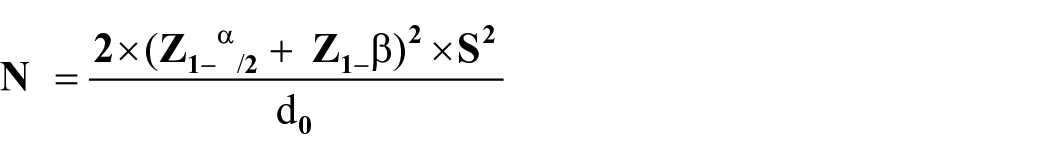Abstract
Objective:
To compare the analgesic efficacy and safety of paracervical block and conscious sedation in the surgical evacuation of the uterus following a first-trimester miscarriage.
Methods:
This is an open-label, randomised control trial comparing pain relief by paracervical block versus conscious sedation during manual vacuum aspiration of first-trimester incomplete miscarriages. One-hundred and sixty-two women were equally randomised into two groups. Group A (paracervical group) received a paracervical block of 4 ml of lidocaine each at the 4 and 8 o’clock positions. Group B (conscious sedation group) received intravenous 30 mg pentazocine and 10 mg diazepam. Data obtained were presented using a frequency table, student t-test, chi-square and Mann–Whitney test and bar chart. A P-value of ⩽0.05 was taken to be statistically significant.
Results:
The mean pain score was lower in the paracervical block group compared to the conscious sedation group (P < 0.05); however, there was no significant difference in women’s satisfaction between the groups (P ⩾ 0.05). Conscious sedation had more negative side effects compared to paracervical block which was significant (P < 0.05).
Conclusion:
Paracervical block has better pain control compared with conscious sedation and has a good side effect profile.
Trial registration:
Pan African Clinical Trial Registry (registered trial no. PACTR202108841661192).
Synopsis
Paracervical block for manual vacuum aspiration (MVA) during first-trimester miscarriage is associated with less pain among women compared with conscious sedation.
Keywords
Introduction
Early pregnancy losses are estimated to occur in 10% of all clinically recognised pregnancies with about 80% occurring in the first trimester. 1 The major cause of morbidity and mortality of early pregnancy loss is incomplete miscarriage which occurs due to complications from haemorrhage and infection. It is the third most common cause of maternal mortality in Nigeria. 2 This associated maternal mortality in Nigeria might increase with a yearly miscarriage rate of 25 per 1000 women. 3
Methods available for the management of miscarriage are expectant care, medical and surgical management. 4 A Cochrane review by Neilson et al. showed that of the surgical methods of evacuation, manual vacuum aspiration (MVA), is safe, quick to perform and less painful.5 –7 It is associated with fewer cases of serious complications, reduced need for general anaesthesia, and a shorter duration of hospital stay.6 –8 MVA involves cervical manipulation and uterine suction that results in pain for the patient. 9 About 97% of women who undergo MVA report pain during and after the procedure, 9 as such making the need for good analgesia during and after the procedure essential. Clinicians need to be aware of the safety and efficacy of the different pain control regimens. 10
For MVA before 13 weeks gestational age, a combination of a paracervical block with local analgesia is safe, and intravenous sedation may also be offered.11,12 Calvanche et al., 13 in their systematic review, concluded that what influences the choice of anaesthesia used for incomplete miscarriage is availability, effectiveness, safety, side effects, practitioners’ choice, cost and woman’s preferences of each method while more evidence supporting each method is awaited. Paracervical block involves the injection of a local anaesthetic agent around the cervix. 14 This prevents transmission of visceral afferent nerve impulses from the uterus and cervix through the Frankenhauser’s ganglia. 14 Conscious sedation on the other hand is associated with a minimally depressed level of consciousness as the patient retains the ability to maintain a patent airway independently and continuously, with the ability to respond appropriately to physical stimulation and/or verbal command. 15
Tangsiriwatthana et al., 14 in their review of literature on the use of analgesia during the management of obstetrics and gynaecological conditions, found that no technique provided reliable pain control. There was no evidence that paracervical block reduced differences in pain perception during or after the uterine intervention when compared to mefenamic acid or pethidine and diazepam or midazolam and fentanyl. 14 On the contrary, Kan et al. 16 reported good pain control using midazolam and fentanyl. Gazvani et al. 17 reported that clients managed with conscious sedation were more satisfied with pain management than those that had patient-controlled anaesthesia. Agostini et al., 18 in France, in their study involving 114 women, compared ropivacaine and lidocaine for a paracervical block during a surgical abortion. They found that the mean intraoperative pain for both groups was 5.23 ± 2.72 vs 4.18 ± 2.77. There was no statistically significant difference. Calvanche et al., 13 in their Cochrane review of trials that compared paracervical block and sedation/analgesia, reported that paracervical block did not improve postoperative pain when compared with the control. The comparison revealed a high clinical heterogeneity and as such trials were not combined. Nafisa 19 in Bangladesh reported a satisfactory outcome with paracervical block in the management of incomplete miscarriage. Because of the conflicting reports on the adequate form of analgesia during MVA, this study was conceived to help fill a knowledge gap, especially in sub-Saharan Africa including Nigeria where there is a paucity of a report on adequate analgesia during MVA. This study is aimed at comparing the analgesic efficacy and safety of paracervical block and conscious sedation in the surgical evacuation of the uterus following a first-trimester miscarriage. Findings from the study will help to improve care and assist in counselling women on the choice of analgesia which will help maternal satisfaction.
Materials and methods
Study design
This is a single-centre open-label randomised controlled trial that was carried out between March 2020 and January 2021 in Alex Ekwueme Federal University Teaching Hospital Abakaliki, Ebonyi State, Nigeria (AEFUTHA).
Study background
Ebonyi state is one of the five (5) states in the South-Eastern part of Nigeria with a population of 2,176,947. 20 The capital of Ebonyi state is Abakaliki. The population of Abakaliki is about 149,683, 20 made up of inhabitants of people from different ethnic groups but predominantly Igbos. The inhabitants are mostly peasant farmers and traders. There are primary, secondary, tertiary, mission and privately owned hospitals in the state which provide health care services for the patients. The study was conducted in the Department of Obstetrics and Gynaecology of AEFUTHA. AEFUTHA is the only tertiary institution in the state and caters for both primary and referral cases from Ebonyi state and environs. It has an emergency unit that provides 24 h/7 days services for obstetrics and gynaecology emergencies. Women diagnosed with incomplete miscarriage are offered manual vacuum aspiration or medical care using misoprostol to complete the miscarriage process.
Study population
The study population were pregnant women presenting with incomplete miscarriage in the first trimester. A diagnosis of incomplete miscarriage was made in a woman with a gestational age of ⩽13 weeks who presented with a history of profuse bleeding per vaginam with or without passage of product of conception. Vaginal examination showing cervical Os dilatation of 2 cm or more with the product of conception extruding from the cervical Os. Bedside ultrasound confirming retained product of conception. The above were vital to help rule out complete miscarriage or threatened miscarriage.
Sample size calculation
The sample size was calculated using the formula for an equivalence, randomised controlled trial (continuous variables) as stated by Zhong 21
where Z = 1.96, β = 0.845; ∞ = 0.05; S is thepolled standard deviation of the means (outcome of interest) 52.4 mm;18,22 and d
Participants per group were 81 after the addition of a 10% attrition rate. The total sample size for the study was 162.
Inclusion and exclusion criteria
Women included in the study were women with the diagnosis of incomplete miscarriage at a gestational age of ⩽13 weeks of gestation. Those excluded were patients with a known history of an allergy to the drugs, psychiatric or neurological diseases, those with active pelvic inflammatory disease and those who refused to give informed consent.
Recruitment
Participants with first-trimester incomplete miscarriages who presented at the Gynaecology emergency unit were recruited using a systematic sampling method with an interval of one. They were thereafter allocated to either paracervical block or conscious sedation group based on the randomisation. Consecutive women who met the inclusion criteria were independently recruited into the study groups until the sample size was met. Ethical standards were maintained throughout the study. Participants were followed up till 24 h after the procedure. Face masks were made available for the participants and the team of researchers to help prevent the transmission of COVID-19 disease.
Study procedure and randomisation
Procedure for randomisation
Randomisation for the study was done by a computer-generated random number using the software Research Randomizer®. Using the software, 81 numbers were randomly generated from a pool of 162 numbers. These first 81 randomly generated numbers were assigned to group A (paracervical block group). The remaining 81 numbers were automatically assigned to group B (conscious sedation group). The numbers with the alphabet of the group allocation (A or B) written by its side (example 1A or 4B etc) were each put in brown paper envelopes, then sealed and put together in a locked container. Participants that met the inclusion criteria and signed the informed consent form were asked to pick one envelope from a container by research assistants. The envelope was thereafter opened by the research assistant to note which arm of the study the participant was allocated to. Group A received 2 ml at the anterior lip of the cervix and 4 ml of 1% lidocaine, for each block at the 4 and 8 o’clock positions. Group B received 30 mg of pentazocine hydrochloride and 10 mg of diazepam intravenously for conscious sedation.
Study procedure
Group A (paracervical block)
Participants allocated to group A received 4 mL of 1% lidocaine, for each block at the 4 and 8 o’clock positions and 2 ml at the anterior lip of the cervix. Participant in this group was placed in the lithotomy position; the perineum was cleaned with a Savlon swab and was draped. The bladder was emptied. A Cusco’s speculum was inserted into the vagina to allow complete visualisation of the cervix. Products of conception and blood obscuring vision were wiped away. The 4 and 8 o’clock positions at the junction of the cervix and vagina were identified. After which, a 10-ml syringe was used to draw up 10 ml of 1% lidocaine solution (equivalent to 100 mg). A 22-gauge spinal needle was connected to the syringe, and then 2 ml was injected superficially at the anterior lip of the cervix after about 2 min, it was held by the sponge holding forceps at the 12 o’clock position. Slight traction was applied to move the cervix to define the transition of the smooth cervical epithelium to vagina tissue. This reflection marks the line of injection. About 4 ml each was injected at the 4 and 8 o’clock positions slowly for 60 s at a depth of 3–7 mm. The syringe’s plunger was withdrawn first to ensure that the needle was not in a vessel. Once the block was given, the Cusco’s speculum was withdrawn and waited for 5 min before commencing the procedure. Participants’ vital signs before, during and after the procedure were checked and recorded. Also, the maximum pulse rate and minimum oxygen saturation during the procedure were recorded.
Group B (conscious sedation)
Participants allocated to group B received intravenous 30 mg pentazocine hydrochloride and 10 mg diazepam. There was a preoperative assessment to ascertain the participant’s fitness as well. Intravenous access was secured with an 18G cannula, and 10 mg diazepam was administered first for over 1 min. Thereafter, 30 mg pentazocine hydrochloride was administered over 1 min and a latency period of 2 min was allowed before the commencement of the procedure for the onset of analgesic function. The participant’s pulse, blood pressure and oxygen saturation were monitored all through the procedure using a pulse oximeter and the values of blood pressure, maximum pulse rate and lowest partial pressure of oxygen were recorded. The vital signs were measured during, within and after the procedure.
Resuscitation tray containing hydrocortisone, promethazine for vomiting, ephedrine, diazepam for aborting seizures, amiodarone for arrhythmias, bag-mask-valve device, drip giving set, syringes and needle, cannula, and normal saline and ringers lactate, laryngoscope, and an endotracheal tube was made available. Oxygen cylinder and rescue analgesia in form of rectal diclofenac were made available. Investigations done for each participant include pack cell volume, blood grouping/typing of blood and retroviral screening after due counselling.
Manual vacuum aspiration
The participant was placed in a lithotomy position with an intravenous infusion of normal saline into which 20 IU oxytocin was added. The surgeon wore a mask, scrubbed, and donned sterile gowns and gloves. A metal catheter was passed into the urethra to empty the bladder; thereafter, bimanual examination was performed to determine the uterine size and position. A Cusco’s speculum was inserted into the vagina to visualise the cervix. The anterior lip of the cervix was grasped using a sponge holding forceps and any products of conception at the cervical Os were removed with an ovum forceps. Afterwards, an appropriate sized and already charged Karman’s syringe was inserted through, a non-touch technique, into the uterine cavity through the cervix and a to and fro movement with both clockwise and anticlockwise rotation of the Karman’s syringe was done until the uterus was empty. This was evidenced by the uterus hugging the cannula, feeling of gritty sensation, foaming blood and no more products entering the chamber of the Karman’s syringe. The cannula was then removed, and the patient was cleaned up, and 600 mcg of misoprostol was inserted per rectum to keep the uterus contracted. Participants’ vital signs were monitored quarter-hourly until stable.
Pain assessment
A research assistant who did not know the type of anaesthesia administered to the participant was made to assess the pain experience and side effects experienced following the use of the analgesic agents at 10 min, 120 min, and 24 h after the procedure. The pain was assessed using a numeric rating scale (NRS), 23 while the side effects profile information was obtained with a data collection form. Participants’ satisfaction score was obtained using the Victoria satisfaction score 24 which was formulated to suit this study and represented on a 5-point Likert-type scale. 25 Please see the attached Supplementary Material (Suppl 1).
The NRS is a continuous scale comprised of a horizontal line, usually numbered 0–10 CM (100 mm) in length, anchored by three verbal descriptors, no pain, mild, moderate pain and severe pain. It was self-completed by the participant or with the help of the research assistant after the women were educated on its use.
Participants follow-up
Participants were reviewed and assessed before discharge. Blood pressure, pulse rate, temperature, packed cell volume and vaginal bleeding were checked, and the participants were allowed home when stable. However, she was counselled to represent to the hospital if bleeding continued if fever developed or if she had a foul-smelling vaginal discharge. Participants were given antibiotics and haematinics. Contraception was discussed with the participants, and they were referred to the family planning clinic.
Outcome measures
Primary outcome
The primary outcome measure was the mean pain score between the paracervical block group and the conscious sedation group.
Secondary outcome
The secondary outcome measures were the incidence of complications/side effects and participants’ satisfaction.
Statistical analysis
Data were analysed with SPSS, version 23.0 (2015, SPSS Inc., Chicago IL, USA) and by the concept of intention-to-treat analysis which included all randomised participants in the groups to which they were randomly assigned, regardless of the treatment received, adherence with the entry criteria and regardless of withdrawal or protocol deviation. Absolute and relative frequencies of categorical variables, mean, and standard deviation of continuous variables were calculated. The independent t-test was used to compare the mean of continuous outcome variables while chi-square was used to compare groups of categorical variables. Fisher’s exact test was used where chi-square was not suitable. A p value of ←0.05 was taken as significant.
Ethical consideration
Ethical approval for the study was obtained from Health Research Ethics Committee, AEFUTHA. The approval number is FETHA/REC/VOL2/2019/225. The trial number, from Pan African Clinical Trial Registry, is PACTR202108841661192.
Informed consent
A signed written informed consent was obtained from every participant before enrolment into the study
Results
As depicted in Figure 1, 188 cases of incomplete miscarriages were managed during the study period. Twenty-six (26) women were excluded, 6 women refused to consent, while 20 women had their gestational age greater than 13 weeks. The remaining 162 patients were randomised into two groups, making 81 patients in each arm of the study. Of the 162 cases randomised, all met the inclusion criteria and were recruited. None was lost to follow-up, and their data were analysed.

Flow chart of patients through the study.
From Figure 2, conscious sedation had more side effects than the paracervical block group with dizziness 25 (31%) being the most commonly felt side effect. Of all these side effects noted, dizziness, nausea and weakness were significantly more common among women who had conscious sedation than paracervical block for surgical evacuation 25 (31%) versus (vs) 0, 16 (19.8%) vs 0, 22 (27.2%) vs 1 (1.2%), respectively with p value < 0.001.

Side effect of analgesia between the two arms.
Table 1 shows that there was no statistically significance difference in the sociodemographic variables of women between the paracervical block and conscious sedation.
Sociodermographic characteristics of study population.

BMI = body mass index.
In Table 2, there was no statistically significant difference in the mean age, gestational age and parity of participants between groups. The median parity is 2.
Obstetric profile of the study population.
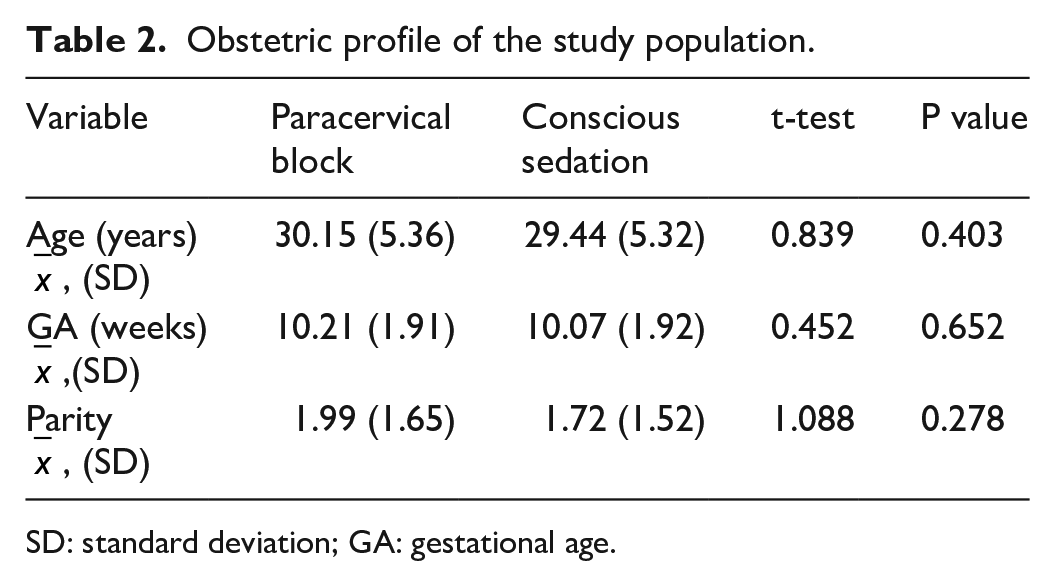
SD: standard deviation; GA: gestational age.
Table 3 shows that 10-min post-evacuation, pain perception were significantly severe (13.6%) and worst (1.2%) among those who had conscious sedation than in those who had paracervical block where none had such pain, p = 0.002. At 120 min (2 h) post–evacuation, there was no significant difference in the pain perception of women who had paracervical block and conscious sedation for surgical evacuation. However, at 24 h post-evacuation, mild pain perception was significantly more in women who had conscious sedation (30.9%) than those who received paracervical block (16%), whereas significantly more participants that had Paracervical block (PCB) (84%) felt no pain when compared with their counterpart that had conscious sedation (69.1%), P = 0.026. Mann–Whitney test was statistically significant at the 10 min and 24 h values with mean rank, (PCB = 70.82, CS = 87.29), U = 2434.500, Z = –3.063, p value = 0.002 and mean rank, (PCB = 75.50, CS = 87.50), U = 2794.500, Z = –2.218, p value = 0.027, respectively.
Comparison of pain scores between treatment groups.

In Table 4, The mean pain score was significantly more among women who had conscious sedation than those who had paracervical block at 10 min (4.99 ± 1.85 versus 4.48 ± 1.33), 2 h (2.38 ± 1.52 versus 1.91 ± 1.45) and 24 h (0.35 ± 0.55 versus 0.16 ± 0.37) with P values of 0.018, 0.046, and 0.013, respectively. These findings were statistically significant.
Mean pain scores between groups.
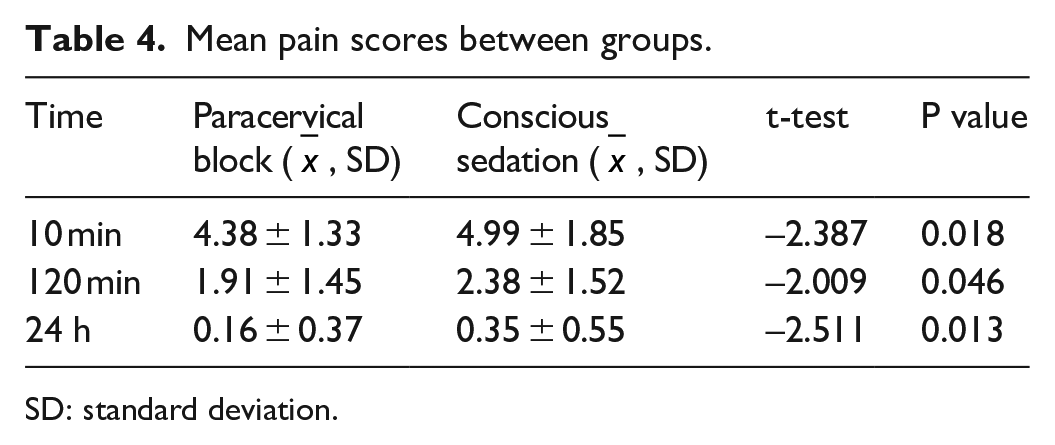
SD: standard deviation.
Table 5 represents women satisfaction on the analgesic efficacy during MVA. There was no significant difference among the two groups on pain control.
Participants’ satisfaction with the analgesia.

Discussion
The mean pain score in this study was significantly more in conscious sedation than in paracervical block at 10 min, 2 h, and 24 h post-evacuation of incomplete miscarriage. Only one participant needed supplemental analgesia and that was in the conscious sedation group. This showed that PCB was a better analgesia for surgical evacuation of the uterus among the groups studied. This agreed with the findings by Nafsia. 19 However, this differed from the findings of Gomez et al. 26 who found that there was no statistically significant difference between paracervical block and psychological support. This finding by Gomez et al. may be a result of not injecting the local anaesthetic agent at the anterior lip of the cervix before grabbing a vulsellum which can be a source of pain. Also, the Cochrane review by Tangsiriwatthana et al. 14 showed no statistically significant difference between paracervical block and conscious sedation.
From the study, the majority of the participants who received paracervical block had no side effects with only one participant each having vomiting and weakness. However, 30.9% of the participants who received conscious sedation had dizziness, 19.8% had nausea and 27% had a weakness. There was a statistically significant difference when dizziness, nausea, and weakness were compared between the two groups with those in the conscious sedation group having more side effects than those in the paracervical block group. There were no participants in both groups who had double vision, fever, seizure, confusion and tremor, whereas, in the conscious sedation group, 4.9% of participants had blurred vision. The reduced side effect profile in the paracervical group is because local administration of paracervical block acts locally and as such does not cause most of these side effects. This follows the recommendation by the World Health Organization (WHO) 12 on the use of pain relief in the surgical evacuation of the uterus for incomplete miscarriage. On the contrary, the greater side effect in the conscious sedation group could be a result of the systemic effect of the drug and findings are similar to the findings by Wong et al. 22
Participants’ satisfaction was not different between the groups we studied, which is similar to the work of Atashkhoii et al. 27 However, this was in contrast with the findings by Wong et al. 22 , which showed that women’s satisfaction was better with the conscious sedation than in the paracervical block. However, Nafsia 19 found that patient satisfaction was better with paracervical block. This could be because both arms in the study by Atashkhoii et al. 27 received paracervical block before the administration of conscious sedation in one arm where its effect could be synergistic while the finding by Nafsia 19 could be because paracervical block was compared with placebo.
Limitations of the study
The study is an open-label study and could be a source of bias, however, participants’ allotment to the groups was at random and pain assessment was done by a different researcher blinded to the study protocol. Pentazocine that was used in the control group could cause dizziness among the women and could interfere with proper pain assessment among the group at 10 min. An effort was however made to obtain a mean pain score among women who had dizziness. Social desirability bias could also be a confounding factor in proper pain report, and to reduce it, participants were counselled on the need to accurately report the level of pain that they perceived using NRS and that reporting will have nothing to do with them as their names were not written on the proforma. This study was done only in participants with first-trimester miscarriage; findings may not be extrapolated to include patients with second-trimester incomplete miscarriages. The short time frame for the pain assessment could affect the ability of these women to give a proper account. This problem was circumvented by assessing pain at three different times.
Conclusion
This study shows that the mean pain scores for the paracervical block group were lower than the control group that had conscious sedation. Unfavourable side effects were more common in the control group. There was no statistically significant difference in participants’ satisfaction between the two groups. We recommended that paracervical block with lidocaine should be used as analgesia during the surgical evacuation of the uterus in patients who have a first-trimester incomplete miscarriage.
Supplemental Material
sj-docx-1-smo-10.1177_20503121221113227 – Supplemental material for Analgesic efficacy and safety of paracervical block versus conscious sedation in the surgical evacuation of the uterus following first-trimester incomplete miscarriages: A randomised controlled trial
Supplemental material, sj-docx-1-smo-10.1177_20503121221113227 for Analgesic efficacy and safety of paracervical block versus conscious sedation in the surgical evacuation of the uterus following first-trimester incomplete miscarriages: A randomised controlled trial by Nnenna A Nweke, Chidebe Christian Anikwe, Richard L Ewah, Osita S Umeononihu and Justus N Eze in SAGE Open Medicine
Footnotes
Author contributions
N.A.N. participated in the study design, data collection/analysis, interpretation of findings and drafting of the manuscript. C.C.A. participated in study design, data analysis, interpretation of findings, and drafting of the manuscript. O.S.U. and C.C.I. participated in the data analysis and the interpretation of findings, R.L.E. and J.N.E. participated in the interpretation of findings and drafting of the manuscript. All participated in the review of the final manuscript. All the authors approved the manuscript.
Availability of data and material
All data generated or analysed during this study are included in this published article.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare that they have no competing interests. To avoid conflict of interest, the study population was randomly selected and allotted to each group that was sealed in an opaque envelope. Pain assessment was carried out by a trained research assistant separate from the authors. The medications used were sourced by the hospital pharmacist independent of the authors.
Ethics approval and consent to participate
Ethical approval for this study was obtained from Health Research Ethics Committee, Alex Ekwueme Federal University Teaching Hospital Abakaliki (FETHA/REC/VOL2/2019/225).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding is borne by the authors.
Informed consent
Written informed consent was obtained from all subjects before the study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.