Abstract
Objectives:
Over the past decade, concerning trends in antimicrobial resistance have emerged in Southern Africa. Given a paucity of pediatric data, our objectives were to (1) describe antibiotic utilization trends at a national referral center in Southern Africa and (2) assess the proportion of patients receiving antibiotics appropriately. In addition, risk factors for inappropriate use were explored.
Methods:
We performed a prospective cohort study on medical and surgical pediatric patients aged below 13 years admitted to the country’s tertiary care referral hospital in Gaborone, Botswana. We collected demographics, clinical, laboratory, and microbiology details, in addition to information on antibiotic use. We separately categorized antibiotic prescriptions using the World Health Organization AWaRe Classification of Access, Watch, and Restrict.
Results:
Our final cohort of 299 patients was 44% female and 27% HIV-exposed; most (68%) were admitted to the General Pediatrics ward. Infections were a common cause of hospitalization in 29% of the cohort. Almost half of our cohort were prescribed at least one antibiotic during their stay, including 40% on admission; almost half (47%) of these prescriptions were deemed appropriate. At the time of discharge, 52 (21%) patients were prescribed an antibiotic, of which 37% were appropriate. Of all antibiotics prescribed, 42% were from the World Health Organization Access antibiotic list, 58% were from the Watch antibiotic list, and 0% were prescribed antibiotics from the Restrict antibiotic list. Univariate analyses revealed that surgical patients were significantly more likely to have inappropriate antibiotics prescribed on admission. Patients who were treated for diseases for which there was a clinical pathway, or who had blood cultures sent at the time of admission were less likely to have inappropriate antibiotics prescribed. On multivariate analysis, apart from admission to the surgical unit, there were no independent predictors for inappropriate antibiotic use, although there was a trend for critically ill patients to receive inappropriate antibiotics.
Conclusion:
Our study reveals high rates of antibiotic consumption, much of which was inappropriate. Promising areas for antimicrobial stewardship interventions include (1) standardization of management approaches in the pediatric surgical population and (2) the implementation of feasible and generalizable clinical pathways in this tertiary care facility.
Introduction
Antimicrobial resistance (AMR) is a global threat with alarming implications, including morbidity, mortality, and costs. 1 Botswana struggles with this problem, as evidenced by rising AMR rates that are well documented in its pediatric population. 2 Over the past decade, the country has experienced a rapid rise in the rates of methicillin-resistant Staphylococcus aureus (MRSA) infections 3 and the prevalence of multiply resistant gram-negative organisms among patients with urinary and enteric tract infections.4,5
Throughout Southern Africa (SA), including Botswana, data reveal the emerging threat of drug-resistant organisms. A study describing the prevalence and trends of MRSA in hospitalized patients in the Republic of South Africa (RSA) detected methicillin resistance in 1231/ 2709 (46%) isolates of S. aureus, with the highest rates measured in children below 5 years of age. 6 A global surveillance study reported that RSA was in the top 10 countries for the prevalence of beta-lactamase production among Enterobacterales isolates and Pseudomonas aeruginosa. 7 Another RSA study revealed that 42% of Salmonella spp. isolates were multidrug-resistant. 8 Such trends have substantial implications for the antibiotic regimens recommended for empiric and definitive therapy of pediatric patients in this region.9,10
A retrospective observational study of antibiotics use in a cohort of pediatric medical admitted to Princess Marina Hospital (PMH), Gaborone, Botswana 11 revealed that the duration of therapy prescribed for laboratory-confirmed infections was excessively long, with patients receiving up to 1.5 times the recommended standard length of therapy. More alarmingly, this study demonstrated that patients who did not have a confirmed infection were prescribed antibiotics for equally long durations of time as those who did. 11 More recent national data have revealed concerning trends in both the inpatient and outpatient realm. A 2017 point prevalence investigation revealed that almost three-quarters of inpatients at 10 hospitals in Botswana received antibiotics at some point in their hospitalization. 10 A recent cross-sectional study of primary healthcare facilities throughout Botswana revealed the large burden of infectious diseases in Botswana nationally, with 42.7% of all prescriptions being for antibiotics, a rate higher that the World Health Organization (WHO) recommendation of < 30%. 12 However, this cohort included a high burden of gynecological and sexually transmitted diseases, and thus not fully generalizable to the pediatric population. With the continued spread of AMR and rising rates of antibiotic consumption worldwide, 13 it is imperative to benchmark the current status of antibiotic utilization of hospitalized children to identify optimal targets for interventions in settings where resources are limited. Here, we describe the first prospective hospital-wide study on pediatric antibiotic use in hospitalized children in Botswana.
Methods
Study design, location, and population
From June to September 2018, we conducted a prospective cohort study of pediatric patients admitted to PMH, the country’s tertiary care referral hospital, in Gaborone, Botswana. All children less than 13 years of age who were admitted to the medical or surgical wards were eligible for inclusion. We excluded children above the age of 13 years as they are admitted to the adult wards. We also excluded neonates as they are admitted to the neonatal unit. The study was approved by the Institutional Review Boards of Princess Marina Hospital, Botswana Ministry of Health Research and Development Committee, the University of Botswana, the Children’s Hospital of Philadelphia, and the University of Pennsylvania. A local study coordinator reviewed the study with each caregiver and obtained written informed consent in the caregiver’s preferred language of either Setswana or English.
Data collection
After consent was obtained, the following data were collected from the medical record: demographics, clinical details, laboratory, and microbiology results, and antibiotic use including choice of agent, indication, dose, interval, and prescribed or planned duration. Comorbidities were noted; defined as the presence of a chronic disorder or illness documented in the patient’s medical record. Severity of illness score was estimated using the inpatient triage, assessment, and treatment (ITAT) score. 14 By enquiring daily with the lead matron, we also collected information on resource limitations, including drug shortages, blood culture bottle availability, and the presence of appropriate intravenous (IV) access. The primary cause of death was documented in the chart by the lead clinician on duty. These were then categorized into infectious versus non-infectious by the study team on review of charts.
Statistical analysis
Our primary outcome was appropriate antibiotic use at the time of admission, defined as the correct choice and dose of agent for the diagnosis in question. Given the heterogeneity of training of pediatric staff, we anticipated there being a range of appropriate antibiotic use, so we consulted local, national, and international guidelines to assist in this determination. These guidelines included the formulary of the Children’s Hospital of Philadelphia (CHOP), PMH guidelines, Pediatric Lexicomp, South African medical formulary (SAMF), British National Formulary (BNF), Nelson’s and PMH pediatric guidelines “Pink Book.” Antibiotic use was deemed appropriate if it aligned with any of these guidelines. Secondary outcomes included appropriate antibiotic dosing and appropriate antibiotic use on discharge. We separately categorized antibiotic prescriptions using the WHO AWaRe Classification of Access, Watch, and Restrict and tracked proportion of use throughout hospitalization. 15 Associations between clinical factors and our outcome of interest, inappropriate antibiotic use, were assessed for using Fisher’s exact test. p-Values ⩽ 0.05 were considered statistically significant. Both clinically relevant variables and those with association using univariate analysis were then subjected to multivariate analysis, where logistic regression was used to calculate the relative risk (RR) of appropriate antibiotic use on admission adjusted for the other variables included. In addition to analyzing the full cohort, we performed the analysis using a sub-cohort of General Pediatrics participants only. This was due to the finding that all surgical participants had inappropriate antibiotic usage so there was no variability in terms of the primary outcome variable for that group. Stata v15 and SAS v9.4 were used for analyses. Power calculation was not performed as this was an observational study.
Results
Demographics
Our final study cohort included a total of 299 patients (Figure 1), of which 56% were male, 27% were HIV-exposed, and 4.4% were HIV-infected. Most participants were admitted to the General Pediatrics ward (n = 202, 68%) and the remainder to the Pediatric Surgery ward (n = 97, 32%). Only 23 (8%) of the cohort were classified as severely ill with an ITAT score of greater than or equal to 4 (Table 1). Infection was the most common reason for admission, and accounted for 29% of hospitalizations. Of these, pneumonia, sepsis, and gastroenteritis were the most common infectious diagnoses (Table 2). Median length of stay was 5 days (IQR 3–8). Outcome data were confirmed in 259 (87%) cases, and 246/259 (95%) survived and 13/259 (5%) died. However, a proportion of all patients were either transferred to another institution (n = 25, 8%) or had an unknown outcome (n = 15, 5%).

Assembling the study cohort.
Patient characteristics.

BMI: body mass index; ITAT: inpatient triage, assessment, and treatment.
Leading diagnoses on admission.

Antibiotic utilization
Approximately half (n = 147, 49%) of the patients in our cohort were prescribed at least one antibiotic throughout their stay, including 119 (40%) who were prescribed at least one antibiotic on admission. Overall days of therapy (DOT) were 749 per 1000 patient days and DOT was greater for medical as compared to surgical patients (966 per 1000 patient days versus 413 per 1000 patient days). At the time of discharge, 52 (21%) patients were prescribed an antibiotic. Of all antibiotics prescribed, 42% were from the WHO Access antibiotic list, 58% were from the Watch antibiotic list, and 0% were prescribed antibiotics from the Restrict antibiotic list. (Figure 2) The most common antibiotics prescribed were third-generation cephalosporins, including ceftriaxone at a rate of 107 per 1000 patient days and cefotaxime at a rate of 75 per 1000 patient days, and co-trimoxazole at a rate of 69 per 1000 patient days. There was also a significant use of piperacillin/tazobactam and ampicillin, both at a rate of 61 per 1000 patient days (Figure 3).

Rate of antibiotic prescribing per 1000 patients by WHO category.

Total rate of individual antibiotic use per 1000 patient days.
Almost half of the antibiotics prescribed at the time of admission (56/119, 47%) were deemed appropriate. Antibiotic use at the time of discharge was appropriate in 37% of cases. A sub-analysis exploring dosing alone revealed that it was appropriate in 64% of cases on admission and 58% on discharge. The median duration of antibiotics prescribed at the time of discharge was 7 days (IQR 5–24), deemed appropriate in 61% of cases.
As expected, significantly more patients received antibiotics on admission for infectious diseases (ID) related diagnoses compared to non-ID diagnoses, (73% versus 26%, p < 0.05). In addition, significantly more patients received antibiotics inappropriately for a non-ID as compared to ID diagnosis (65% versus 42%, p < 0.05). Notably, only 58% of patients with an ID diagnosis received appropriate prescribing of antibiotics on admission. The most common indications for prescription of antibiotics were pneumonia, meningitis, and sepsis. Of those with a diagnosis of pneumonia, 39% were prescribed antibiotics inappropriately; most (62%) were determined to be inappropriate due to dosing. Similarly, over one-third of patients with bacterial meningitis (38%) were prescribed antibiotics inappropriately; 67% were inappropriate due to dosing errors. Most children diagnosed with sepsis received appropriate antibiotics; only two cases were inappropriate, one of these due to dosing errors.
We examined additional factors associated with inappropriate antibiotic use. Univariate analyses revealed that surgical patients were significantly more likely to have inappropriate antibiotics prescribed on admission. All surgical patients received inappropriate antibiotics on admission. In univariate analyses, patients who were treated for diseases for which there was a clinical pathway for the suspected diagnosis, or who had blood cultures sent at the time of admission were less likely to have inappropriate antibiotics prescribed. Younger age (<6 months) and presence of a comorbidity were associated with an increased likelihood of receiving appropriate antibiotics in univariate analyses, as did HIV exposure, which trended toward significance (Table 3). Given that the entire surgical cohort received inappropriate antibiotics, we analyzed factors associated with inappropriate prescribing in both the entire cohort and in medical patients only. We did not identify any additional statistically significant predictors of inappropriate antibiotic use, although there was a trend toward an increased risk of inappropriate use among those deemed severely ill on admission (Table 4).
Univariate analyses of factors associated with inappropriate antibiotic use.
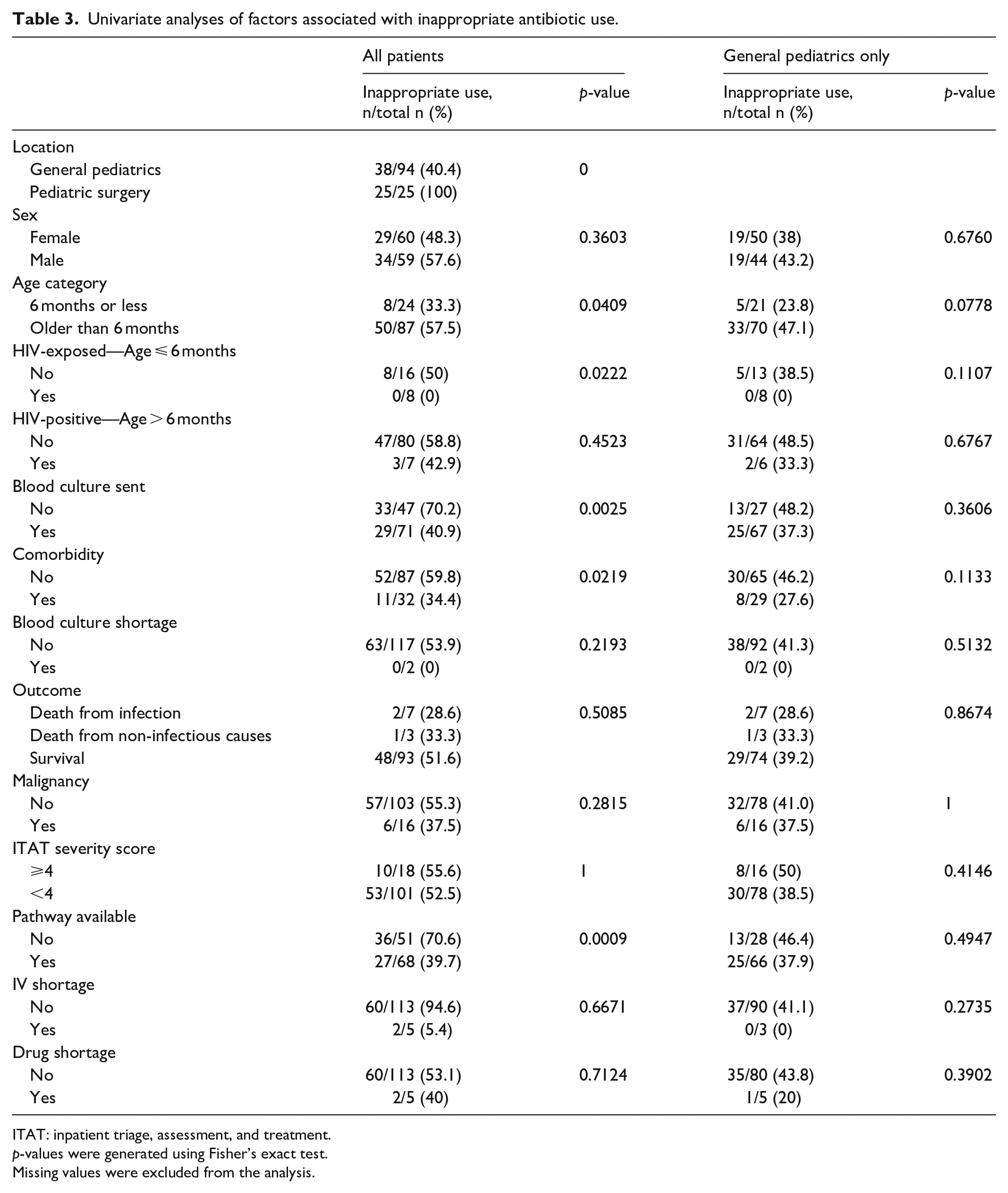
ITAT: inpatient triage, assessment, and treatment.
p-values were generated using Fisher’s exact test.
Missing values were excluded from the analysis.
Multivariate analyses of factors associated with inappropriate antibiotic use.

ITAT: inpatient triage, assessment, and treatment.
Potential impact of resource limitations
We considered the impact of drug shortages on prescribing which was difficult to quantify because the existence of a drug shortage was often verbally reported to the study team, rather than systematically documented by the pharmacy. However, drug shortages affected 11 (4.3%) of the 262 patients for which these data were reported. Additional system-based barriers were noted, including six instances when antibiotics were ordered but not given for unclear reasons. Separately, there were nine instances when IV antibiotics were ordered but not given because the patient did not have an indwelling IV cannula. Blood cultures returned positive in a median of 4 days (IQR 3–5). However, the method of culture reporting varied between laboratory reporting versus verbal reporting. Teams were also frequently unaware of blood culture results and there was lack of documentation of these in the patient’s medical chart. In addition, it was documented that four patients did not have blood cultures sent due to the unavailability of a blood culture bottle.
Discussion
Antibiotic utilization
The Worldwide Antibiotic Resistance and Prescribing in European Children (ARPEC) point prevalence study published in 2016 revealed that 36.7% of hospitalized children received at least one antibiotic during their stay. Notably, only four African countries were represented in this international study including Gambia, Ghana, Malawi, and RSA. 16 Our results reveal that there was significantly more antibiotic consumption in our Botswana cohort where 49% received at least one antibiotic during their stay. While higher than reported in the ARPEC point prevalence study, these findings are additionally supported by the findings from a recent survey performed in Botswana. 10 Here, data from over a quarter (28%) of hospitals in the country were reviewed and it was revealed that over 70% of patients were receiving antimicrobials at the time of data collection. 10 Pediatric patients were included in the patients surveyed, although not separately analyzed. While it must be noted that a high burden of infections was documented in this population, as in similar low- and middle-income countries (LMIC) settings, concerns were highlighted about the variable infrastructure currently in place to improve the standardization of future antibiotic use in these high-risk groups. 10
Pediatric specific data in neighboring countries remain concerning. In Ghana, a multicenter point prevalence study of 10 healthcare facilities revealed that 71% of admitted children and adolescents were prescribed antibiotics. 17 In Zimbabwe, almost all (93.1%) admitted patients were prescribed an antibiotic at least once during their hospital stay. 18 A point prevalence study performed across 18 hospitals in RSA revealed that 49.7% (627/1261) received at least one antibiotic during their stay. 19 In addition, studies from other LMIC settings, including Gambia and Ethiopia, have reported a wide range of antibiotic prescribing patterns of between 54% and 86% of patients during their hospitalization.20,21
Our findings reveal a concerningly high proportion of inappropriate antibiotic use on admission of 53%. In West Africa, a review of prescriptions across 48 health centers in Burkina Faso, it was noted that while children < 5 years of age received antibiotics for the appropriate indication of pneumonia in the majority of cases, there was a significant proportion of likely inappropriate prescribing including for a diagnosis of malaria alone (7.3%), non-bloody diarrhea (20%), and isolated diagnosis of cough (40%). 22 The above-mentioned Zimbabwean study noted that while 93% of patients received antibiotics while admitted, only 58% of these were adherent to national guidelines. 18 Such data strongly argue for the urgent development and implementation of pediatric antimicrobial stewardship programs which we know are lacking in this region of need, despite demonstrating clear improvement in outcomes. 23
Surgical sub-cohort
All patients on the surgical unit in our cohort received antibiotics inappropriately. Almost two-thirds of the cohort 17/26 (65%) had antibiotics prescribed with an incorrect dose on admission. We suspect this was largely due to the lack of documented weight for appropriate dosing as 34/97 (35%) of all patients had no weight documented for their entire stay compared with 2/202 (1%) of the medical cohort. Although we did not capture the use of surgical prophylaxis in the cohort for individual patients, due to often limited documentation, we did note that the median DOT per patient was 8 (IQR 4–11.5), despite the majority of patients being admitted for elective procedures. This, along with the high DOT of 413 per 1000 patient days, argues that this is an area to focus future stewardship efforts. System-based contributors include a lack of clinical pathways in this subset of patients which were noted to be less likely to be associated with the inappropriate use of antibiotics in our univariate analysis. In our surgical cohort, only five patients (5%) had a pathway available for use compared with 109 (54%) in the medical cohort. Despite widespread inappropriate antibiotic use, our surgical cohort was surprisingly medically stable with just 11 (12%) reported to have a comorbidity compared with 73 (36%) of the medical cohort. Previous studies corroborate our findings with data from RSA showing that almost half of a pediatric surgical cohort received antibiotics without a documented indication, with significant variation noted in surgical antibiotic prescribing practices in general. 24 With 80% of pediatric patients routinely receiving antimicrobials for surgical prophylaxis in Africa, our results are generalizable to other LMIC in SA and highlight the urgent need for stewardship interventions in this high-risk group. 24
Dosing and duration concerns
Dosing was deemed inappropriate in 36% of admission prescriptions and 42% of discharge prescriptions. Data from other LMIC are similar; where in Ethiopia, 18.2% of patients received inappropriate antibiotics due to the incorrect dose and 10.1% receiving the incorrect duration. 25 A cross-sectional study performed in a large teaching hospital in Northern Ghana revealed a high proportion of prescription errors where almost 30% received the incorrect duration, a large proportion of these pediatric patients. 26
A systematic study exploring the appropriateness of ceftriaxone revealed that across Africa, three in five patients received inappropriate prescribing, from either dose, duration, or frequency. 27 As is similar across the continent, our cohort received a high proportion of ceftriaxone prescribing given the most common infectious indications of pneumonia, sepsis, and meningitis, with a total rate of prescribing of 103 per 1000 patient days (Figure 3). Overall, 39% of patients received this medication inappropriately.
There are many reasons for dosing and duration concerns in LMIC that may seem out of our control, including variability in training of providers and familiarity with guidelines. However, other factors are potentially modifiable. As previously discussed, in many cases in our cohort (n = 36, 12%), the exact weight of the child was not documented in the chart, making accurate weight-based dosing impossible. There was often a lack of child-appropriate formulary preparations available and no clear discharge duration which we hypothesize contributed to the increased median duration of antibiotics that was 7 days (IQR 5–24), deemed appropriate in 61% of cases. These findings underscore the need for dedicated antimicrobial stewardship (ASP) or pediatric clinical pharmacy expertise and laboratory support and funding to ensure a standardized approach that is generalizable and feasible. 28
Pathways and comorbidities
Our univariate analyses noted significantly improved antibiotic use when a pathway was available for the diagnosed condition. Similarly, the presence of HIV exposure or a comorbidity was less likely to be associated with inappropriate antibiotic use, as did sending a blood culture at the time of admission. When stratified by location and a sub-analysis was performed on the General Pediatric cohort alone, results did not remain statistically significant; however, overall trends were similar. We therefore hypothesize that pre-existing mental models of known conditions allow more rapid and accurate clinical reasoning in these patients, leading to more streamlined appropriate antibiotic use. Previous studies have highlighted that the use of clinical pathways contributes toward both reduced unnecessary antibiotic use 29 and improved patient outcomes. 30 Given the severe burden of infectious diseases in LMIC, including in our cohort where almost one-third had an infection as their primary diagnosis, we feel clinical pathways are potentially under-utilized in LMIC settings, particularly in surgical populations, and offer the potential to offload the mental burden particularly associated with staffing shortages and different educational backgrounds. A previous study exploring factors associated with compliance with treatment guidelines highlighted the need to tailor guidelines for the facility at hand, keeping in mind the accessibility of both the guideline and the recommended antibiotic agents, the requirement for evidence-based up-to-date guidance, the degree to which it is user-friendly, and the incorporation of key personnel including nursing and pharmacists. 31
Drug shortages/barriers
Drug shortages remain a continuing significant global issue and are of major concern in LMIC where patients are unable to access suitable first- and second-line agents which may lead to unnecessary, inappropriate, or no antibiotic exposure. 13 Access to appropriate first-line appropriately used antibiotics have well demonstrated benefits to population health and reduction in child mortality. 32 While likely underestimated, drug shortages were present in at least 4.3% of our cohort. We strongly suspect that a significant proportion of our inappropriate prescribing could have been mitigated with consistent access to key first-line agents or formulations, or with combination therapy of available agents at the time of prescribing. In-depth knowledge of the spectra of available antibiotics and of effective combinations of second-line agents is a key part of any ASP initiative but one that will need to be expanded to LMIC settings where we must continue to balance the need for access to appropriate antibiotics against the risk of inappropriately over prescribing.
Limitations
Certain documentation was missing, particularly on the surgical floor where we were often unable to determine whether the indication was prophylactic or therapeutic. Similarly, we collected data on comorbidities with an intention to include body mass index (BMI) as a covariate to infer nutritional status in our cohort. However, due to missing height of many patients, including 76/97 (78%) on the surgical floor, we were unable to reliably document this for the majority of patients. As noted above, power calculation was not performed as this was an observational study. Finally, we acknowledge that this is a single center in Botswana thus many not be generalizable to all SA countries. However, it is the nation’s biggest tertiary care facility with a large catchment area.
Conclusion
In conclusion, our analyses reveal high rates of antibiotic consumption yet significant practice gaps with respect to appropriate antibiotic choice, dose, and duration of therapy, both on admission and discharge, in children in Botswana’s national referral center. Key areas for ASP interventions highlighted include both standardization of approaches in the pediatric surgical population in addition to the implementation of feasible and generalizable clinical pathways in this tertiary care facility. While we acknowledge that this is a single-center study, we hypothesize that many of our findings are generalizable to similar LMIC throughout SA and highlight the urgent need for dedicated antibiotic stewardship in children in these settings.
Supplemental Material
sj-docx-1-smo-10.1177_20503121221104437 – Supplemental material for Assessing antibiotic utilization among pediatric patients in Gaborone, Botswana
Supplemental material, sj-docx-1-smo-10.1177_20503121221104437 for Assessing antibiotic utilization among pediatric patients in Gaborone, Botswana by Eimear Kitt, Molly Hayes, Lance Ballester, Kgotlaetsile B Sewawa, Unami Mulale, Loeto Mazhani, Tonya Arscott-Mills, Susan E Coffin and Andrew P Steenhoff in SAGE Open Medicine
Footnotes
Acknowledgements
This work is dedicated to and inspired by the memory of Dr Melissa Ketunuti. As a compassionate pediatrician and researcher, she worked to improve health for vulnerable children in Botswana and around the world. The authors thank Kristel Emmer, PhD, Kara (Catalyst) Twomey, RN, MPH, and Catherine Sayikanmi, MPH for their assistance in data collection.
Author contributions
E.K., M.H., K.B.S., U.M., L.M., T.A.-M., S.E.C., and A.P.S. contributed to conception and design; E.K., M.H., L.B., K.B.S., S.E.C., and A.P.S. contributed to analysis; E.K. drafted the article; M.H., L.B., K.B.S., U.M., L.M., T.A.-M., S.E.C., and A.P.S. critically revised the article. All authors agree to be accountable for all aspects of work ensuring integrity and accuracy, and gave final approval.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Melissa Ketunuti Memorial Global Health Fund. S.E.C. receives research support from Merck unrelated to this project. Otherwise, the authors have no pharmaceutical, industrial, or additional funding to disclose.
Ethical approval
Ethical approval for this study was obtained from (1) Ministry of Health Research and Development Committee, Republic of Botswana. Protocol No. PMH 5/79(403-1-2017); (2) Princess Marina Hospital Ethics Committee Protocol Reference: PMH 2/2A(7)/75; (3) University of Botswana IRB: UBR/RES/IRB/BIO/50; and (4) University of Pennsylvania IRB: IRB PROTOCOL#: 828029.
Informed consent
Written informed consent was obtained from legally authorized representatives before the study.
Data availability
Data are available on request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
