Abstract
Objective:
Etonogestrel sub-dermal implant discontinuation is very common in the study area. But evidence on the determinants was limited. So, this study aimed to identify determinants of early discontinuation of etonogestrel sub-dermal implant among reproductive-age women in Legambo district, Northeast Ethiopia.
Methods:
An institution-based unmatched case–control study was conducted on 252 reproductive-age women (84 cases and 168 controls) from 1 February up to 30 April 2020. Systematic random sampling was used to select women, and the data were collected by face-to-face interview using a structured, pretested, and interviewer-administered questionnaire. The collected data were entered into Epi Data version 3.1 and analyzed by SPSS version 23. A binary logistic regression model was used to identify determinants of early discontinuation of etonogestrel sub-dermal implant. Statistical significance was declared at a p value of less than 0.05, and adjusted odds ratio with a 95% confidence interval was used to identify determinants of early discontinuation of etonogestrel sub-dermal implant in the final model.
Result:
In this study, not receiving pre-insertion counseling (adjusted odds ratio = 3.19, 95% confidence interval: (1.61, 6.30)), having a history of abortion (adjusted odds ratio = 2.89, 95% confidence interval: (1.50, 5.54)), contraceptive side effects (adjusted odds ratio = 2.45, 95% confidence interval: (1.99, 3.91)), and not receiving an appointment for a follow-up visit at the time of insertion (adjusted odds ratio = 3.45, 95% confidence interval: (1.89, 5.99)) were determinants of early discontinuation of etonogestrel sub-dermal implant.
Conclusion:
Not receiving pre-insertion counseling, having a history of abortion, contraceptive side effects, and not receiving an appointment for a follow-up visit at the time of insertion were predictors of early discontinuation of etonogestrel sub-dermal implant. Thus, healthcare professionals should give an appointment for a follow-up visit at the time of insertion and provide detailed counseling for all women, not only for those who had had an abortion and contraceptive side effects.
Introduction
Etonogestrel sub-dermal implant is a highly effective long-acting and reversible sub-dermal contraceptive implant with a length of 40 mm and a diameter of 2 mm delivering 68 mg of etonogestrel at a dose sufficient to suppress ovulation in every cycle throughout the 3 years of use.1,2 This single-rod progesterone-releasing contraceptive method is 99% effective in preventing unintended pregnancy within 3 years of use compared to other short-acting contraceptive methods. 3 It was developed due to the need to reduce some of the problems associated with six implant systems and inhibit ovulation within 1 day of insertion. 4 It also requires little user compliance and is associated with prompt fertility return after removal. 5
Globally, more than 4.5 million women have used etonogestrel sub-dermal implant. 6 However, despite its good tolerance, reversibility, and effectiveness, 18%–63% of women in developing countries who are still in need discontinued contraception.7,8 Early etonogestrel sub-dermal implant discontinuation is defined as the removal of the implant less than 3 years of use after the insertion. 9 The discontinuation rate of etonogestrel sub-dermal implant varies across countries. A study conducted in the United States indicated that 25.2% of women discontinue the implant within 10.4 months. 10 Studies conducted in the United Kingdom and Malaysia, the Netherlands, and Jordan also indicated that 23%, 28%, and 45% of women discontinue etonogestrel sub-dermal implant within 1 year of initiating the method, respectively.11–13
The discontinuation rate is significantly higher in Africa. Studies conducted in Egypt, South Africa, Nigeria, Liberia, and Yemen showed that 36, 43%, 19%, 23%, and 69% of women discontinue etonogestrel sub-dermal implant before the completion of 3 years, respectively.11,14–16
Etonogestrel sub-dermal implant is provided free of charge for women in Ethiopia. In addition, the Ethiopian Federal Ministry of Health (FMOH) developed a program to scale up this contraceptive method at the community level to meet the huge unmet need in family planning since the beginning of 2010. 17 Despite this effort, the early discontinuation rate within 12 months increased from 5% in 2011 to 11% in 2016.18,19 In Legambo district, the annual report showed that 11.3 % of reproductive-age women use etonogestrel sub-dermal implant, and among these, 31% of them discontinued before 3 years of use for unknown reasons. 20
Contraceptive discontinuation has a substantial contribution to the occurrence of negative reproductive health outcomes. It places a woman at risk of unintended pregnancy, induced abortions, and ultimately can lead to maternal and child mortality.21–26 It also increases the total fertility rate, reduces educational attainment, and may result in negative social and economic consequences.11,27–30
Socioeconomic factors such as age, marital status, educational status, parity, dissatisfaction with services,31–35 contraceptive side effects,36–39 desire to become pregnant, spouse disapproval, and switching to another method37,38,40,41 were the common factors affecting early discontinuation of etonogestrel sub-dermal implant.
Improving the utilization rate of modern contraceptive methods, in general, and etonogestrel sub-dermal implant, in particular, is of paramount importance for tackling different maternal and child health problems to achieve Sustainable Developmental Goals (SDGs) and Growth and Transformation Plan Two (GTPІІ).42,43 Early discontinuation of etonogestrel sub-dermal implant in Legambo district was higher. But evidence on the determinants was limited. So, this study aimed to identify determinants of early discontinuation of etonogestrel sub-dermal implant among women in Legambo district, Northeast Ethiopia, 2020.
Methods and materials
Study area, period, and design
An institution-based unmatched case–control study was conducted from 25 February to 10 April 2020 in public health facilities of Legambo district. The district is located 623 km away from Bair Dar, the city of Amhara regional state, and 501 km away from Addis Ababa, the capital city of Ethiopia. The district has 40 administrative kebeles with a total population of 203,676. Of these, 29,987 were women of reproductive age. It has one primary hospital and nine health centers.
Population
The source population for the cases and controls: All reproductive-age women who were still using etonogestrel sub-dermal implant at the time of the study.
The study population for the cases: All reproductive-age women who request early removal of etonogestrel sub-dermal implant before 3 years following insertion during the study period.
The study population for the controls: All reproductive-age women who request the removal of etonogestrel sub-dermal implant at three completed years following insertion during the study period.
Inclusion and exclusion criteria
All women who requested early removal of etonogestrel sub-dermal implant before 3 years following insertion in the health facilities during the study period were included as cases and all women who requested the removal of etonogestrel sub-dermal implant at 3 years following insertion in the health facilities during the study period were included as controls. Women who had severe illnesses and were not able to provide the required information were excluded.
Sample size determination and sampling procedure
The sample size was calculated using Epi Info version 7.1 by taking the assumption of early discontinuation of etonogestrel sub-dermal implant among women who did not experience contraceptive side effects as 56.5% based on the study done in Mekelle city, 44 the ratio of exposed to unexposed as 1:2, Adjusted Odds Ratio (AOR) as 2.66, power of 80%, and confidence level of 95%. Thus, the required sample size after adding a 10% non-response rate becomes 252 (84 cases and 168 controls).
Among the 10 health facilities in the Legambo district, 5 of them were selected by simple random sampling method. Then after proportional allocation to the selected health facilities, women were selected using the systematic random sampling technique until reaching the final sample size. Women were selected in every second case, followed by two succeeding controls from their sequence of family planning visits in each health facility during the study period. The random start, which was selected by the lottery method, was the second woman who requested the removal of the etonogestrel sub-dermal implant before the expiry date.
Data collection tools, procedures, and measurements
Data were collected by face-to-face interview using a structured and pretested questionnaire taken from previous similar works of literature. The questionnaire was composed of socio-demographic characteristics, obstetric characteristics, contraceptive-related characteristics, and client-related characteristics.11,31,45,46 One Bachelor of Science (BSc) midwife supervisor and two diploma midwifery and three diploma nurses were assigned for the data collection. Data were collected in the ratio of one case for every two succeeding controls in each health facility until reaching the final sample size.
Early etonogestrel sub-dermal implant discontinuation: removal of etonogestrel sub-dermal implant by the healthcare provider before 3 years following insertion. 31
Knowledge: Women’s level of knowledge toward etonogestrel sub-dermal implant was assessed using seven “yes” or “no” answer questions, that is, Does etonogestrel sub-dermal implant delay pregnancy upon discontinuation? Can it provide effective protection from pregnancy for up to 3 years? Does it require a minor surgical procedure for removal? Does it have any interference with sexual intercourse or desire? Can it be discontinued at any time? Does it require little attention after insertion? Is it inserted in the left upper arm? It was measured as “having good knowledge” if respondents scored at or above median value and “having poor knowledge” if respondents scored below the median value.47,48
Attitude: Attitude toward etonogestrel sub-dermal implant was assessed using six questions, that is, etonogestrel sub-dermal implant insertion and removal are highly painful, it has severe side effects, using etonogestrel implant restricts normal activities, etonogestrel sub-dermal implant causes severe changes in menstrual bleeding pattern, it is difficult to remove etonogestrel sub-dermal implant, and etonogestrel sub-dermal implant requires frequent follow-up. Each question has five response options taking a score between 1 and 5 (1 = strongly disagree, 2 = disagree, 3 = neutral, 4 = agree, and 5 = strongly agree). Since the data were not normally distributed, the median score was used as a cut-off point to categorize women’s attitudes toward etonogestrel sub-dermal implant. As a result, those women who scored at or above the median were considered to have a positive attitude and those women who scored below the median were considered to have a negative attitude toward Implanon.47,48
Data quality control
To ensure consistency, the questionnaire developed in English was translated into Amharic language and again translated back to English by language experts. The supervisor and data collectors were trained for 1 day on the objective of the study, the contents of the questionnaire, and the data collection procedure. The questionnaire was pretested on 5% (13) of women (4 cases and 9 controls) at Genete Health Center, one of the health centers in the district, and based on the feedback obtained from the pretest, the necessary modification was done accordingly. During the study period, the collected data were checked continuously on a daily basis for completeness by the principal investigator and supervisor in the respective health facilities.
Data processing and analysis
The collected data were cleaned, coded, and entered into Epi Data version 3.1 and exported to Statistical Package for Social Science (SPSS) version 23 statistical software for statistical analysis. Descriptive statistics, such as frequencies, proportion, and summary measures (median with Inter-Quartile Range (IQR)), were computed. The association between independent variables and early discontinuation of etonogestrel sub-dermal implant was tested using a binary logistic regression model, and all independent variables having a p-value of less than or equal to 0.2 were included in the multivariable logistic regression model. Multicollinearity was checked using standard error, and the Hosmer and Lemeshow test was used to check model fitness. Statistical significance was declared using Odds Ratios (ORs) with 95% CIs and p-value; p-value less than 0.05 and AOR with 95% CI non-inclusive of 1 were considered as statistically significant and used to determine predictors of early etonogestrel sub-dermal implant discontinuation in the final model. Besides, survival analysis was carried out to see the effect of some important variables on early discontinuation of etonogestrel sub-dermal implant.
Results
Socio-demographic characteristics
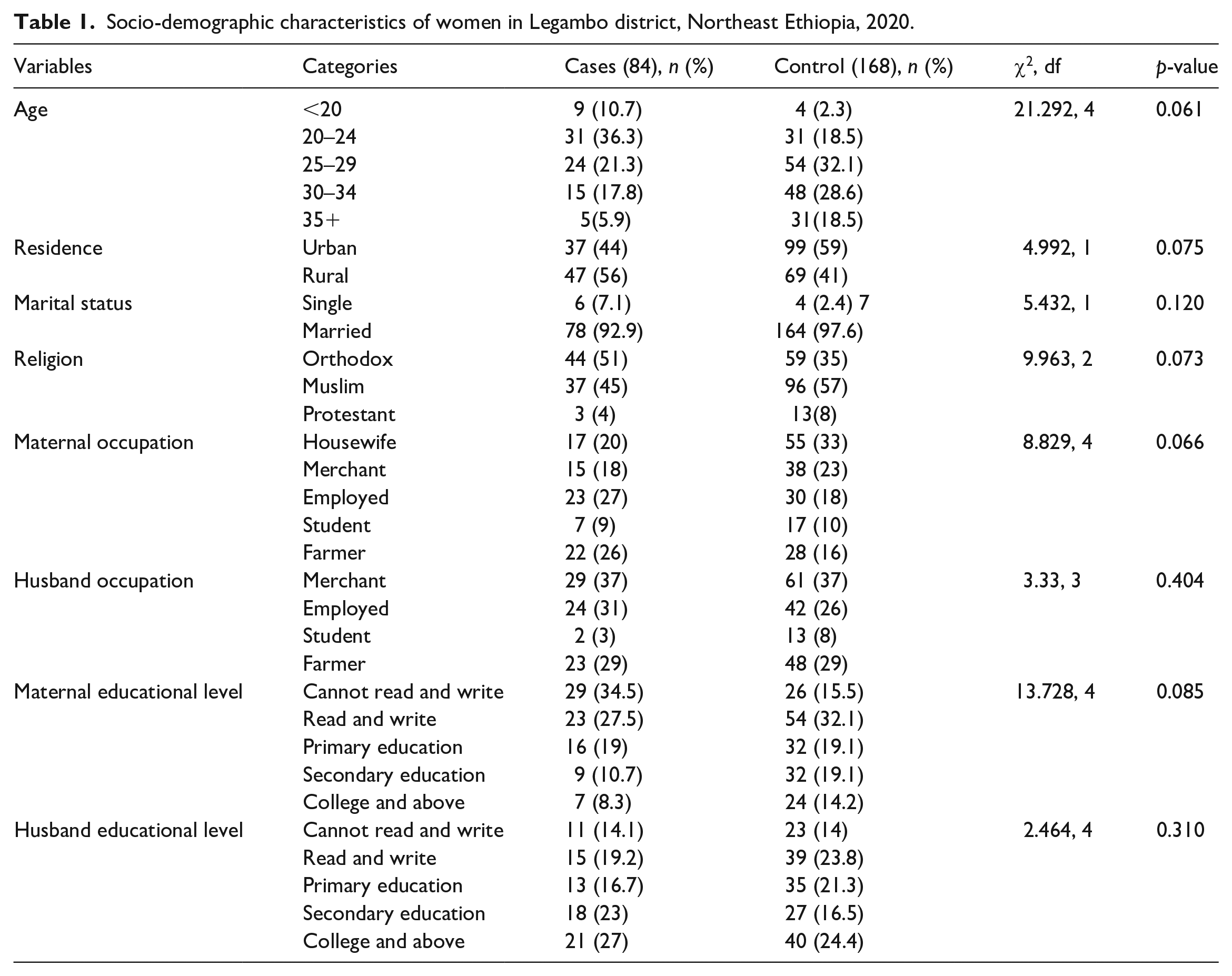
A total of 252 respondents (84 cases and 168 controls) have participated in the study with a response rate of 100%. The median age of the study participants was 27 years with an IQR of 8 years. The median (IQR) duration of etonogestrel sub-dermal implant usage among cases was 18 (10) months, with the duration ranging from 5 to 33 months. Most study participants in this study were married (Table 1).
Socio-demographic characteristics of women in Legambo district, Northeast Ethiopia, 2020.
Obstetric, contraceptive, and client-related characteristics
The majority of study participants were multiparous and had less than or equal to three children. Many of them have been counseled about the side effects of etonogestrel sub-dermal implant, and most of them have good knowledge and a positive attitude toward etonogestrel sub-dermal implant (Table 2).
Women obstetric, contraceptive, and client-related characteristics living in Legambo district, Northeast Ethiopia, 2020.

HEW: health extension workers.
Determinants of early discontinuation of etonogestrel sub-dermal implant
Both bivariable and multivariable logistic regression analyses were done. As a result, history of abortion, contraceptive side effects, not receiving pre-insertion counseling, and not receiving an appointment for a follow-up visit at the time of insertion were factors significantly associated with early etonogestrel sub-dermal implant discontinuation.
Those women who had a history of abortion were almost 3 times more likely to discontinue etonogestrel sub-dermal implant early than those women who had no history of abortion (AOR = 2.89; 95% CI: (1.50, 5.54)). The study also revealed that the odds of discontinuing etonogestrel sub-dermal implant early were 2.5 times higher among women who experienced contraceptive side effects as compared to their counterparts (AOR = 2.45, 95% CI: (1.99, 3.91)). Women who have not received pre-insertion counseling about etonogestrel sub-dermal implant were 3.2 times more likely to discontinue etonogestrel sub-dermal implant early than those women who have been counseled before insertion (AOR = 3.19; 95% CI: (1.61, 6.30)). Similarly, those women who have not received an appointment for a follow-up visit at the time of insertion were 3.45 times more likely to discontinue early than those who received a follow-up appointment (AOR = 3.45; 95% CI: (1.89, 5.99)) (Table 3).
Factors associated with early etonogestrel sub-dermal implant discontinuation among women in Legambo district, Northeast Ethiopia, 2020.
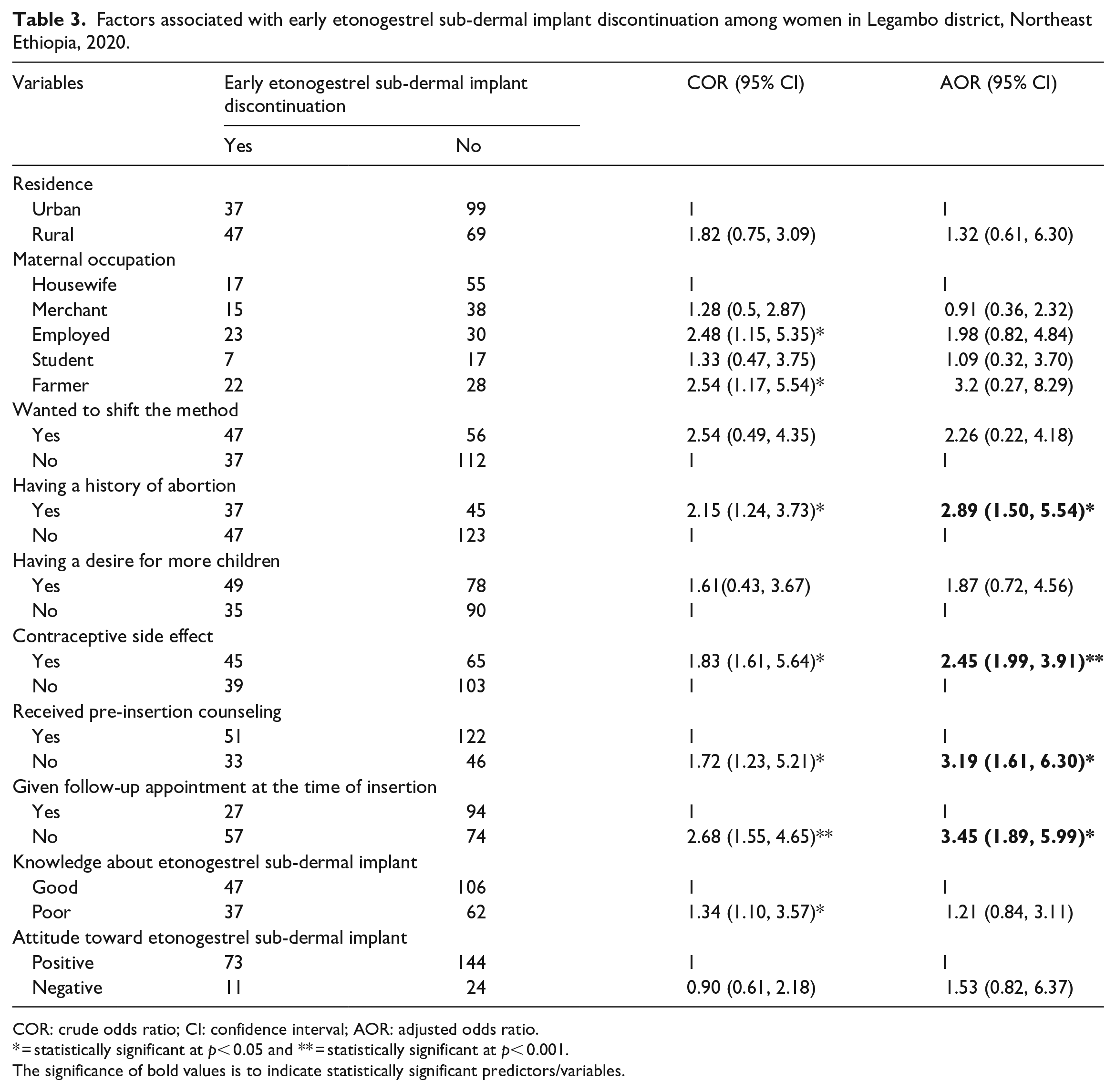
COR: crude odds ratio; CI: confidence interval; AOR: adjusted odds ratio.
* = statistically significant at p< 0.05 and ** = statistically significant at p< 0.001.
The significance of bold values is to indicate statistically significant predictors/variables.
In addition, Cox proportional hazard model was fitted to see the effect of history of abortion, contraceptive side effects, and receiving an appointment for a follow-up visit at the time of insertion on early discontinuation of etonogestrel sub-dermal implant. As a result, the hazard of early discontinuation of etonogestrel sub-dermal implant was 1.8 times higher among women who had a history of abortion compared to their counterparts (Adjusted Hazard Ratio (AHR) = 1.79; 95% CI: (1.17, 2.96)). Similarly, women who have not received an appointment for a follow-up visit at the time of insertion were 1.69 times more likely to discontinue the method early than their counterparts (AHR = 1.69; 95% CI: (1.26, 3.71)) (Table 4).
Cox regression of factors associated with early discontinuation of etonogestrel sub-dermal implant among women in Legambo district, Northeast Ethiopia, 2020.

CHR: crude hazard ratio; CI: confidence interval; AHR: adjusted hazard ratio.
Note. * = statistically significant at p < 0.05 and ** = statistically significant at p < 0.001.
The significance of bold values is to indicate statistically significant predictors/variables.
Discussion
To effectively confront the unreserved contraceptive discontinuation among women in Ethiopia, in general, and Legambo district, in particular, determinant factors influencing early discontinuation of etonogestrel sub-dermal implant need to be investigated. In this study, having a history of abortion, contraceptive side effects, not receiving pre-insertion counseling, and not receiving an appointment for a follow-up visit at the time of insertion had higher odds of early etonogestrel sub-dermal implant discontinuation.
Having a history of abortion had a positive association with early etonogestrel sub-dermal implant discontinuation. This finding is consistent with previous studies conducted in Diguna Fango district and Australia.8,49 It is also supported by a survival analysis result. The possible explanation would be that women who had a history of abortion might discontinue etonogestrel sub-dermal implant early due to fear of childlessness as a result of the double burden of abortion and contraception.
Women who experienced etonogestrel sub-dermal implant side effects were at high risk of early etonogestrel sub-dermal implant discontinuation. The finding was in agreement with other studies conducted in Ethiopia,46,50 Uganda, 51 Egypt, 52 Kinshasa, 53 Turkey, 54 Jos Nigeria, 55 and Jordan. 56 The possible reason might be inadequate pre-insertion counseling about the possible side effects. Studies indicated that the early discontinuation rate was higher among women who had not been adequately counseled about the side effects of the Implanon.5,46,57 The other reason might be women’s little intolerance to the newly occurring minor side effects of the method. Although the etonogestrel sub-dermal implant has no serious effects on the health of the women, they might be more worried about their sexual experience as a result of unfavorable bleeding patterns and would want the etonogestrel sub-dermal implant to be removed early in order not to interfere with their sexual relationship.
Not receiving an appointment for a follow-up visit at the time of insertion also had a positive association with early etonogestrel sub-dermal implant discontinuation similar to studies conducted in Diguna Fango district and Debre Tabor town.46,49 The finding is also supported by the survival analysis result. Women who have not been appointed for follow-up may not get further detailed counseling regarding the side effects, and those with the complain of etonogestrel sub-dermal implant side effects could not get management and supportive treatment from healthcare providers and hence they could discontinue the method early.
Women who have not received pre-insertion counseling discontinued etonogestrel sub-dermal implant early compared to their counterparts. This finding is witnessed by other studies conducted in Debre Markose, 35 Mekelle, 44 and Uganda. 51 The possible reason for this might be women who did not get effective pre-insertion counseling may not accept and tolerate minor side effects, and possibly they may seek early removal of the method. On the other hand, the information provided during counseling can clear their concerns or misconceptions related to the method causing early discontinuation. 58
Strength and limitations
This study had strength and limitations. As a strength, the study can minimize selection bias as it used hospital controls. As a limitation, this study was a facility-based study, the result of which might not be generalized to the general population. In addition, it is prone to recall bias as the client might experience the difficulty of remembering the date of insertion and removal of etonogestrel sub-dermal implant.
Conclusions
Having a history of abortion, not receiving pre-insertion counseling, not receiving an appointment for a follow-up visit at the time of insertion, and contraceptive side effects were determinants of early etonogestrel sub-dermal implant discontinuation. Healthcare providers should give detailed counseling regarding etonogestrel sub-dermal implant side effects before insertion to increase their retention. They should also give special emphasis to those women who had a history of abortion and should counsel about the benefit of etonogestrel sub-dermal implant for the long-term preparation of women for the next pregnancy. Health professionals should also give appointments for follow-up for close monitoring and management of etonogestrel sub-dermal implant side effects and to increase etonogestrel sub-dermal implant continuation rate. Further research with a qualitative study design would be recommended to explore the unknown factors.
Footnotes
Acknowledgements
We would like to express our deepest gratitude to Wollo University for providing the opportunity and Legambo District Health Office for providing the necessary information to conduct the study. We also would like to thank supervisors, data collectors, and study participants for their cooperation during the data collection process.
Author’s contribution
YY: initiated the research concept, involved in the data collection process, proposal development, data analysis, and interpretation; NC: involved in writing and reviewing the manuscript. YD: involved in writing and reviewing the manuscript; all the authors have read and approved the final manuscript.
Data availability
The datasets used for the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from the Ethical Review Committee (ERC) of Wollo University College of Medicine and Health Sciences (CMHS-2134/2020). The letter of permission to conduct the study was obtained from each health facility administration.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
After explaining the purpose of the study, written informed consent was obtained from all study participants before the data collection. In the case of minor subjects (aged less than 18 years), written informed consent was obtained from their legally authorized representatives. They were informed that participating in the study is voluntary and the right to withdraw from the study at any time during the interview was assured. Privacy of the client and confidentiality of information they give were secured at all levels and personal identifiers were not used. The information they provide was used for the study purpose only. The study also compiles and fulfilled the ethical principles of the Helsinki Declaration in research.
