Abstract
Introduction:
Heart failure is a major public health problem worldwide. Since heart failure with reduced ejection fraction and preserved ejection fraction are different clinical entities, in-hospital mortality may occur at different rates. This study aimed to assess the management, survival, and predictors of mortality among hospitalized heart failure patients at Debre Markos comprehensive specialized medical ward.
Methods:
A prospective cohort study was conducted on 228 heart failure patients at Debre Markos Comprehensive Specialized Hospital medical wards. A structured data collection tool was used to collect data. Data were analyzed using SPSS version 21.0. The Kaplan–Meier survival curve was used to investigate if there was a difference in the in-hospital survival between heart failure with a reduced ejection fraction and heart failure with a preserved ejection fraction. Those variables having p-value < 0.05 were considered statistically significant.
Results:
From the 228 participants, 126 (55.3%) were females with a mean age of 53.32 ± 15.68 years. One hundred thirty-three (58.3%) patients were presented with preserved (⩾50%) level of ejection fraction. The all-cause in-hospital death rate was 12.7%, and the risk of in-hospital mortality was higher in heart failure patients with reduced ejection fraction (7.4% vs 5.3%; p = 0.005). Current occupation (p = 0.041), elevated serum creatinine (p = 0.010), reduced ejection fraction (p = 0.017), and asthma/chronic obstructive pulmonary disease comorbidity (p = 0.002) were the independent predictors of high hospital mortality.
Conclusion:
The rate of in-hospital mortality among heart failure patients was high. Healthcare providers should provide effective education activities and define disease management strategies for patients with reduced ejection fractions.
Keywords
Introduction
Heart failure (HF) is a major public health problem internationally. In 2015, there were 26 million individuals with HF worldwide.1,2 Currently, it is one of the major clinical challenges in developing countries. HF is common in developing countries and continues to increase in incidence and prevalence with minimal improvement in prognosis.1,3,4 In sub-Saharan Africa, HF has been shown to affect young and middle-aged adults most likely to be in New York Health Association (NYHA) Functional Class IV compared with those from Asia, the Middle East, and South America. 3
In Ethiopia, there has been an epidemiological transition in the burden of diseases from communicable to non-communicable diseases (NCDs). 4 Moreover, in developing countries, priority is given to acute disorders, child and maternal healthcare, and control of communicable diseases, giving very little emphasis on chronic NCD.3,4 Different studies conducted in Ethiopia showed that HF is the commonest cardiovascular disease (CVD) in Ethiopians.5–7
Treatment outcomes of HF significantly vary in HF patients with preserved ejection fraction (HFpEF) and reduced ejection fraction (HFrEF). In the United States, 30-day mortality was 10% lower in patients with HFpEF than HFrEF. 8 A study conducted in Italy showed that diuretics were the most commonly prescribed class of drugs during hospitalization and at discharge (81% vs 90%). 9 while a study on seven Gulf countries showed that intravenous frusemide and nitrates were administered in 93% and 20% of the patients, respectively. 10
A prospective multicenter longitudinal study in New Zealand (NZ) and Singapore on 2039 patients showed that after adjusting for age, sex, and clinical risk factors, patients with HFpEF had a lower risk of death compared with those with HFrEF (hazard ratio = 0.62, 95% confidence interval (CI) = 0.46–0.85). 11 A Report from the OPTIMIZE-HF Registry indicated that the risk of in-hospital mortality was lower in patients with HFpEF as compared to with (2.9% vs 3.9%). 12 A report from the Acute Decompensated Heart Failure National Registry Database showed that in-hospital mortality was lower in patients compared with patients with HFrEF (2.8% vs 3.9%). This study analysis identified that elevated blood urea nitrogen (BUN), lower systolic blood pressure (BP), low sodium, older age, elevated creatinine, presence of dyspnea at rest, and absence of chronic beta-blocker use as mortality risk factors. 13
A study conducted in Korea showed that in-hospital mortality was 5.2% and low blood pressure and azotemia were the most important predictors of in-hospital mortality. 14 In Sweden, mortality risk was greater for those with HFpEF (HFrEF (7.5% vs 5.9%)), respectively. In contrast, other studies indicate that preserved ejection fraction (EF) was associated with a lower risk of death (13% of patients with preserved EF died, compared with 21% of patients with depressed EF). 15
In Nigeria, mortality rate at 30 days was 4.2% and female gender, being single, HF with normal EF, lower blood pressure, higher heart and respiratory rates, higher body temperature, anemia, high creatinine levels, and higher total white blood cell counts. 16 In Botswana, the in-hospital mortality rate of HF patients was 10.9%, and hyponatremia elevated N-terminal (NT)-pro hormone BNP (NT-proBNP), urea levels, and hyperuricemia were significantly associated with in-hospital mortality. 17
Although several studies have been conducted in Europe and the United States on the clinical characteristics, treatment, and outcome of HF patients, little information is available on this subject in Ethiopia, particularly in the study area.8,12,18 Therefore, this study aimed to assess the management, survival, and predictors of mortality among patients hospitalized with HF at Debre Markos comprehensive specialized medical ward.
Methods
Study design and site
A prospective cohort study was conducted from 1 November 2019 to 30 April 2021, at Debre Markos Comprehensive Specialized Hospital (DMCSH). It is located in east Gojjam, Amhara region, Ethiopia. 7
Study population
All adult hospitalized patients aged ⩾18 years with a diagnosis of decompensated or new-onset HF at DMCSH and who were voluntary to participate in the study were included in the study. HF patients with concomitant malignancy or World Health Organization stage 4 HIV infections and patients with incomplete information were excluded from the study.
Sample size determination and sampling method
We used a previous study conducted in Korea, in 2014, which reported that the rate of in-hospital mortality was 3.3% and 8.8% in patients with HFpEF and HFrEF, respectively, for the sample size calculation. 19 Then, we used the following formula
where p0 is the proportion of HFpEF in-hospital mortality = 0.033, p1 is the proportion of HFrEF 1-year mortality = 0.088, q0 = (1 − p0) = 1.0 − 0.033 = 0.967, q1 = (1 − p1) = 1.0 − 0.088 = 0.912, Z1 − α/2 = 1.96 is the value of the standard normal distribution (with 95% CI), and Z1 − β = 0.84 is the value of Z from standard normal distribution (β = 0.20). A sample size of 104 for each group was calculated from the above formula, which was adjusted with the assumption of 10% non-response, to yield a sample size of 114 HF patients for each group and a random sampling technique was used.
Study variables
The dependent variable was the 30-day in-hospital mortality rate. The independent variables included in this study were socio-demographic variables: age, sex, educational status, residence, body mass index (BMI), alcohol use, salt un-restricted, smoking chewing chat, drinking coffee, physical inactivity, vital signs, clinical presentation, laboratory, and other diagnostics results. Clinical and medication-related variables were: co-morbid illnesses, current medications, and adjuvant medication use, guideline-based treatment.
Data collection tool and procedures
A structured data collection tool which includes a questionnaire and data abstraction format was prepared by reviewing different kinds of literature for important variables to this study and used to extract all necessary information. The data collection tool’s feasibility, readability, consistency of style and formatting, the likelihood HF patients would be able to answer the questions, and content area and the clarity of the language used was evaluated by five experts. The internal consistency reliability of the tool was also established. The overall Cronbach’s alpha of the modified tool was 0.67. Data from parents/caregivers were collected by face-to-face interview and secondary data such as prescribed drugs, laboratory and diagnostic results, and co-morbid illness were collected from the patient’s medical chart daily.
The EF of the participants was measured using an echocardiogram during their first admission. Only the first HF hospitalization was used to determine hospital mortality. Data were collected by two clinical nurses.
Data quality assurance
A 1-day training was given to the data collectors by the principal investigator regarding the data collection method and appropriate use of the data collection instruments. The questionnaire was translated to the local language, Amharic, for consistency. A pre-test of data collection format was performed on 5% of the sample before conducting the study. Then, the final tool was developed with some modifications after reviewing the results of the pre-test. The quality of data was also checked at data entry, analysis, and interpretation.
Data processing and analysis
The data were entered into a computer using Epidata version 3.1 and exported to the Statistical Package for Social Science (SPSS) version 21.0 for analysis. Continuous variables were presented as mean values (standard deviation). Categorical variables were presented as frequency and percentages. Bivariate analysis was done to see associations between the dependent and independent variables. Variables having a p-value of less than 0.25 were included in multivariate Cox regression analysis. Those variables having p-value < 0.05 were considered statistically significant. The Kaplan–Meier survival analysis was done to compare in-hospital survival differences between HF with reduced and preserved EF. Tables, figures, and texts were used to present the finding accordingly.
Operational definitions
Thirty-day in-hospital mortality rate: defined as death in hospital following the index admission date to the next 30 days before discharge. 10
Censored: defined when participant lost before an event (death) occurs or death not observed within the study period.
Co-morbidity: it is the presence of one or more additional disorders (diseases) co-occurring with a primary disease or disorder. 7
Physically active: classified as if they reported that they exercise greater than 30 min greater than 5 days of the week, otherwise they were classified as physically inactive.
Preserved EF: expressed as a percentage of how much blood the left ventricle (LV) pumps out with each contraction (normal ⩾50%) other ways considered as reduction EF. 10
Hypertension: defined when blood pressure ⩾130/80 mm Hg for all age groups and comorbidities. 7
Results
Demographic characteristics of study participants
A total of 228 patients were included in the final analysis. Of these, 126 (55.3%) were females and 58.3% of patients had HFpEF. The mean age of participants was 53.32 ± 15.68 years. More than one-half of the participants (n = 120) were from rural centers and 44.3%% of the patients have no formal education. Patients with HFpEF were less physically active than patients with HFrEF; however, they had good salt intake restriction (Table 1).
Demographic characteristics of patients admitted with heart failure according to left ventricular EF.

HF: heart failure; BMI: body mass index.
Clinical characteristics of patients admitted with HF at DMCSH
More than one-third (42.5%) of patients were presented with acute de novo HF and there was a higher prevalence of de novo HF in the HFrEF (52.6%) than in the HFpEF (33.6%) group. Objective atrial fibrillation (AF) was documented in 48.0% of HFrEF and 52.0% of HFpEF patients. Pulmonary congestion (67.2%), cough (62.0%), and fatigability (60%) were more frequently experienced in HFpEF. Patients with HFrEF showed high mean systolic blood pressure, diastolic blood pressure, and higher mean heart rate at admission compared with patients with HFpEF. Similarly, mean potassium level was higher in patients with HFrEF than patients with HFpEF (Table 2).
Clinical characteristics of patients admitted with heart failure at DMCSH.

HF: heart failure; NYHA: New York Health Association; JVP: jugular venous pressure; SBP: systolic blood pressure; DBP: diastolic blood pressure; BUN: blood urea nitrogen; RBS: random blood sugar; FBS: fasting blood sugar; COPD: chronic obstructive pulmonary disease.
Others include thyroid disease, thromboembolism, diastolic blood pressure, chronic kidney disease, and angiotensin receptor blockers.
Admission, and discharge medications among HF patients at DMCSH
During the index admission, more than two-third (74.1%) of participants had received diuretics, whereas 41.7% had received digoxin. During discharged diuretics, beta-blockers and angiotensin-converting enzyme inhibitors (ACEIs) were the most frequently prescribed class of medication (64.5%, 44.7%, and 42.5%), respectively. Compared with HFrEF patients, the use of diuretics (40.4%) was higher in the HFpEF group whereas the use of digoxin, angiotensin receptor blockers (ARBs), and the inotropic agent was lower during admission in HFpEF (Table 3).
Admission and discharged medication for hospitalized heart failure patients.

HF: heart failure; ACEIs: angiotensin-converting enzyme inhibitors; ARBs: angiotensin receptor blockers.
Thirty-day follow-up survival analyses and determinants of in-hospital mortality among HF patients
The all-cause in-hospital death rate was 29 (12.7%), of which 17 (7.4%) occurred in patients with HFrEF and 12 (5.3%) in patients with HFpEF. The mean survival time was 23.43 ± 1.05 and 27.17 ± 0.74 days for HFrEF and HFpEF, respectively. Using Kaplan–Meier curves, mortality rates were compared based on exposure to EF (Figure 1). Patients with HFrEF experienced a higher in-hospital mortality rate than patients with HFpEF (p = 0.005) and survival time decreased as the length of hospital stay increased.

Overall survival of heart failure patients with reduced (HFrEF) and preserved ejection fraction (HFpEF).
Twenty variables were with p-value less than 0.25 in bivariate analysis. Hence, these variables were included in the multivariate Cox proportional hazards models. However, only current occupation, serum creatinine, EF level, asthma/chronic obstructive pulmonary disease (COPD) comorbidity, and the use of ARBs were significantly associated with in-hospital mortality in multivariate Cox regression. Accordingly, government employees were almost three (adjusted hazard ratio (AHR) = 2.79, CI = 1.04–7.46, p = 0.041) times more likely associated with in-hospital death than non-government employees. Similarly, serum creatinine (AHR = 3.65, CI = 1.36–9.79, p = 0.010) and EF level (AHR = 3.38, CI = 1.24–9.18, p = 0.017) were also independently associated with a worse outcome in hospitalized HF patients. Participants who had asthma/COPD comorbidity were almost two times (AHR = 1.75, CI = 0.60–5.08, p = 0.002) more likely to die than without this comorbidity. In contrast, the use of ARBs decreased the risk of mortality, that is participants who were on ARBs were 0.52 times (AHR = 0.52, CI = 0.19–1.42, p = 0.003) less likely to have in-hospital mortality (Table 4).
Binary and multivariate Cox proportional hazard analysis to identify factors associated with in-hospital mortality among heart failure patients at DMCSH.
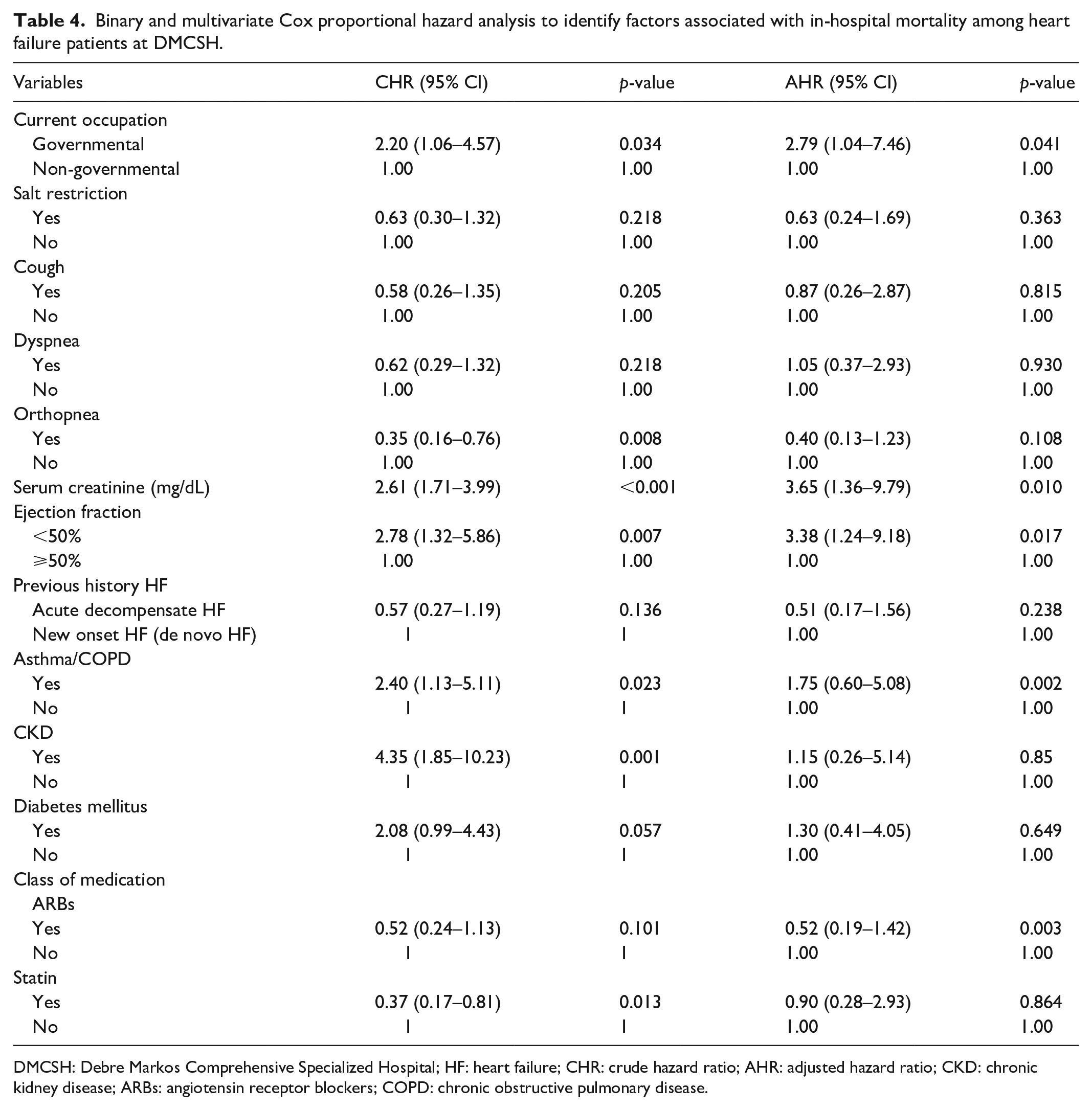
DMCSH: Debre Markos Comprehensive Specialized Hospital; HF: heart failure; CHR: crude hazard ratio; AHR: adjusted hazard ratio; CKD: chronic kidney disease; ARBs: angiotensin receptor blockers; COPD: chronic obstructive pulmonary disease.
Discussion
In this study, we compared treatment and in-hospital mortality of hospitalized HF patients based on EF levels. The result of this study showed that more than one-half of (58.3%) HF patients had preserved EF. More than half of patients with HFrEF were presented with new-onset (de novo) HF and had high mean systolic pressure and heart rate. This may be since uncontrolled hypertension is the main cause of left ventricular hypertrophy, where the heart muscle is weakened, which affects its ability to pump properly, and increasing heart rate is an immediate compensatory mechanism of decreasing cardiac output in new-onset HF. 20 Pulmonary congestion, cough, and fatigue were more frequently experienced in HFpEF. The result was in line with a study conducted in Korea. 21 This may be because patients with HFpEF were presented with impaired relaxation and stiff myocardium which resulted from an increasing load to the LV attributed to the stiff arterial system (vascular uncoupling which leads to pulmonary edema and cough).
The finding of this study showed that the overall mortality rate of hospitalized HF patients was 12.7%%. 18 This result was closely similar to studies done in India (12.5%), England, and Wales (12%).22,23 However, mortality in England and Wales was higher among those with HFpEF (11%) as compared with HFrEF (8%). The difference might be due to the large sample size in England and Wales (6170) and variation in cut point of EF (<40% and ⩾40%). All causes of hospital mortality were similar with the result from Canada (10.7%) and Sweden (13.4%) (7.5% vs 5.9% in HFpEF and HFrEF) and Botswana (10.9%).17,19,20 In contrast, the total in-hospital and subgroup mortality was lower than studies in St. Paul’s Hospital Millennium Medical College in Addis Ababa, Ethiopia (24.4%) and Emory University School of Medicine in Atlanta (16.3% in patients with HFrEF and 13.2% in patients with HFpEF), respectively. 18 This difference might be due to the study design and duration of the study (5 years retrospective cohort study in St. Paul’s Hospital Millennium Medical College and 3 years retrospective cohort study in Atlanta). In the same way, several studies reported lower rates of mortality among HFpEF as compared with HFrEF.13,15,19
In contrast, the overall hospital mortality rate in this study was higher than studies done in the United States (7%), Italy (6.4%), Portugal (5.5%), and Korea (5.2%).8,14,22 The low level of mortality rate in these countries may be due to better medical and pharmaceutical care to patients, and better education and awareness of patients to heart treatment. It was also higher than studies done in Nigeria (3.8%), Egypt (5%), and Tikur Anbessa Specialized Hospital, Ethiopia (3.6%).24–26 This difference in the mortality might be due to inclusion criteria, sample size, and clinical characteristics of participants. Study participants in Nigeria were, for example, from the urban community and 91.8% of them had new-onset HF which implies they present early to healthcare institutions before developing different complications (90% of the subjects were in NYHA Functional Class II or III as compared to 93.0% had NYHA Class IV in this study). Similarly, in Egypt, participants were from the ambulatory clinic and only 14.8% of them had Asthma/COPD comorbidity as compared to 42.5% participants in this study which was significantly associated with hospital mortality. 24 However, variation from the study done in Tikur Anbessa Specialized Hospital, Ethiopia, may be due to a smaller sample size (169 vs 228 participants) than this study. Our results showed that survival was better in patients with HFpEF. Numerous other investigators have supported this finding.13,19,27,28 However, it was noted that although mortality for patients with HFpEF may be lower, the absolute burden of mortality is still substantial. These findings provide important insights into the factors that contribute to HF mortality and have important implications for the care of patients hospitalized with HF. Asthma/COPD comorbidity is also another independent predictor for short-term survival. The poor treatment outcomes and higher mortality with asthma/COPD might be associated with the disease’s worse prognosis that leads to altered structure (e.g. hypertrophy or dilatation) and/or impaired function of the right ventricle that results from pulmonary hypertension.8,20 Importantly, the use of ARBs reduces the mortality rate among HF patients. Randomized controlled trials established that ARBs reduce morbidity and mortality in HFrEF. 29 It is also true that ARBs promote regression of ventricular hypertrophy and reduce the incidence of sudden cardiac death in patients with LV dysfunction. 30 This observation highlights the importance of developing and implementing quality improvement programs in the evidence-based management of HF.
In this study, a significant association was observed between patients’ current occupations. Government employees had a higher HF mortality rate. The exact mechanism was unclear, but it might be due to time constraints to come to the health facility for routine follow-up and difficulty remembering to take all their medicine on time resulting in hospitalization.
Another factor that influences patient survival was high serum creatinine. The result was similar to those studies conducted in different countries.13,16,27 This finding is also supported by the ACEF (age, creatinine, EF) score study which showed that two of three components of the ACEF score, that is, age and serum creatinine level, are strong predictors of ischemic and bleeding events in the setting of acute coronary syndrome. 31 This explained that patients with renal impairment often develop the cardio-renal syndrome, which is caused by low cardiac output. In addition, these patients often develop multiple alterations at the vascular level, leading to endothelial dysfunction, coagulation abnormalities, insulin resistance, hyper-homocysteinemia, and activation of the sympathetic nervous system, as well as the renin–angiotensin and aldosterone system resulting in further deterioration.
Diuretics were administered in most patients admitted for HF. Indeed, relief of pulmonary and peripheral congestion is the main goal of acute heart failure (AHF) to stabilize the patients.9,10,27 The use of ARBs, digoxin, and inotropic agents during hospitalization significantly increased patients with HFrEF compared to that HFpEF. This finding is in line with the 2017 ACCF/AHA Guideline for the Management of Heart Failure recommendation and is also supported by different investigators.13,17,32 In contrast, the use of ACEIs and beta-blockers was higher among patients with HFpEF. This variation may be explained that sub-optimum guideline-based medical treatment even though it was not statically significant.
The use of ARB but not ACEIs showed protective effects in patients with HFrEF in this study. However, a study in South Korea showed that both ACEIs and ARBs decrease hospital mortality among HFrEF. 21 Beta-blockers were the second most prescribed discharge medications following diuretics which were also significantly higher among the HFpEF group. The rate finding was in line with the OPTIMIZE-HF Registry, however in less in a patient with HFrEF. 12 However, In Egypt and Gulf acute HF registry, the second most frequent discharge medications were ACE/ARBs after diuretics.10,24 This implicated that the use of beta-blocker gets attention after the acute phase of HF treatment in most healthcare settings. In addition to pharmacological treatment of HF, an intra-aortic balloon pump as a mechanical support device can be used in intensive cardiac care unit (ICU) patients in order to improve patient outcomes. 33
Limitations of the study
This is a prospective cohort study, addressing the relation of a broad scope of determinants with in-hospital mortality. However, the study was conducted in one setting; the findings cannot be generalized to reflect the healthcare setting in Ethiopia. In addition, we followed patients for 30 days after the index of admission; we could not ascertain the exact cause of death for patients after 30 days and who died outside the hospital environment. There is no consensus in the published data supporting one most appropriate definition of preserved EF. However, even analyses with the most restrictive definition showed similar findings, so analyses with current quantitative EF may not have a significant impact on the findings.
Conclusion
The rate of in-hospital mortality among HF patients is still high and the risk of death was higher in patients with HFrEF. During hospitalization, digoxin appeared to be overused in HFrEF while ACEIs were most commonly used at the discharge for patients with HFpEF. These findings indicated that the clinical outcomes of HF are influenced by multiple factors beyond EF and implementation of guidelines. The clinical practice must take into account the phenotype (heterogeneity) of each patient to improve patient outcomes. In addition, these patients may need to be investigated separately with a large sample size, in multicenter outcomes studies and clinical trials. Healthcare providers should provide effective education activities and define disease management strategies for patients with reduced EFs.
Footnotes
Acknowledgements
The authors thank data collectors, study participants, and the health professionals working at DMCSH for their cooperation.
Author contributions
B.K. made contributions to the conception and design of the study. B.K., B.D., M.G., Y.M., and B.B. did data analysis and interpretation of data, took part in the preparation of the manuscript, agreed to submit to the current journal, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by the Ethical Reviewer, College of Health Science Debre Markos University with a reference number HSC/R/C/Ser/Co/347/06/12. An official letter was written to Debre Markos comprehensive specialized hospital. This study was conducted following the Declaration of Helsinki ethical principles, where informed written consent was taken from each participant. 34 Participants were informed about the objective of the study, procedures of selection, and assurance of confidentiality, and their names were not registered to minimize social desirability bias and enhance anonymity. Patients were not forced to participate and received any monetary incentive, and it was solely voluntary-based. Each participant was informed about his or her right of withdrawing from the study without restriction whenever necessary. The collected data were handled and secured with the principal investigator on every data collection day.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Debre Markos University.
Informed consent
Written informed consent was obtained from all subjects before the study.
Data availability statement
The data used to support the finding of this study will be available from the corresponding author upon request.

