Abstract
Aims:
To assess whether baseline characteristics (age, New York Heart Association functional class, left ventricular ejection fraction, glomerular filtration rate, systolic blood pressure, and N-terminal pro-brain natriuretic peptide) predicted different survival functions in patients with advanced heart failure, treated with intermittent inotropics.
Methods:
Survival analysis based on a retrospective cohort of patients with advanced heart failure, treated with intermittent infusion of levosimendan and managed at heart failure clinic of the Hospital Universitario San Ignacio, in Bogotá (Colombia). We evaluated which baseline characteristics were associated with worse survival outcomes, using Kaplan–Meier curves and log rank test.
Results:
Forty-two patients with advanced heart failure were included (mean age: 69.5 ± 13.2 years, 97.6% New York Heart Association III–IV, 54.7% of the population had quadruple therapy). A total of 39.5% died during 1-year follow-up. Survival functions were worse in patients with N-terminal pro-brain natriuretic peptide levels >6000 pg/mL (HR: 2.72; 95% CI: 1.01–8.01, p = 0.0493). In contrast, no significant difference in mortality was found for those with basal GFR <60 versus ⩾60 ml/min (p = 0.1030), left ventricular ejection fraction ⩽20% versus >20% (p = 0.1040), systolic blood pressure <90 mmHg versus ⩾90 mmHg (p = 0.2463), according to functional class (p = 0.840) or age > 65 years (p = 0.169). A significant increase of left ventricular ejection fraction measurements was observed in 26 surviving patients at 12 months of follow-up (20% vs 34%, p < 0.0001).
Conclusion:
Our preliminary data suggest that the only characteristic that could predict worse survival in patients with advanced heart failure treated with levosimendan is N-terminal pro-brain natriuretic peptide levels greater than 6000 pg/ml. Future research is needed to confirm our findings.
Keywords
Introduction
Heart failure is a major public health problem. It affects about 64 million people worldwide, with prevalence projected to increase by 46% in the United States. 1 Annually, the medical costs exceed $10,000 USD per person and is estimated to be $53 billion for the total population by 2030. 2
Despite optimal evidence-based treatment, advanced heart failure occurs in about 1%–10% of the heart failure population, with a 5-year mortality rate of 80%. 3 At this stage, additional therapeutic strategies such as heart transplantation, implementation of ventricular assist, intermittent inotropics, or end-of-life care are needed.3,4
The use of inotropic drugs has confirmed improvement in hospitalization rate, functional class, N-terminal pro-brain natriuretic peptide (NT pro-BNP) levels and recently, the overall 1-year survival (86%) with a low incidence of adverse events, as was shown in the RELEVANT-HF (REpetitive LEVosimendan in AdvaNced refracTory Heart Failure) registry collaboration. 5 However, it has not been assessed whether the baseline characteristics of the patients might suggest an impact on survival in this population.
This study seeks to identify whether the baseline characteristics of the population (age, New York Heart Association (NYHA) functional class, and left ventricular ejection fraction (LVEF)) predict different survival functions in adult patients with advanced heart failure undergoing intermittent infusion with levosimendan between 2020 and 2022 in a reference hospital in the city of Bogota, Colombia.
Methodology
We conducted a survival analysis study based on a retrospective cohort of patients with advanced heart failure treated with intermittent infusion of levosimendan in the heart failure clinic of the San Ignacio University Hospital, Bogota (Colombia), between 2020 and 2022. Patients over 18 years of age, with a diagnosis of advanced heart failure (defined according to the criteria proposed by the Heart Failure Association of the European Society of Cardiology), 3 and with availability of basal NT pro-BNP levels, glomerular filtration rate, systolic blood pressure, LVEF, and functional class (NYHA) evaluation, were included. Patients were excluded if they required a left ventricular assist device (LVAD) from the time of diagnosis, had drug intolerance due to significant hypotension (defined as systolic blood pressure <80 or <100 mmHg associated with symptoms) according to LIONHEART (Levosimendan Intermittent administration in Outpatients: effects on Natriuretic peptides in advanced chronic HEART failure) definitions, 6 and did not complete cycles of levosimendan intermittent infusion. The study was approved by the ethics committee of the Hospital Universitario San Ignacio and the Faculty of Medicine of the Pontificia Universidad Javeriana. Written informed consent was exempted by the Institutional Review Board.
The data were obtained from a thorough review of the institutional electronic medical records performed by the cardiology group at each consultation. Sociodemographic variables, comorbidities, time of diagnosis of heart failure, date and adverse events of levosimendan infusion, natriuretic peptide levels, functional class during follow-up, hospital admission for heart failure, and survival were systematically collected in a standardized format. Missing and extreme data and patients lost to follow-up were checked and verified to ensure data quality.
Treatment compliance was defined as completion of 6 cycles of intravenous infusions of levosimendan (dose: 0.2 µg/kg/min) for 6 h every 2 weeks, according to LIONHEART protocol. 6 The criterion of hospitalization for decompensated heart failure was validated by review of medical records. Functional class and ejection fraction were estimated using the NYHA Scale and the Simpson method by echocardiography, respectively. Measurements of NT pro-BNP levels were performed by immunoassay techniques using chemiluminescence in the central laboratory. The cut point of 6000 pg/ml to analyze subgroups according to NT pro-BNP levels were taken from Pereira-Barretto et al.’s 7 study. Categories of GFR were defined with KDIGO (1–2 vs 3–5). Subgroup analysis according to LVEF used a cut point of 20% as there were not previously defined categories in patients with LVEF < 30%.
Finally, survival, defined as the time that elapses from the date of diagnosis and start of treatment until the patient is still alive, was supplemented by means of the Administrator of the Resources of the General Social Security Health System (ADRES) information system, a national and updated database that has high quality and verified information on the vital status of persons enrolled in the Colombian health system (https://servicios.adres.gov.co/BDUA/Consulta-Afiliados-BDUA).
Statistical analysis
We estimated the sample size using Freedman’s method, allowing for an alpha error of 0.05 with a power of 80%, assuming a delta of HR of 0.40. The calculated sample size was 42 patients. 8 Categorical variables were described as absolute and relative frequencies. Continuous variables were presented as mean and standard deviation or as median and interquartile range according to the distribution of the data. The Shapiro–Wilk test was used to estimate the normality assumption. Kaplan–Meier curves and Cox proportional model were used to assess survival functions and the Log Rank test statistic was used to compare these functions by proBNP levels, basal LVEF, age, functional class, systolic blood pressure, and glomerular filtration rate. Cardiac transplant events were not considered as competing risk as no patient presented mortality in the time period evaluated. Additionally, a sensitivity analysis was performed that excluded patients who underwent cardiac transplantation, and similar results were obtained. To evaluate changes over time in LVEF and NT pro-BNP levels, the paired t-test was used, considering that these were repeated measurements of the same population. Statistical analyses were performed with Stata software (Stata Statistical Software: Version 17; Stata Corp LLC, College Station, TX, USA).
Results
A total of 42 patients were finally included in the analysis. The baseline characteristics at study entry are shown in Table 1. A majority of the population were older adults and were male (69%); the main comorbidities were chronic kidney disease (76.1%), acute myocardial infarction (50%), arterial hypertension (40.4%), type 2 diabetes mellitus (42.8%), and atrial fibrillation (38.1%). A total of 97.6% of the patients had NYHA functional classes III–IV and 38% had NT pro-BNP levels above 6000 pg/ml. A total of 54.7% of the population had quadruple therapy with beta-blocker management in its entirety. A total of 25.5% of the patients required hospital admission for decompensated heart failure. Nobody had drug intolerance or required a ventricular assist device.
Demographic characteristics, clinical variables and NT pro-BNP levels of patients with advanced heart failure at the start of treatment with levosimendan.

NYHA: New York Heart Association, NT pro-BNP: N-terminal pro-brain natriuretic peptide, IQR: Interquartile range, ACEI/ARA2: Angiotensin-converting enzyme inhibitor/angiotensin II receptor antagonists, ARNI: Angiotensin/neprilysin receptor inhibitors, iSGLT2: sodium-glucose cotransporter type 2 inhibitor, iSGLT2: sodium-glucose cotransporter inhibitor.
Chronic kidney disease: defined as GFR less than 60 ml/min/1.73 m2.
The outcomes associated with the intervention are presented in Table 2. Four patients did not complete the proposed 6 cycles as they died during the first 12 weeks of follow-up. At 1 year follow-up, the mortality rate was 39.5%. Of the total number of patients, six received cardiac transplantation and none had management with LVADs. The main complication of intermittent infusion of levosimendan was hypotension, which was not significant (6.98%).
Indications for infusion, clinical variables, treatment, and complications during infusion and clinical outcomes in patients with advanced heart failure undergoing treatment with levosimendan.

Dose taken from the last infusion session.
Low dose: bisoprolol < 5 mg/day or metoprolol <50 mg/day.
When evaluating survival according to the baseline characteristics of the population in the univariate model, we found higher mortality in patients with NT pro-BNP levels >6000 versus <6000 (HR: 2.72; 95% CI: 1.01–8.01, p = 0.0493; Figure 1). In contrast, a not significant difference in mortality was found according to basal GFR (<60 vs ⩾60 ml/min; p = 0.1030; Figure 2), and with LVEF ⩽20% versus >20% (p = 0.1040; Figure 3). Similarly, there were no differences for those older than 65 years versus younger than 65 years (p = 0.169), or according to functional class (p = 0.840) or systolic blood pressure (p = 0.246). Similar results were obtained from the sensitivity analysis that excluded patients who underwent cardiac transplantation, demonstrating that NT pro-BNP levels >6000 (p = 0.05) were the only characteristic associated with different survival. The multivariate Cox model showed again that the only characteristic associated with mortality was proBNP levels >6000 (HR: 3.09, 95% CI: 1.06–11.04, p = 0.049).

Kaplan–Meier survival curves according to NT pro-BNP levels.
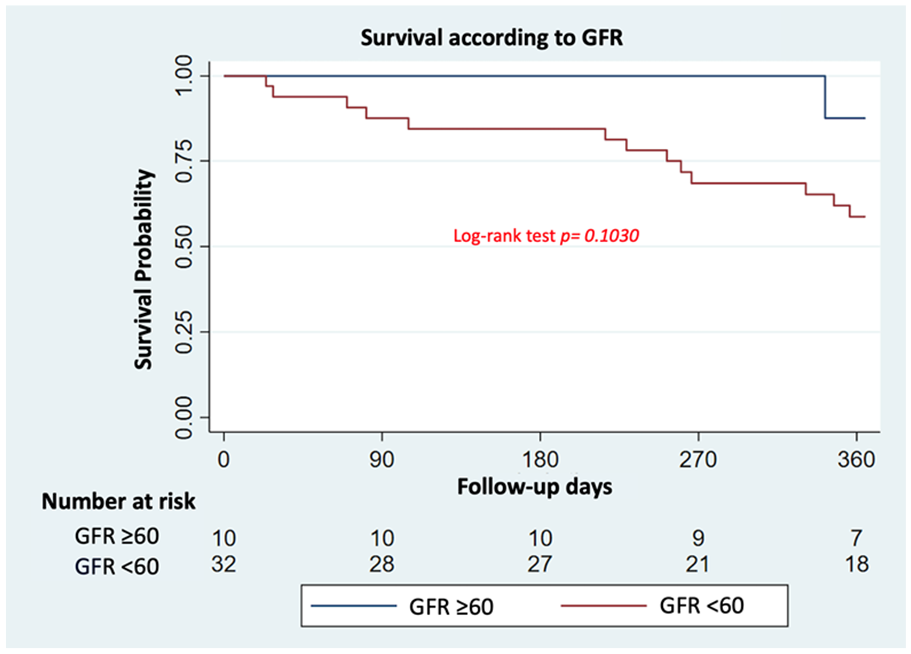
Kaplan–Meier survival curves according to glomerular filtration rate.

Kaplan–Meier survival curves according to LVEF.
Changes in LVEF and NT pro-BNP at 12 months are shown in Table 3. When comparing the changes in LVEF measurements, a significant increase was observed in 26 surviving patients at 12 months (p < 0.0001). Regarding NT pro-BNP levels at 6 months in the 35 survivors, no significant reduction was observed (6721 ± 11.728 vs 4859 ± 5839 pg/ml, p = 0.176). Similarly, at 12 months, the survivors did not have a reduction in peptide levels (p = 0.7560; Table 3).
Changes over time in NT pro-BNP and LVEF levels in patients with advanced heart failure who underwent treatment with levosimendan.

n: number of patients at 1 year of follow-up; LVEF: left ventricular ejection fraction; NT pro-BNP: N-terminal pro-brain natriuretic peptide.
Discussion
In this study, we evaluated whether baseline characteristics predict 1-year survival in a population with advanced heart failure who received management with levosimendan. Our results showed a significant difference according to NT pro-BNP levels with a cut-off point greater than 6000 pg/ml; with no evidence of statistically representative changes when assessing GFR, systolic blood pressure, age, LVEF, or functional class.
A comparison of our overall survival data with that of other published studies reveals a mortality rate of 11.6% at the 6-month follow-up point, which is lower than the rates reported in the LION-HEART 6 and LevoRep 9 trials. It is noteworthy that, despite the demographic and clinical similarities between our population and those in the aforementioned studies, the pharmacological management differed with respect to the use of angiotensin receptor-neprilysin inhibitor and sodium-glucose cotransporter-2 inhibitors (iSGLT2). Additionally, our patients were administered beta-blockers exclusively, and 54.7% of them received quadruple therapy. The introduction of these pharmaceutical agents has had a substantial influence on the outcomes of hospitalization and mortality due to heart failure, largely due to their anti-inflammatory and antifibrotic effects.10,11 This reflects the growing importance of advances and adherence in the management of heart failure over the past decade.
Our findings indicate that a baseline level of NT pro-BNP exceeding 6000 pg/ml is predictive of reduced survival. This finding is consistent with the results of a previous study conducted in South America, which demonstrated that patients with advanced heart failure and NT pro-BNP levels above 6000 pg/ml had a higher probability of mortality within 90 days. 7 Furthermore, NT pro-BNP levels are elevated not only by cardiac dysfunction but also by aging, the presence of atrial fibrillation, and deterioration in the glomerular filtration rate. These conditions are associated with increased mortality and adverse clinical outcomes. 12 Our findings suggest that when intervening with intermittent inotropic therapy in patients with advanced heart failure, subpopulations should be differentiated according to NT pro-BNP levels. This allows for a more rigorous and frequent follow-up of those with values above 6000 pg/ml, as they are at a higher risk of mortality.
When evaluating the impact of chronic kidney disease, defined as GFR < 60 ml/min/1.73 m2, we observed a nonsignificant trend of lower survival. These findings are consistent with those reported in the literature,13–16 which indicates that renal impairment is a predictor of poor prognosis in patients with heart failure and is associated with an increased risk of premature death and hospitalizations. Furthermore, there is an important limitation in the administration of optimal medical therapy, as most randomized clinical trials have excluded this population. This is due to the fact that several drugs affect renal hemodynamics and generate severe hydroelectrolytic alterations. In our study, despite the high prevalence of Chronic kidney disease (CKD), the majority of cases were classified as stages 3a, 3b, and 4. This allowed us to administer quadruple therapy without compromising its efficacy and potentially delaying the progression of heart disease and the deterioration of renal function.
An ejection fraction of less than 20% does not correlate with a reduced probability of survival. This finding is consistent with the literature, which indicates that advanced heart failure has a higher mortality rate due to its inherent severity. There are no differences in survival when comparing groups with preserved ejection fraction, slightly reduced ejection fraction, or reduced ejection fraction. 17 It is important to note that previous studies have documented additional echocardiographic findings that are associated with an increased risk of mortality. These include indexed left atrial volume, mitral E velocity, septal and lateral e’ velocity, as well as the E/e’ ratio. These findings highlight the importance of diastolic dysfunction in the prognosis of patients with heart failure. 18 Our findings suggest that further studies should be conducted to evaluate the echocardiographic variables of both diastolic and systolic function of both ventricular cavities, as well as their relationship with mortality in patients managed with intermittent inotropy.
Another finding of our study is that the group of adults older than 65 years showed a tendency to a lower survival rate, which is consistent with the findings in the literature, where morbidity and mortality increase linearly with age, as well as the number of hospitalizations for decompensated heart failure and treatment failure. 19
The results of our study indicate that the administration of levosimendan did not lead to a reduction in NT pro-BNP levels. This finding may seem inconsistent with the established efficacy of levosimendan in reducing congestion, which is attributed to its positive lusitropic effect, specifically the reduction of left atrial pressure and tricuspid regurgitation velocity. 17 These hemodynamic changes are postulated to reduce ventricular wall stress and peptide release. However, it is important to recognize that decreases in NT pro-BNP in heart failure are a favorable sign but do not always parallel changes in ejection fraction. Renal impairment and atrial fibrillation may confound interpretation by increasing NT pro-BNP independent of myocardial wall stress. Furthermore, a significant proportion of our population had dilated cardiomyopathy associated with Chagas disease, in which the prognostic role of natriuretic peptides remains uncertain. 20 This finding reminds us to always interpret NT pro-BNP in the context of volume status, rhythm, renal function, and other clinical markers (such as symptoms, echo, and weight changes).
Furthermore, alterations in ejection fraction have been among the echocardiographic parameters assessed in published trials involving levosimendan, demonstrating a notable enhancement in LVEF and even cardiac power at rest. 17 The results of our study were comparable, with an absolute 10% increase in LVEF observed. This finding can be attributed to the positive inotropic effects of levosimendan, which enhances the interaction between the mechanical pump and peripheral vascular resistance, thereby optimizing cardiac output and myocardial metabolism. 21 It is important to note that these findings were evaluated only in patients who survived the 1-year follow-up period (26 patients). This suggests that an improvement in LVEF may serve as a favorable prognostic factor.
Regarding the use of levosimendan in patients with acute decompensated heart failure due to Chagas’ disease, the BELIEF study conducted in Brazil included 28 patients with this etiology. The results showed that levosimendan was as effective in these patients as in those with other causes of heart failure, although a higher incidence of hypotension was observed in the Chagas group. 22 Currently, there is no specific published evidence on the use of the LIONHEART regimen in patients with advanced Chagas heart disease. Given the unique characteristics of Chagas heart disease, such as a higher predisposition to arrhythmias and thromboembolic events, it is essential to conduct specific studies to evaluate the efficacy and safety of the LIONHEART regimen in this population. In our study, 13.9% of patients had Chagas as the etiology of heart failure, with results similar to those reported for other etiologies. As this information is preliminary, the use of levosimendan in patients with Chagas disease should be considered with caution and based on an individualized assessment of each case.
With regard to the study’s limitations, the relatively small sample size limits the likelihood of identifying small differences in population survival and makes it challenging to assess the individual impact of each factor in a multivariate analysis (Cox regression model). However, our study represents the largest cohort in Latin America and have a similar sample size compared to the pivotal trials of levosimendan.6,9 To achieve more precise assessments and control for multiple confounding factors, larger multicenter trials will be needed in the future. Similarly, some missing data as the number of hospitalizations in the years previous to the admission to our program could limit the evaluation of the basal severity of heart failure, and the missing information about hospitalizations in other institutions during the follow-up may have been present; however, the loss of this information was reduced by telephone follow-up by the failure clinic. This did not affect the mortality estimates, as we had access to a complementary, updated, and high-quality database through ADRES.
Conclusion
Our preliminary data suggest that the only characteristic that could predict worse survival in patients with advanced heart failure prior to intermittent infusion with levosimendan, is NT pro-BNP levels greater than 6000 pg/ml. Serial measurements of NT pro-BNP levels may prove useful in identifying patients at higher risk and guiding management in heart failure clinics. Future research is needed to confirm our results.
Footnotes
Acknowledgements
We thank our patients and their families for their trust, collaboration, and support.
Ethical considerations
The Ethics Committee of Hospital Universitario San Ignacio approved the study, approval number FM-CIE-0320-21.
Consent to participate
Written Informed Consent was exempted by the Institutional Review Board.
Author contributions
Angel Alberto Garcia Peña: Conceptualization; Formal analysis; Methodology; Project administration; Supervision; Validation; Visualization; Writing – review & editing. Alejandro Mariño: Conceptualization; Formal analysis; Methodology; Supervision; Validation; Writing – review & editing. Daniela Saa González: Conceptualization; Data curation; Formal analysis; Investigation; Methodology; Project administration; Software; Validation; Visualization; Writing – original draft; Writing – review & editing. Oscar Mauricio Muñoz-Velandia: Conceptualization; Formal analysis; Investigation; Methodology; Project administration; Software; Supervision; Validation; Visualization; Writing – original draft; Writing – review & editing.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
