophylaxis, immunocompromise (febrile neutropenia < 1000 cells/mm3 or functional asplenia), seizures with risk of aspiration, gastrointestinal bleeding or obstruction, patient refusal to take oral antibiotics, and antimicrobial use with unknown indication.
Sampling technique and sample size
A consecutive sampling technique was used, in which the researcher enrolled whom they could at the time with inclusion criteria. This was owing to unavailability of registered data regarding the number of patients with infectious disease specifically, patients on IV antimicrobial which is related to our objective.
Data collection procedure and tools
A semi-structured data abstraction tool was prepared with a slight modification of the tools which was used for a similar purpose by previous studies.8,28 The collected data included sociodemographic, clinical, medication/antimicrobial usage, costs, and outcomes.
This information was collected from patients’ charts, after 24 h of IV antibiotics, switch to PO was considered daily by two 5th-year Pharmacy students under the supervision of senior Clinical Pharmacists. They evaluated whether the patients had satisfied the criteria for early IV to PO antimicrobial switch based on meeting the following pre-defined criteria indicative of stable or improving infection8,17,28,29: temperature < 38°C for >24 h, pulse rate < 100 bpm, respiratory rate < 25 breath/min, oxygen saturation > 92%; tolerating oral fluids/medication or food; white blood cell count stable/normalizing <15 × 103 cells/mm3 (if available).
The cost of failing to switch appropriately from IV to PO was considered from Day 3 onwards. From the day they could have switched (e.g., Days 3, 4, 5, and at end of treatment duration) for each day the patient inappropriately continued IV, the additional costs of the antimicrobial itself (IV antimicrobial cost minus oral antimicrobial cost) and the cost of an inpatient bed (bed plus meal = 40 Ethiopian Birr (ETB)) per day was calculated. These costs were taken from the AURH Liaison office and Pharmacy cost list.
Early IV to PO switch and no early switch group
The study participants were categorized into two groups based on the status of early IV to PO antimicrobial switch. Before categorizing the study participants as early IV to PO switch/intervention and no early switch/comparator, they were followed until the fulfillment of the criteria (clinical stability) to consider IV to PO switch for 72 h. Then, the allocation was done by non-random method (by treating physician) to early IV to PO switched (36) and not switched (56).
Outcome measure
The primary endpoints were the mean cost difference between the two groups. Secondary endpoints were the proportion of early IV to PO switch, length of hospital stay, proportion patients that met criteria for early IV to PO switch, the rate of complications during the IV therapy, that is, phlebitis/injection site infection/pain/hospital-acquired pneumonia, and in-hospital mortality/treatment outcome. Per this study, antimicrobial is equivalent with antibiotic.
Data quality control and statistical analysis
To maximize the accuracy of this study, the data collection tool was pre-tested on 5% of the total study population from the medical ward; to check whether it can measure the intended objectives of the study, but removed from final analysis. During each step of data collection, processing, and analysis, any erroneous data were checked and corrected on daily basis through communication with a data collector and senior clinical pharmacists, ahead of patient discharge. The final data were analyzed using SPSS version 20. The results were presented with mean ± standard deviation for continuous variables after checking for outlier, while frequency and percentage were used for categorical variables. Linear regression for continuous variables and multivariate logistic regression for categorical variables were done to search for predictors variables for the given outcome. The mean difference between the intervention and comparator groups was analyzed using an independent t-test.
30
Significance was set at
Results
Sociodemographic and clinical characteristics of the study participants
During the 3-month period, 117 patients met initial inclusion criteria and were followed until Day 3 of IV antimicrobial. Of these, 92 (78.6%) subsequently met criteria for early IV to PO switch and are the cohort investigated in this study. Regarding the sociodemographic characteristics of the study participants, there was no significant difference between the two groups (Table 1).
Sociodemographic characteristics of the study participants at AURH, Ethiopia (
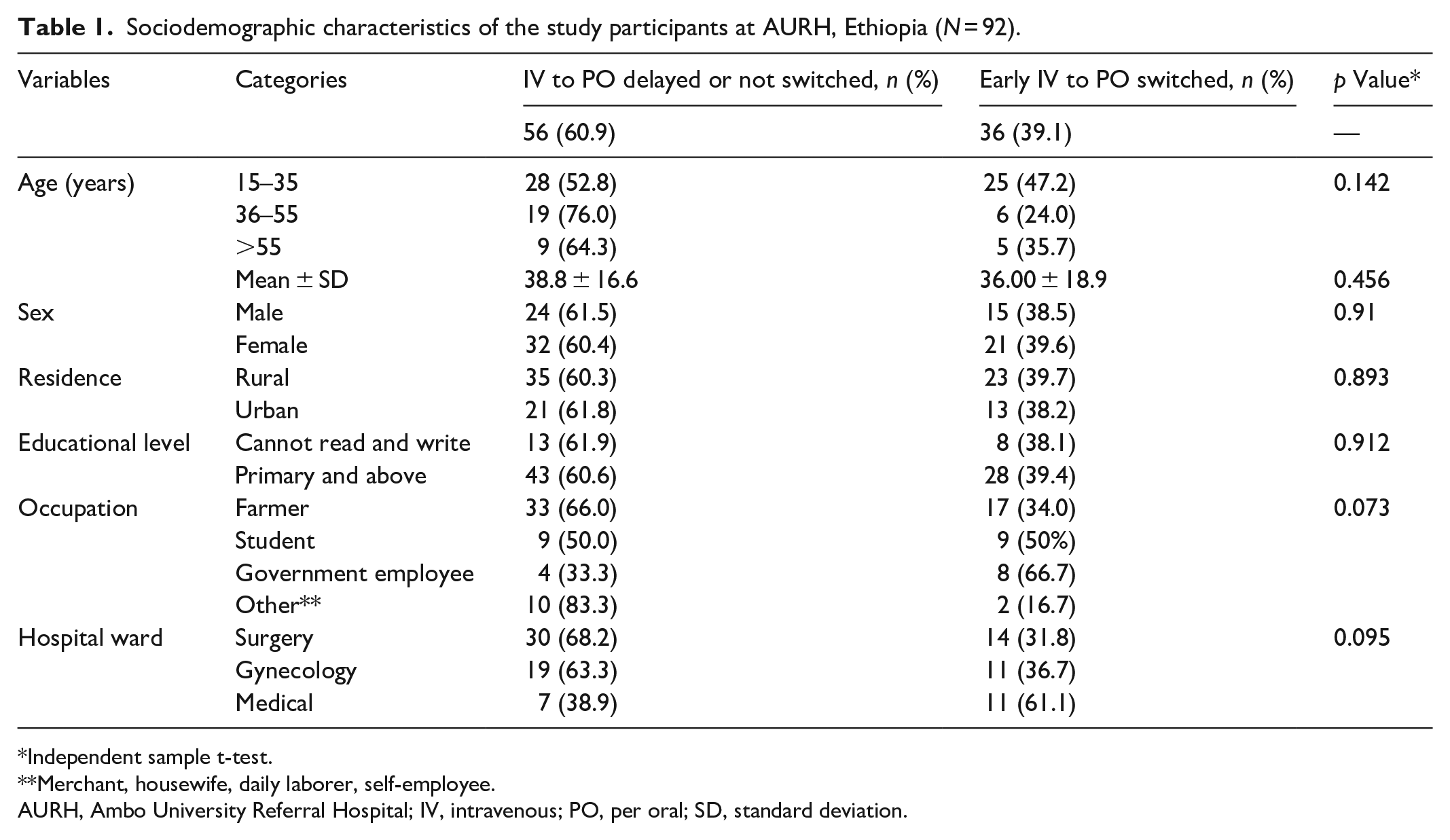
Independent sample t-test.
Merchant, housewife, daily laborer, self-employee.
AURH, Ambo University Referral Hospital; IV, intravenous; PO, per oral; SD, standard deviation.
The infections that they were on IV antimicrobial were gastrointestinal tract-related infection, Skin and soft tissue infection and community-acquired pneumonia. The most commonly prescribed IV antimicrobials for the study participants were ceftriaxone and metronidazole (Table 2).
Clinical and medication history of the study participants at AURH, Ethiopia.

Ceftazidime, fluconazole, acyclovir, and cotrimoxazole.
There are multiple responses.
Tongue biting injury, mesenteric ischemia, tonsillopharyngitis, and malaria
Alcohol intoxication, organophosphate poisoning, severe acute malnutrition, goiter, acute kidney injury, PUD/dyspepsia, and HIV/AIDS.
Nifedipine/amlodipine, RHZE, metoclopramide, atorvastatin, spironolactone, heparin, insulin, salbutamol, morphine, pyridoxine, tetanus antitoxoid.
AURH, Ambo University Referral Hospital; BPH, benign prostatic hyperplasia; CAP, community-acquired pneumonia; CPD, cephalo-pelvic disproportion; DM, diabetes mellitus; GIT, gastrointestinal tract; IV, intravenous; PO, per oral; PUD, peptic ulcer disease; SD, standard deviation; SSTI, skin and soft tissue infection.
Early IV to PO antimicrobial switch and associated features
Of 117 patients who were initially on IV antimicrobials, 92/117 (78.6%) of the study participants satisfied the criteria for early IV to PO switch/clinically stable within 72 h. However, early IV to PO antimicrobial switch was done only for 36/92 (39.1%) patients (Figure 1). The only independent predictors for lack of early IV to PO antimicrobials switch was poly-pharmacy (adjusted odds ratio = 3.4 at 95% confidence interval: 1.036–11.16;

Clinical stability of patients and the practice of IV to PO antimicrobial switch, Ethiopia.
Predictors for early IV to PO switch of antimicrobials at AURH, Ethiopia.

AOR, adjusted odds ratio; AURH, Ambo University Referral Hospital; CI, confidence interval; IV, intravenous; PO, per oral.

IV antimicrobials that were least likely switched to oral, Ethiopia (
Outcomes of early IV to PO antimicrobial switch
This study results have shown that there was a significant difference in terms of mean length of hospital stay in day (8.80 ± 3.57 versus 3.17 ± 0.74;
Overall treatment of outcome of patients with early IV to PO antimicrobials switch.

Chi-square or independent t-test.
Either of HAI, injection site pain or injection site infection.
Based on the visual analog scale of 0–10 (no pain is 0; mild is 1–4; moderate is 5–6; severe is 7–10 out of 10).
Extra cost of IV antimicrobial plus the cost of bed and meal paid out of pocket; at the time of this report writing (30/01/2020) 1 US$ is equal to 31.81 ETB, from National bank of Ethiopia.
Improved better response or totally cured at a time of discharge.
ETB, Ethiopian Birr; HAP, hospital-acquired pneumonia; IV, intravenous; N/A, not applicable; PO, per oral.
Moreover, the independent predictors for the length of hospital stay for patients on IV antimicrobials were found to be late IV to PO antimicrobial switch (delay by 48 h increase hospital stay by three times (
Predictors for the length of hospital stay for patients on IV antimicrobial, Ethiopia.

ref- is to mean reference.
CI, confidence interval; IV, intravenous; PO, per oral.
Discussion
This study was a 3-month study on 92 sample size with 36 intervention and 56 comparator groups from medical, surgery, and gynecology and obstetrics wards, that was conducted with the aim of assessing the proportion, associations, and outcomes of early IV to PO antimicrobial switch at AURH. Accordingly, the practice of early IV to PO antimicrobial switch was low and it has been contributing to prolonged hospitalization, in-hospital complication, and extra cost to the patients. The first draft of this document was available elsewhere as a preprint. 31
Early IV to PO switch of antimicrobial treatment among hospitalized patients is often neglected owing to time constraints, the inaccurate belief that IV therapy is always superior to oral therapy by physicians and/or low risk of re-infection if IV was continued, unavailability of direct oral equivalent for the IV antimicrobials, and rapid switch in staffing.8,19 According to our study finding, the presence of poly-pharmacy was an independent predictor for lack of early IV to PO antimicrobials switch. This might be attributed to patients with poly-pharmacy being considered by the physician as having a more severe condition that may not respond to PO antimicrobials. In other studies, IV antimicrobials have often continued longer than 72 h unnecessarily in spite of eligibility for early IV to PO switch.3,32 Even though the rate of early IV to PO switch at our hospital fell short of the potential 100% at only 39%, this is in line with other studies. For example, studies of early switch in eligible patients in Switzerland showed a rate of 61%, 9 Lebanon 33%, 3 and Ethiopia 21%. 8 The possible reason for this difference was attributed to the variation in the service provided (clinical pharmacy service and presence of antimicrobial stewardship in Switzerland) which promotes the practice of early IV to PO switch.
As evidenced by different studies, this issue has been contributing to incremental cost, longer hospital stay, risk of thrombophlebitis, and increased risk of hospital-acquired infection (HAI).5,11,33 Indeed, the finding of our study was not far apart from these reports. Thus, if early IV to PO antimicrobial was done for all patients who satisfied the criteria, we can avoid the extra cost for the study participants. This cost was higher than the study report from Jimma University Medical Center (JUMC); 4,080.06 (
This means changing IV to PO alone is not sufficient to reduce the extra cost, but, giving due consideration for the time or as soon as possible early conversion has a significant role in reducing the extra cost of IV antimicrobial. This finding was supported by studies from Brazilian hospitals and Lebanese hospitals (significantly longer IV antimicrobial which may contribute to a higher extra cost).3,34
In addition, the current study findings revealed that the independent predictors for an incremental length of hospital stay were delay IV to PO switch from the time of fulfillment of criteria for IV to PO antimicrobial switch and failure to fulfill IV to PO antimicrobial switch criteria throughout the hospital stay. This study was supported by studies from JUMC and Brazilian hospitals.2,8,34
Among the roles of an antimicrobial stewardship program in the given hospital, reducing HAIs, reducing the duration of IV antimicrobial and consecutively hastening early discharge, reducing extra cost related with unnecessarily prolonged IV antimicrobial, and improving overall treatment outcome of patients diagnosed with the infectious disease were few among the shortlisted activities. Thus, the AURH should aim to establish the functional antimicrobial stewardship program to improve the practice of early IV to PO antimicrobial switch based on the predefined criteria. By doing this, the hospital can avoid the negative consequences of the IV to PO not switched antimicrobials. Moreover, the antimicrobial stewardship program in the hospital is among the five strategies recommended by the World Health Organization and Ethiopian Ministry of health on antimicrobial resistance prevention and containment.27,35
The strength of this study is that it provides data around the practice and outcomes of early IV to PO switch in a resource-limited setting, where data are scarce. The first limitation of this study was a small sample size, selection bias due to non-probability sampling, and being a single center, which affects its generalizability to all Ethiopian University referral/teaching hospitals. Among the factors that affect early clinical stability and early IV to PO antimicrobial switch, one is the severity of the disease 36 including non-communicable diseases. But, the current study failed to assess the severity of the disease by using different validated tools.
Conclusion
The current study finding revealed that, even though the majority of the study participants have had early clinical stability, the practice of early IV to PO antimicrobial switch was low. Poly-pharmacy and acid suppressant use were the predictors for failure to early IV to PO switch of antimicrobials. This practice has been contributing to unnecessary costs and prolonged hospital stay. There was a significant difference between the intervention and comparator groups in terms of length of hospital stay, in-hospital complication, and extra cost. This shows an important gap in the rational use of antimicrobial therapy and the need for an intervention that promote the practice of early IV to PO switch.
Supplemental Material
sj-docx-1-smo-10.1177_20503121231161192 – Supplemental material for Opportunities, associations, and impact of early intravenous to oral antimicrobial switch for hospitalized patients in Ethiopia
Supplemental material, sj-docx-1-smo-10.1177_20503121231161192 for Opportunities, associations, and impact of early intravenous to oral antimicrobial switch for hospitalized patients in Ethiopia by Gosaye Mekonen Tefera, Tesemma Sileshi, Mekonnen Derese Mekete and Gurmu Tesfaye Umeta in SAGE Open Medicine
Footnotes
Acknowledgements
We would like to express our deepest gratitude to our study participants and AURH staff for their co-operation and genuine information.
Authors contributions
Tefera GM and Mekete MD: conceived the study, participated in its design, and project management. Mekete MD: Done the data collection and statistical analysis. Sileshi T, Umeta GT, Mekete MD, and Tefera GM: contributed to study design and statistical analysis. Tefera GM and Sileshi T contributed to drafting of the manuscript, study design, and statistical analysis. All authors read and approved the final manuscript.
Availability of data and material
The data used for analysis and report of this manuscript were available from corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
The ethical clearance with ref. number of AU/Phar/117/2019 was obtained from Ambo University College of Medicine and Health sciences school of Pharmacy. Written informed consent was obtained from the study participants and/or parents or guardian for ages of 15–17 years. Confidentiality was secured to protect the privacy of the participants.
Informed consent
Written informed consent was obtained from the study participants and/or parents or guardian for ages of 15–17 years.
Trial registration
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
