Abstract
Pyoderma gangrenosum is a rare neutrophilic dermatosis often associated with systemic inflammatory or malignant conditions. We report a case of recurrent pyoderma gangrenosum injection site reactions following subcutaneous Maveropepimut-S (previously known as DPX-Survivac), an anti-cancer vaccine, in a 31-year-old woman undergoing immunotherapy for stage 3C ovarian cancer. The diagnosis was confirmed histologically and clinically after repeated ulcerative lesions developed at injection sites. The lesions showed a rapid response to corticosteroid therapy. This case highlights the importance of recognizing pyoderma gangrenosum as a potential cutaneous adverse event in immunotherapy treatments and suggests that timely dermatologic evaluation may help avoid delays in diagnosis and treatment.
Introduction
Pyoderma gangrenosum (PG) is an uncommon, painful, ulcerative dermatosis characterized by neutrophilic infiltration. It has a predilection for women and is frequently associated with systemic disorders, including inflammatory bowel disease, rheumatoid arthritis, and hematologic malignancies.1,2 Clinically, conventional PG presents with tender papulopustules that evolve into necrotic ulcers with undermined violaceous borders. 3 Several variants exist: ulcerative (most common), pustular, bullous, and vegetative, each with differing clinical courses. Cribriform scarring upon healing and pathergy (lesion induction by trauma) are hallmark features. 1
The pathogenesis of PG remains unclear, and diagnosis is clinical, made after excluding infections and other ulcerative conditions. In 2018, Maverakis et al. proposed a diagnostic criterion for the ulcerative form of PG that consists of one major and four out of eight minor criteria (Table 1). 4
Diagnostic criteria for ulcerative pyoderma gangrenosum. To diagnose ulcerative pyoderma gangrenosum, the major criterion must be present along with at least four of the eight minor criteria proposed by Maverakis et al.

Case report
A 31-year-old woman with stage 3C ovarian cancer underwent total abdominal hysterectomy with bilateral salpingo-oophorectomy, omentectomy, and pelvic lymph node dissection, followed by six cycles of carboplatin and paclitaxel. Upon progression of disease, she enrolled in a clinical trial (PRIMA, NCT03836352) receiving pembrolizumab, low-dose cyclophosphamide, and DPX-Survivac subcutaneous injection, a novel anti-cancer immunotherapy vaccine targeting solid tumors.
Following her first injection of DPX-Survivac (0.25 mg) into the right medial thigh, the patient developed a painful papule that rapidly ulcerated within 2 weeks at the site of drug administration. The ulcer exhibited violaceous, undermined borders with a purulent base (Figure 1(a)). There were no systemic symptoms or relevant personal or family history of autoimmune or inflammatory conditions. Conservative management (hydrogel dressing with sodium chloride and topical antibiotics) was initiated by oncology, and the ulcer healed over 4 months, leaving a cribriform scar.
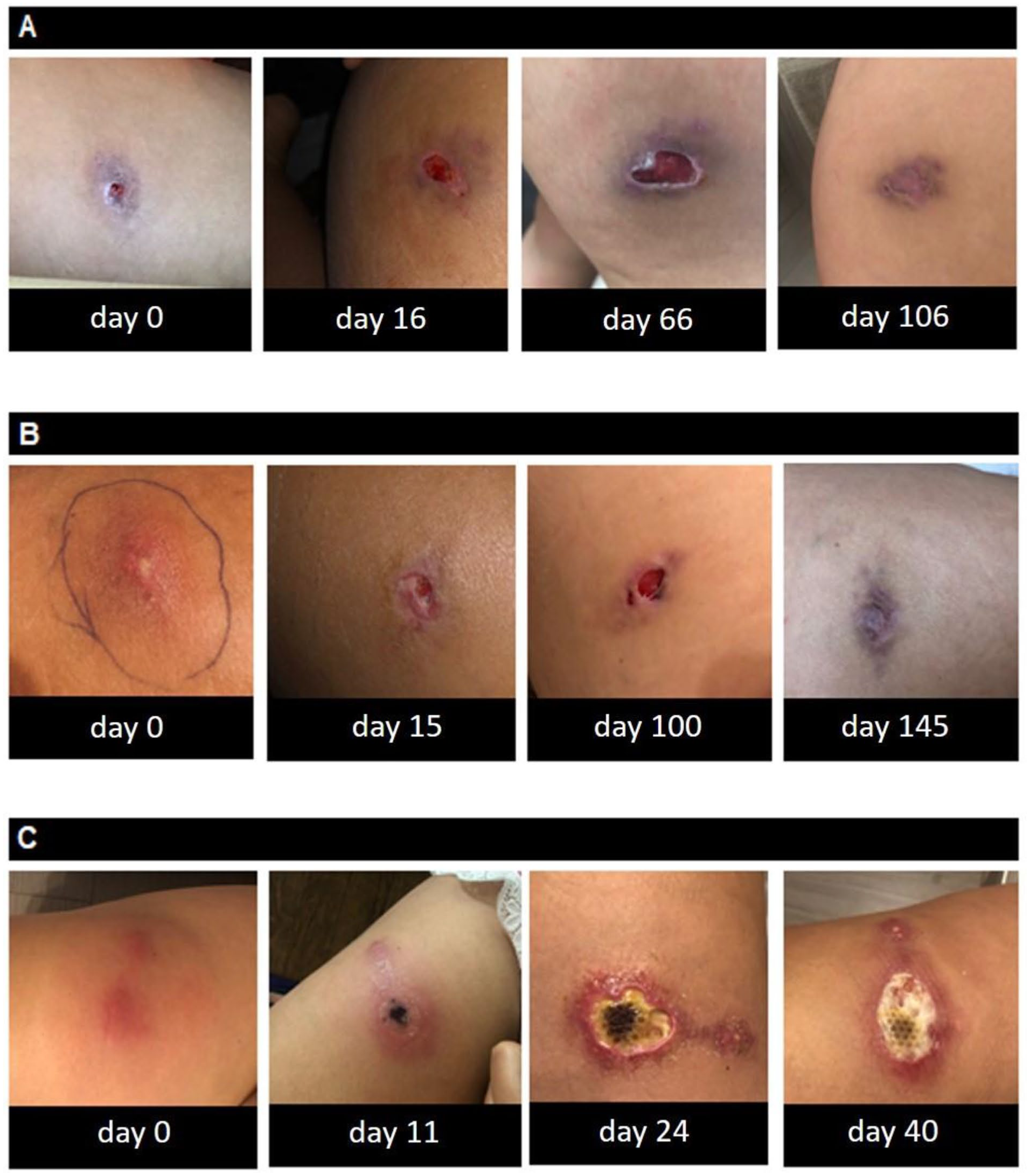
Progression of the ulcers. The first injection caused an ulcer with a necrotic, undermined, violaceous border, which eventually healed with a cribriform pattern of scarring (a). The second injection ulcer started as a papulopustule on a background of erythema that underwent necrosis and subsequently healed with a cribriform scarring pattern (b). After the third injection, rapid progression of an ulcer with an irregular, undermined, violaceous border with an erythematous rim and a purulent base was noted (c).
A similar ulcer developed at her second injection site on the left thigh, which resolved over 3 months with identical management (Figure 1(b)). Following the third injection into the right anterior thigh, another painful ulcer appeared (Figure 1(c)), but this time with minimal improvement after conservative wound care. A dermatology consultation was sought.
Biopsies for histology and culture were performed. Histopathology revealed a dense neutrophilic infiltrate without evidence of infection. Based on the histological findings (major criterion) and presence of four minor criteria (exclusion of infection, history of papule, vesicle, or pustule ulcerating within 4 days, peripheral erythema, underminedborders with tenderness at the site of ulcer, and cribriform scars), a diagnosis of drug-related injection site PG was made.
The patient received an intralesional injection of triamcinolone acetonide (40 mg/mL) and was started on clobetasol propionate 0.05% ointment twice daily under foam dressing. At follow-up, 2 weeks later, there was a significant reduction in ulcer size and pain. The patient documented with photography her ulcerating lesions and shared them with written informed consent for publication (Figure 1).
Discussion
This case illustrates a recurrent and localized form of PG temporally associated with Maveropepimut-S (previously known as DPX-Survivac) injections. While PG is classically linked to systemic inflammatory and hematologic diseases, this case suggests that novel immunotherapies may act as potential triggers (including at injection sites), possibly through mechanisms involving vaccine-induced immune activation or pathergy. 5 Although PG has not been previously reported in association with Maveropepimut-S, its clinical presentation here aligns with known patterns of neutrophilic dermatoses. 2
Given the increasing use of cancer immunotherapy, clinicians should remain vigilant for rare but significant cutaneous adverse events. A high index of suspicion and timely dermatologic evaluation can lead to accurate diagnosis and appropriate immunosuppressive treatment, minimizing patient morbidity.
Conclusion
This case highlights the importance of recognizing PG as a potential adverse effect of Maveropepimut-S immunotherapy. Clinicians should include PG in the differential diagnosis of painful post-injection ulcers, especially in the context of cancer immunotherapy. Early recognition and prompt initiation of immunosuppressive therapy can help prevent complications and reduce morbidity.
Footnotes
Consent for publication
Patient provided written informed consent to use images for publication and/or teaching.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
