Abstract
Background:
Acute heart failure affects ~9% of patients with hematological malignancies. This study aimed to evaluate the impact of acute heart failure on in-hospital mortality among patients with hematological malignancies and to identify independent predictors of mortality in this population.
Methods:
The National Inpatient Sample (2016–2021) was queried for adults (⩾18 years of age) with hematological malignancies and those who experienced in-hospital mortality. Among these, we identified patients who were hospitalized with a diagnosis of acute heart failure. Univariate analysis and multivariate regression were employed to analyze the mortality risk. A p ⩽ 0.05 was considered significant.
Results:
Among 40,246 patients with hematological malignancies who died in the hospital, the univariate analysis showed a higher in-hospital mortality in the acute heart failure cohort compared to the non-acute heart failure group (8.08% versus 4.52%, p < 0.001). However, after adjusting for sociodemographic factors and comorbidities, acute heart failure was associated with a lower risk of in-hospital mortality (OR 0.87, 95% CI 0.84–0.9, p < 0.001). Predictors of mortality in the acute heart failure cohort were age >60 years, hematopoietic stem cell transplant, severe sepsis, arrhythmias, infective endocarditis, bleeding from any source, acute respiratory failure, hepatic dysfunction, metabolic encephalopathy, and invasive ventilation (all p < 0.01). On subgroup analysis, acute myeloid leukemia, chronic myeloid leukemia, chronic lymphocytic leukemia, and multiple myeloma had a higher in-hospital mortality in the acute heart failure cohort compared to the non-acute heart failure group (p < 0.001).
Conclusion:
Acute heart failure in patients with hematological malignancies was associated with poor outcomes in the presence of severe sepsis, organ dysfunction, and the need for mechanical ventilation. Close monitoring and a multidisciplinary team approach are recommended to optimize the treatment plan for this population.
Introduction
Hematological malignancies (HMs) represent 3.3% of cancer incidence in the United States and carry a 3.8% mortality. 1 These patients often show increased cardiac remodeling and dysfunction, due to the inflammatory and fibrotic effects of clonal hematopoiesis and direct cardiotoxicity of chemotherapy.2–5 The prevalence of heart failure in HMs is ~9%.6–9 Comorbidities such as anemia, hyperviscosity, arrhythmias, sepsis, diabetes mellitus, hypertension, obesity, and dyslipidemia may increase the risk of heart failure.2–5,10,11
There is a lack of comprehensive, large-scale studies on acute heart failure (AHF) and the use of cardiotoxic drugs in this population.12–14 Prior studies often relied on post-mortem evaluations, which may have introduced selection bias and failed to account for comorbid conditions.15,16 Some of the contributing factors to mortality may still be unrecognized, limiting clinicians’ ability to develop effective treatment plans.9,17–19 Despite existing risk evaluation strategies, there is no universally accepted method for risk stratification or guiding the management of AHF.12,13
Our study aimed to retrospectively analyze the National Inpatient Sample (NIS) database from 2016 to 2021. The primary objective was to evaluate in-hospital mortality among patients with AHF and identify factors contributing to mortality.
Methods
Study design, population, and database description
We conducted a retrospective analysis of patients aged 18 years or older diagnosed with HMs. The study utilized the NIS database, capturing data from 2016 to 2021. It included patients with International Classification of Diseases, Tenth Revision, and Clinical Modification (ICD-10-CM) codes for HMs.20–22 Supplemental Table 1 lists the ICD-10 codes used in this study. Patients younger than 18 years were excluded from the analysis. A predetermined sample size was not calculated. The design of this sample was stratified by probability, ensuring that it represents all non-federal acute care hospitals nationwide. Subsequently, a random sample including 20% of all hospitals was collected. After each hospital discharge, a weight was assigned based on the total number of discharges from all acute care hospitals in the United States divided by the number of discharges included in the 20% sample. This weighting was performed to create a data model that accurately represents the entire nation. The dataset, spanning from 2016 to 2021, comprised over seven million annual discharges and accurately reflected 95% of hospital discharges nationwide.
Study outcomes
The primary outcome was in-hospital mortality, as reported in the NIS, which reflects all-cause mortality. We aimed to evaluate the mortality risk in patients with AHF compared to those without AHF. Patients with HMs included individuals with both active disease and those in remission. These patients were further stratified based on disease activity, and comparisons were made between the AHF and non-AHF cohorts. Secondary outcomes assessed mortality risk across each type of HMs, including acute myeloid leukemia (AML), chronic myeloid leukemia (CML), chronic lymphocytic leukemia, and multiple myeloma (MM), by comparing AHF versus non-AHF cohorts. Also, patients with CML who tested positive for Breakpoint Cluster Region–Abelson leukemia (BCR/ABL) tyrosine kinase mutations were identified and subsequently divided into AHF and non-AHF cohorts for comparative analysis. Tertiary outcomes focused on identifying the predictors of in-hospital mortality among patients with AHF.
Statistical analysis
Quantitative variables were analyzed with means, and qualitative variables were analyzed using the chi-square test. A non-parsimonious multivariate logistic regression model was developed to estimate the mortality risk of AHF compared to the non-AHF cohort. The regression models were built by including all confounding variables that showed an association with the outcome on univariate analysis, with a p value cutoff of 0.3. The literature review helped identify the key variables that were incorporated into the models. Additionally, regression models were built to determine the predictors of mortality. We set a significance level of p ⩽ 0.05. Stata v18 (StataCorp, TX, USA) was used to perform the analysis.
Ethical considerations
The NIS is a de-identified, publicly accessible database. Before accessing the database, we adhered to the Agency for Healthcare Research and Quality (AHRQ) data user agreement. The database complies with the Health Insurance Portability and Accountability Act (HIPAA) Privacy Rule for limited data sets, contains no explicit patient identifiers, and is exempt from Institutional Review Board approval, consent, and written informed consent requirements due to the use of de-identified, publicly available data.
Results
Hematological malignancies
We extracted 821,530 patients with HMs from the NIS database. Among these, we focused on 40,246 patients who died in the hospital and compared the mortality risk between those with and without AHF. The mean age was higher in the AHF cohort compared to the non-AHF cohort (73.58 versus 69.55 years). Gender distribution was comparable between the groups (p > 0.05). Racial distribution varied slightly, with a higher proportion of American White patients (73.53% versus 69.58%) and a lower proportion of African American (13.04% versus 14.3%) and Hispanic patients (7.12% versus 9.17%) having AHF (p < 0.001). Comorbidities such as hypertension, diabetes, chronic kidney disease, cardiovascular grafts, arrhythmia, and use of long-term anticoagulants and antiplatelets were more prevalent in patients with AHF (all p < 0.001). The proportion of patients who received inpatient chemotherapy and underwent hematopoietic stem cell transplantation (HSCT) was lower in the AHF cohort (p < 0.001). Table 1 describes the Sociodemographics and Comorbidities of patients with hematological malignancies.
Sociodemographics and comorbidities in hematological malignancies patients with mortality.
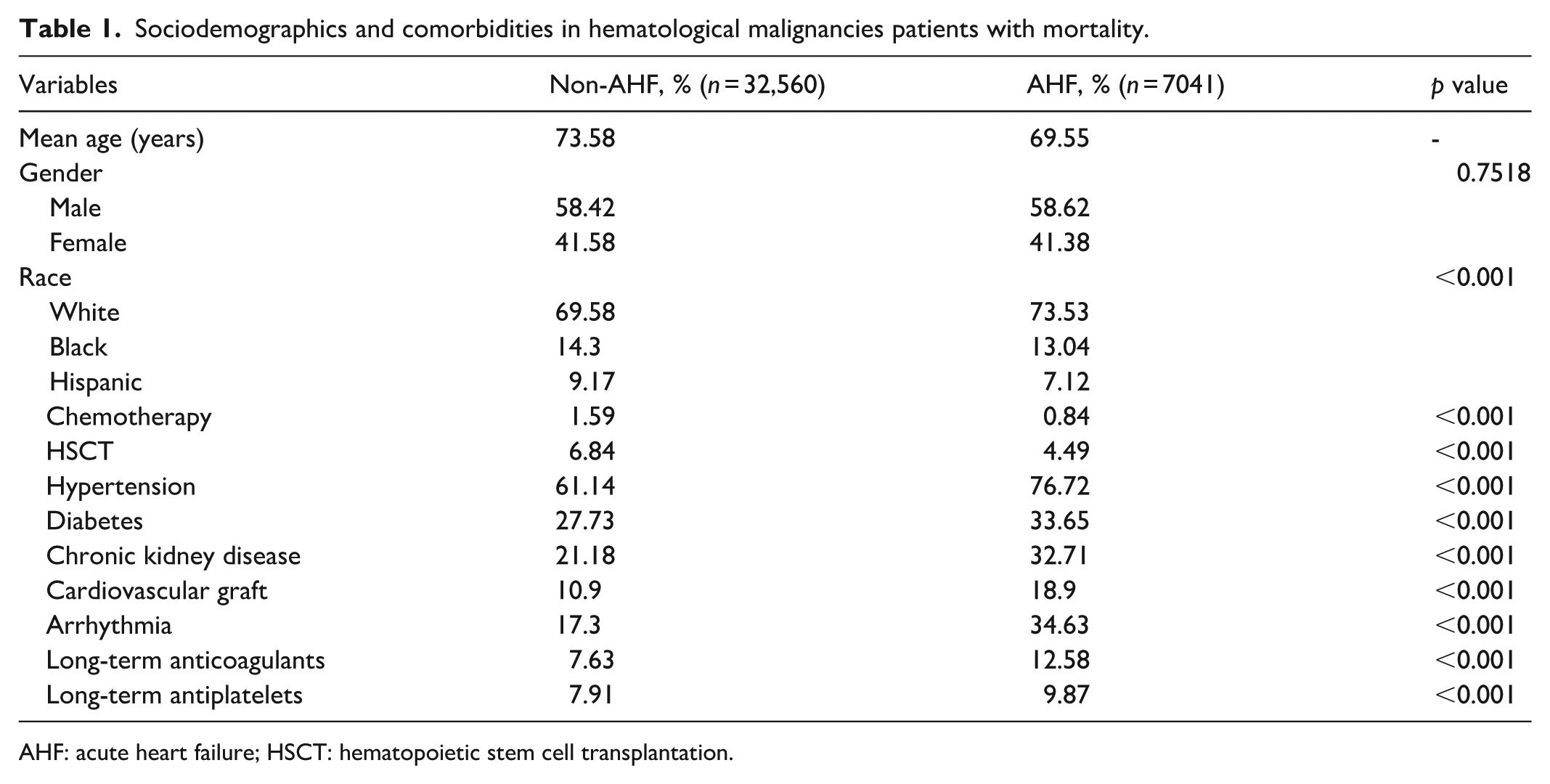
AHF: acute heart failure; HSCT: hematopoietic stem cell transplantation.
On univariate analysis, in-hospital mortality was significantly higher in the AHF cohort compared to the non-AHF group (8.08% versus 4.52%, p < 0.001). However, after adjusting for sociodemographic factors and comorbidities, AHF was associated with a lower risk of in-hospital mortality (OR 0.87, 95% CI 0.84–0.9, p < 0.001) as noted in the multivariable regression model. Table 2 illustrates the results of the mortality of patients with HMs and AHF. Predictors of mortality in the AHF cohort were age >60 years (p < 0.001), HSCT (p < 0.001), severe sepsis (p < 0.001), arrhythmias (p < 0.001), infective endocarditis (p < 0.01), bleeding from any source (p < 0.001), acute respiratory failure (p < 0.001), hepatic dysfunction (p < 0.001), metabolic encephalopathy (p < 0.001), and invasive ventilation (p < 0.001). Mortality risk was lower among African Americans (p < 0.001), patients with diabetes (p < 0.001), hypertension (p < 0.001), and those receiving long-term anticoagulation (p < 0.001) and antiplatelet therapy (p < 0.001). Table 3 presents the results of mortality predictors in patients with HMs and AHF. Supplementary Table 2 describes the complications of HMs.
Mortality in patients with hematological malignancies and AHF.
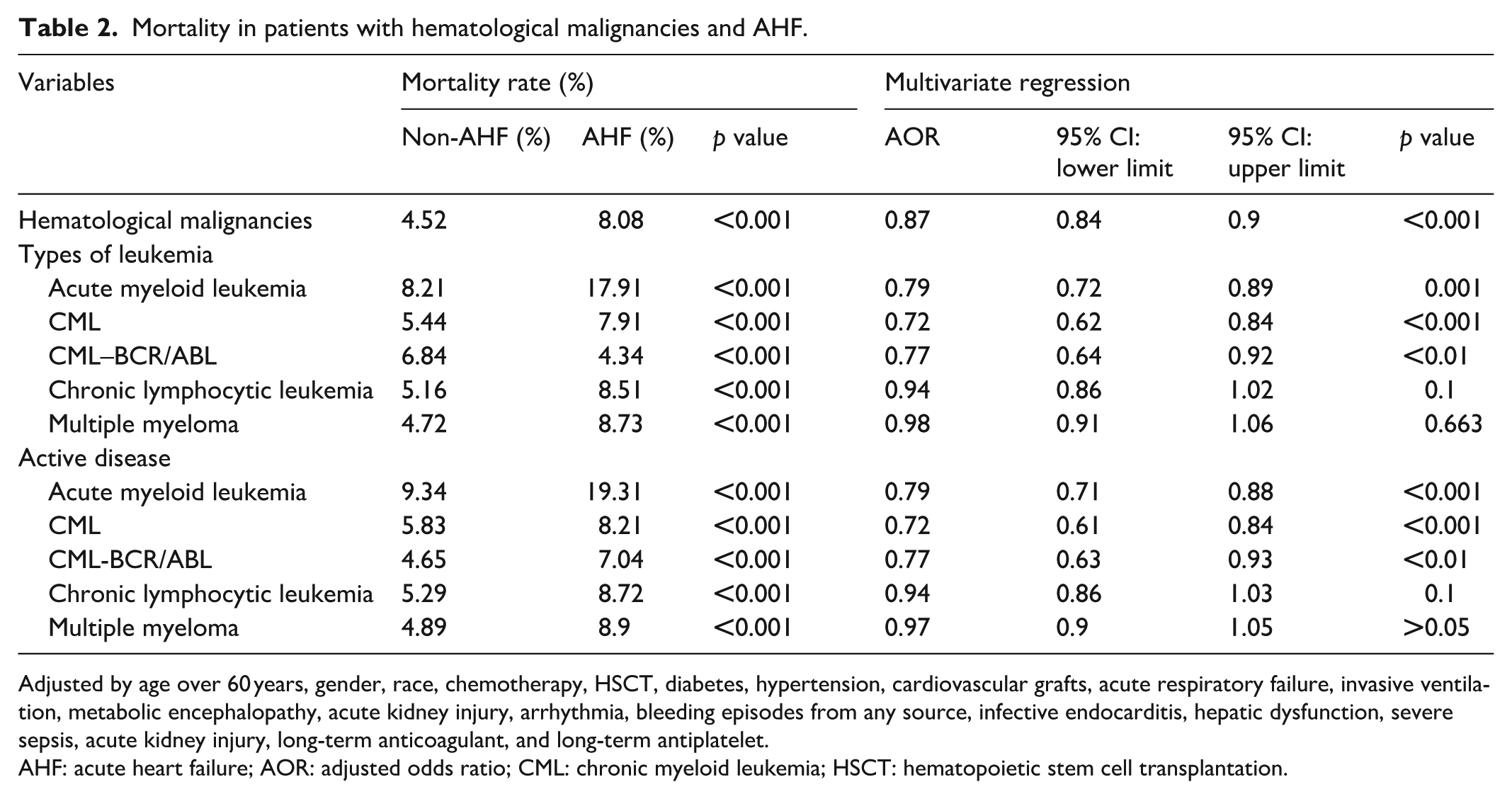
Adjusted by age over 60 years, gender, race, chemotherapy, HSCT, diabetes, hypertension, cardiovascular grafts, acute respiratory failure, invasive ventilation, metabolic encephalopathy, acute kidney injury, arrhythmia, bleeding episodes from any source, infective endocarditis, hepatic dysfunction, severe sepsis, acute kidney injury, long-term anticoagulant, and long-term antiplatelet.
AHF: acute heart failure; AOR: adjusted odds ratio; CML: chronic myeloid leukemia; HSCT: hematopoietic stem cell transplantation.
Mortality predictors in patients with hematological malignancies and acute heart failure.

AOR: adjusted odds ratio; HSCT: hematopoietic stem cell transplantation.
Arrhythmia included atrial fibrillation, atrial flutter, ventricular fibrillation, and ventricular tachycardia.
Acute myeloid leukemia
Of the 79,100 patients with AML, the unadjusted mortality was significantly higher in the AHF group (17.91% versus 8.21%, p < 0.001). AHF was associated with reduced mortality (OR 0.79, 95% CI 0.72–0.89, p = 0.001). Predictors of mortality in the AHF cohort were age >60 years (p < 0.01), severe sepsis (p < 0.001), bleeding from any source (p < 0.05), acute respiratory failure (p < 0.001), hepatic dysfunction (p < 0.001), metabolic encephalopathy (p < 0.001), and invasive ventilation (p < 0.001). Mortality risk was lower in patients with hypertension (p < 0.05), chemotherapy (p < 0.01), and long-term anticoagulation (p < 0.01). Supplementary Table 3 describes the characteristics of the subgroups of HMs.
Subset analysis of 65,792 patients with active AML revealed a higher in-hospital mortality between AHF and non-AHF cohorts (19.31% versus 9.34%, p < 0.001). After adjustment, AHF remained associated with lower odds of mortality (OR 0.79, 95% CI 0.71–0.88, p < 0.001).
Chronic myeloid leukemia
Of the 34,737 patients with CML, the in-hospital mortality was significantly higher in the AHF cohort compared to the non-AHF group (7.91% versus 5.44%, p < 0.001), with an adjusted odds ratio of 0.72 (95% CI 0.62–0.84, p < 0.001). Predictors of mortality in the AHF cohort were age over 60 (p < 0.001), severe sepsis (p < 0.001), acute respiratory failure (p < 0.001), hepatic dysfunction (p < 0.001), metabolic encephalopathy (p = 0.001), and invasive ventilation (p < 0.001). Also, mortality risk was lower in African Americans (p < 0.05), diabetes (p < 0.05), hypertension (p < 0.001), and long-term antiplatelet therapy (p < 0.05).
Of the 30,281 patients with active CML, the unadjusted mortality was significantly higher in the AHF group (8.21% versus 5.83%, p < 0.001), and AHF was associated with reduced mortality (OR 0.77, 95% CI 0.63–0.93, p < 0.01).
Patients with both CML and BCR/ABL-positive mutations (CML/BCR–ABL+) were numbered at 27,240. Their unadjusted mortality was higher in the AHF group (6.84% versus 4.34%, p < 0.001) but was associated with reduced mortality risk (OR 0.77, 95% CI 0.64–0.92, p < 0.01). Patients with only the active disease were noted to be 23,131. Similarly, their unadjusted mortality was higher and adjusted mortality was lower in the AHF cohort (7.04% versus 4.65%, p < 0.001) and (OR 0.77, 95% CI 0.63–0.93, p < 0.01), respectively.
Chronic lymphocytic leukemia
Of the 95,552 patients with chronic lymphocytic leukemia, the in-hospital mortality was higher with AHF (8.51% versus 5.16%, p < 0.001), but there was no significant difference in adjusted mortality (OR 0.94, 95% CI 0.86–1.02, p = 0.1). In the AHF cohort, mortality risk was higher in patients with age over 60 (p < 0.001), severe sepsis (p < 0.001), acute respiratory failure (p < 0.001), hepatic dysfunction (p < 0.001), metabolic encephalopathy (p < 0.001), and invasive ventilation (p < 0.001). Mortality risk was lower in females (p < 0.01), diabetes (p < 0.01), hypertension (p = 0.001), and long-term anticoagulation (p < 0.01) or antiplatelet (p < 0.001).
Of the 86,996 patients with active chronic lymphocytic leukemia, the in-hospital mortality of the AHF cohort was higher than that of the non-AHF cohort (8.72% versus 5.29%, p < 0.001), but the association was not significant on multivariable analysis (OR 0.94, 95% CI 0.86–1.03, p = 0.1).
Multiple myeloma
Of the 141,033 patients with MM, the in-hospital mortality of the AHF cohort was higher than that of the non-AHF cohort (8.73% versus 4.72%, p < 0.001), and adjusted analysis showed no significant association (OR 0.98, 95% CI 0.91–1.06, p = 0.663). In the AHF cohort, mortality risk was higher in age over 60 (p < 0.05), severe sepsis (p < 0.001), bleeding from any source (p < 0.001), arrhythmias (p < 0.001), acute respiratory failure (p < 0.001), hepatic dysfunction (p < 0.001), metabolic encephalopathy (p < 0.001), and invasive ventilation (p < 0.001). Also, mortality risk was lower in African Americans (p < 0.01), hypertension (p < 0.001), long-term anticoagulation (p = 0.001), and antiplatelet therapy (p < 0.001).
Of the 129,379 patients with active disease, the in-hospital mortality of the AHF cohort was higher than that of the non-AHF cohort (8.9% versus 4.89%, p < 0.001), but after adjustment, mortality did not differ between the two cohorts (0.97, 95% CI 0.9–1.05, p > 0.05).
Discussion
In our study, patients with AHF exhibited a higher in-hospital mortality compared to those without AHF across the entire HMs cohort, and their subtypes. However, after adjusting for sociodemographic factors and comorbidities, mortality risk was lower among patients with HMs (p < 0.001) and the subtypes of AML (p < 0.001) and CML (p < 0.001) in the AHF cohort. No significant difference in mortality was observed between the AHF and non-AHF groups in the chronic lymphocytic leukemia (p > 0.05) and MM (p > 0.05) patients. Based on our analysis, AHF alone does not appear to be the primary driver of mortality. Multivariate regression across the HMs cohort indicates that comorbid conditions such as arrhythmias and a history of HSCT, along with complications like severe sepsis, acute renal failure, bleeding from any source, hepatic dysfunction, and invasive ventilation, independently elevated the mortality risk.
We identified that the co-occurrence of atrial fibrillation, atrial flutter, ventricular fibrillation, and ventricular tachycardias was associated with increased risk of mortality. Alexandre et al. demonstrated that the incidence of atrial fibrillation varies substantially depending on the anticancer agent used, with the highest annualized rates observed with ibrutinib (4.92/100 person-years), clofarabine (2.38), and ponatinib (2.35). Notably, the incidence of atrial fibrillation increases in patients with acute leukemia, chemotherapy-induced cardiomyopathy, and acute decompensation due to sepsis or organ dysfunction. 23 Similarly, QT prolongation, ventricular arrhythmias, and bradyarrhythmias can also negatively impact clinical outcomes in this population.24,25 In our study, bleeding from any source was associated with increased mortality. Bleeding in patients with HMs can occur due to bone marrow failure or infiltration, invasive procedures, and therapy-related toxicities. 26 This finding is strongly supported by prior data in patients receiving anthracycline-based induction chemotherapy. Clinically significant bleeding was associated with a fivefold increased risk of mortality within the first 60 days (HR: 5.24; 95% CI: 2.50–10.96; p < 0.001). 27 Hemorrhagic complications also occur in the absence of chemotherapy or anticoagulation use.28–30 Cordella et al. reported that eight out of nine hemorrhagic events occurred in patients not receiving anticoagulation. 31 The incidence of major bleeding was 14.4%, with the most common sites being the central nervous system, gastrointestinal tract, and pulmonary system.26,32–34 The use of fresh-frozen plasma or antithrombin concentrate in managing such bleeding complications remains controversial. 35 Therefore, close monitoring and individualized risk assessment are essential to guide transfusion decisions and improve outcomes in this high-risk population.
HSCT and the usage of cardiotoxic agents such as anthracyclines, proteasome inhibitors, and tyrosine kinase inhibitors (TKIs) are common in this population. Their use has been associated with increased risk of heart failure and cardiovascular death.36–39 We identified that in patients with AML and AHF, a lower mortality risk was associated with administration of inpatient chemotherapy (p = 0.01) and HSCT (p < 0.1). However, the incidence of heart failure at 5 years post-HSCT transplant rises to 5.0% in autologous and 6.0% in allogeneic HSCT recipients. 40 Hence, cardiovascular risk assessment and surveillance in HMs both before and after HSCT are crucial. 41 Anthracyclines have been used in both upfront and salvage treatment of AML. In patients with AML, heart failure often occurs within the first year of anthracycline exposure. The prevalence of symptomatic heart failure in this group has been reported at 9%, higher than the 2%–5% seen in the general cancer population receiving anthracyclines. 9 TKIs have significantly improved CML outcomes but are associated with a 2.4-fold increased risk of heart failure (OR 2.4, 95% CI 2.2–2.6).42–44 MM patients are at a higher risk of cardiac amyloidosis and anemia. 45 Treatments for MM were associated with high-grade cardiac toxicity reported for immunomodulatory drugs (OR ~2) and proteasome inhibitors (OR 1.67–2.68). 46 Similarly, in a prospective study investigating the incidence of cardiac events following idecabtagene vicleucel (CAR T cell therapy for relapsed and refractory cases), over 50% of patients experienced grade 3 or higher cardiovascular adverse events. The most common adverse event was heart failure (20%). 47
These findings suggest that the prognostic impact of AHF may differ across HM subtypes and is potentially influenced by underlying disease biology or treatment responsiveness. 48 AHF is recognized as a serious adverse cardiac effect, marked by sudden decompensation of cardiac function, often triggered by cardiotoxic therapies or disease-related cardiac stress. Supporting this, HM patients have been shown to exhibit lower collagen fiber alignment, fewer cardiomyocyte nuclei, and elevated B-type natriuretic peptide levels, all of which reflect compromised myocardial integrity and increased susceptibility to cardiac dysfunction. 15 Moreover, another study reported a 3.22-fold higher risk of heart failure in cancer survivors with hematopoietic and lymphatic malignancies compared to cancer-free individuals (95% CI 2.51–4.18). 49 This study analyzed a large sample of AHF in HMs and their subtypes, yielding several notable findings.
Limitations were present in this study that must also be acknowledged. The NIS database lacked detailed information on laboratory values and specific pharmacological treatments, such as immunomodulators, chemotherapeutic regimens, vasopressors, and diuretics. The study was unable to determine the indication for invasive ventilation. The assessment of patient-specific outcomes, such as volume status and quality of life, was limited. Due to the cross-sectional design, follow-up information and data on hospital readmissions were not available for analysis. Since this is a registry-based database, a predetermined sample size was not calculated, and there is a potential for coding errors that could introduce bias.
Conclusion
AHF appeared as a significant clinical marker in patients with HMs. A large proportion of those who died had AHF, suggesting a potential association. However, after adjusting for confounding factors, AHF was paradoxically associated with a lower mortality risk. This may be influenced by the higher mortality seen in patients with severe complications such as sepsis and multiorgan dysfunction, which often overshadow the impact of AHF itself. Thus, early recognition of severe sepsis and a systematic approach to managing severe organ dysfunction may contribute to better outcomes. In an effort to improve patient outcomes, we recommend close monitoring and a multidisciplinary team approach to effectively manage severe sepsis and organ dysfunction. There is limited research directly linking AHF with outcomes in patients with HMs, and the specific impact of AHF alone remains unclear. Further prospective studies are needed to better understand the long-term outcomes and prognostic significance of AHF in this high-risk population.
Supplemental Material
sj-docx-1-smo-10.1177_20503121251370940 – Supplemental material for Acute heart failure in patients with hematologic malignancies: A nationwide analysis 2016–2021
Supplemental material, sj-docx-1-smo-10.1177_20503121251370940 for Acute heart failure in patients with hematologic malignancies: A nationwide analysis 2016–2021 by Barath Prashanth Sivasubramanian, Diviya Bharathi Ravikumar, Siva Naga S. Yarrarapu, Ragavendar Saravanabavanandan, Ibthisam Ismail Sharieff, Shashvat Joshi, Deepthi Karunakar, Visalakshi Kannathal Vairavan, Indrajith Devandra Balamurugan, Umabalan Thirupathy, Vikramaditya Samala Venkata, Yash Garg and Aditya Kumar Ghosh in SAGE Open Medicine
Footnotes
Author contributions
Barath Prashanth Sivasubramanian: conceptualization, methodology, data curation, formal analysis, writing – original draft. Diviya Bharathi Ravikumar: investigation, methodology, writing – original draft, writing – review and editing. Siva Naga S. Yarrarapu: conceptualization, methodology, writing – review and editing. Ragavendar Saravanabavanandan: validation, writing – review and editing. Ibthisam Ismail Sharieff: writing – original draft. Shashvat Joshi: writing – original draft. Deepthi Karunakar: writing – original draft. Visalakshi Kannathal Vairavan: writing – original draft. Indrajith Devandra Balamurugan: writing – original draft. Umabalan Thirupathy: supervision, writing – review and editing. Vikramaditya Samala Venkata: supervision, writing – review and editing. Yash Garg: supervision, writing – review and editing. Aditya Kumar Ghosh: conceptualization, supervision, writing – review and editing. All authors reviewed and approved the final article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
