Abstract
Objectives:
The aim of this study was to determine if carvedilol improved structural and functional changes in the left ventricle and reduced mortality in patients with hypertensive heart disease.
Methods:
Blood pressure, heart rate, echocardiographic parameters, and laboratory variables, were assessed pre and post treatment with carvedilol in 98 eligible patients.
Results:
Carvedilol at a median dose of 50 mg/day during the treatment period in hypertensive heart disease lowered blood pressure 10/10 mmHg, heart rate 10 beats/min, improved left ventricular ejection fraction from baseline to follow-up (median: 6 years) (36%–47%)) and reduced left ventricular end-diastolic and end-systolic dimensions (62 vs 56 mm; 53 vs 42 mm, respectively, all p-values <0.01). Left ventricular ejection fraction increased in 69% of patients. Patients who did not have improved left ventricular ejection fraction had nearly six-fold higher mortality than those that improved (relative risk; 5.7, 95% confidence interval: 1.3–25, p = 0.022).
Conclusion:
Carvedilol reduced cardiac dimensions and improved left ventricular ejection fraction and cardiac remodeling in patients with hypertensive heart disease. These treatment-related changes had a favorable effect on survival.
Keywords
Introduction
The impact of beta-blockers on morbidity and mortality due to heart failure with various etiologies has been demonstrated in large studies.1–3 Reverse remodeling of the left ventricle may occur more or less frequently according to the etiology of heart failure. Cardiac remodeling includes structural and functional changes of cardiac muscle, interstitium, and vessels, 4 which reflect genetic, electric, 5 and cellular factors. 6 Hypertension is an important stimulus to cardiac remodeling. 7 Cardiac modifications from hypertension reflect hemodynamic overload, ischemia, neurohumoral alterations, and activation of inflammatory cytokines. 4 Collectively, these alterations can lead to hypertensive heart disease (HHD). 8 The clinical manifestations of HHD include left ventricular (LV) hypertrophy, myocardial ischemia, arrhythmia, and heart failure (HF). 9
HF is more frequent in elderly people with a prevalence of 11.5% in the American population (⩾80 years) between 2009–2012. 10 Hypertension, as the only risk factor, accounts for approximately 4% of HF among adults in North America 11 and a similar prevalence of HF in Europe, 12 while hypertension in concert with other risk factors precedes HF in 75% of patients. 13 Left ventricular hypertrophy (LVH) by echocardiography is a frequent finding in hypertensive subjects, 14 and the frequency increases with the severity of hypertension. Hypertensive subjects with symptomatic HF can present with preserved (⩾50%), mid-range (40%–49%), and reduced (<40%) ejection fraction (EF). 15 Antihypertensive treatment can improve LVH and reduce HF in hypertensive patients. 16 More recently, the question has been raised on whether antihypertensive treatment can reverse structural and functional changes in HHD.
The current literature is limited to a few studies with small numbers of HHD patients.17–19 In one study, long-term antihypertensive therapy failed to normalize LV systolic dysfunction in hypertensive patients with HF and reduced EF. 19
Beta-blockers, including carvedilol, metoprolol, and bisoprolol have improved left ventricular ejection fraction (LVEF) and decreased morbidity and mortality from HF. 20 Carvedilol reduced the risk of death and hospitalization for cardiovascular causes in HF patients receiving digoxin, diuretics, and renin-angiotensin system inhibitors. 17 In hypertensive patients, carvedilol also reduced cardiovascular morbidity, mortality, and HF post-myocardial infarction (MI).1,2 However, the effect of carvedilol on LV structure and function as well as mortality in patients with HHD has not been reported.
The aim of the study was to evaluate whether carvedilol is effective in reversing structural and functional changes in the left ventricle and can improve the survival of patients with HHD.
Materials and methods
Study and ethic approval
This retrospective study was conducted in 2015. The sample consisted patients who were treated in a public hospital between 1 January 2003 and 31 December 2013. The Ethics Committee of the Universidade Nove de Julho—UNINOVE determined that informed consent (IC) was not required for this retrospective study devoid of personal identifiers. This study was approved by Ethic Committee from Universidade Nove de Julho—UNINOVE with the number 665293.
Inclusion and exclusion criteria
We selected 1920 patients for this study. From this total, 1822 were excluded; 736 were excluded due to insufficient data, 1025 had exclusion criteria, and 61 did not have a second control echocardiography. The patients were selected by convenience sample. Exclusion criteria included any valvulopathy, non-hypertensive cardiomyopathy, hypothyroidism, hyperthyroidism, infiltrative cardiovascular disease, angina, documented or clinical suspected coronary artery disease (CAD), chronic kidney disease (estimated glomerular filtration rate <30 mL/1.73 m2/min), secondary hypertension, cancer, previous use of chemotherapy, grade III obesity (body mass index (BMI) ⩾40 kg/m2), organ transplantation, diabetes using insulin, or having implantable cardioverter defibrillators.
Collected variables
Hemodynamic evaluation
Heart rate and systolic, diastolic, and mean BP were evaluated before and after carvedilol use. Blood pressure and heart rate were measured with the patients in seated position. Mean blood pressure was calculated.
Biochemistry and hematologic tests
Total cholesterol, low-density lipoprotein (LDL)-cholesterol, high-density lipoprotein (HDL)-cholesterol, glucose, creatinine and hemoglobin, and hematocrit were evaluated with commercial kits prior to and after carvedilol therapy.
Echocardiography
Left ventricular end-diastolic dimension (LVEDD), left ventricular end-systolic dimension (LVESD), aortic root dimension (AoD), left atrium diameter (LAD), interventricular septum thickness (IVST), posterior wall thickness (PWT), EF, left ventricular mass index (LVMI), and relative wall thickness (RWT) were evaluated before and after carvedilol use. 21
Myocardial scintigraphy and coronary angiography
Of 98 study participants, 66 (67%) were evaluated by one or both of these tests. A total of 36 patients had coronary angiography. A total of 30 patients had only myocardial scintigraphy, and 11 patients had both tests.
Statistical analysis
SPSS version 22 (IBM corporation, Armonk, USA) was used for statistical analysis. Normality was tested with Kolmogorov–Smirnov method. Since data for most variables were not normally distributed, findings are reported as median and interquartile range (25th to 75th percentile). Continuous numeric parameters were compared by Wilcoxon test for paired samples; while non-paired variables were compared with Mann–Whitney method. Bivariate analysis with chi-square (χ2) and Fisher exact test were used for categorical variables. The Kaplan–Meier method and log-rank test were used to estimate survival among HHD with and without improvement of EF during carvedilol treatment. P-values <0.05 were considered statistically significant.
Results
Study population
A total of 98 hypertensive (females and males) patients with reduced EF (<45%) were evaluated. Carvedilol was added to their treatment regimens, which included diuretics and renin-angiotensin system blockers for hypertension and systolic HF. Before adding carvedilol half of the hypertensive patients had already attained blood pressure control. Subjects were evaluated at baseline and 6 years (median) after beginning treatment with carvedilol. CAD was excluded by cinecoronariography, computed tomography (CT) scan, or cardiac scintigraphy. Sample size and power estimates were not performed, as this was a convenience sample.
Baseline demographic data and other selected descriptive data are shown in Table 1. All subjects were taking diuretics and renin-angiotensin system blockers and kept medication’s dose during treatment (Table 2). Baseline BMI, blood pressure, heart rate, and biochemistry values for all patients are provided in Table 3. Seven patients (8%) had side effects; two men reported erectile dysfunction, and five patients reported other side effects such as wheezing, dyspnea or dizziness or symptoms consistent with claudication orhypotension.
Demographic and clinical parameters from all subjects.

Antihypertensive, antidiabetic, hypolipidemic, antiarrhythmic, digoxin and RAS inhibitors, frequency use, and doses.

Baseline biochemical, anthropometric, and hemodynamic variables and comparison after carvedilol use.
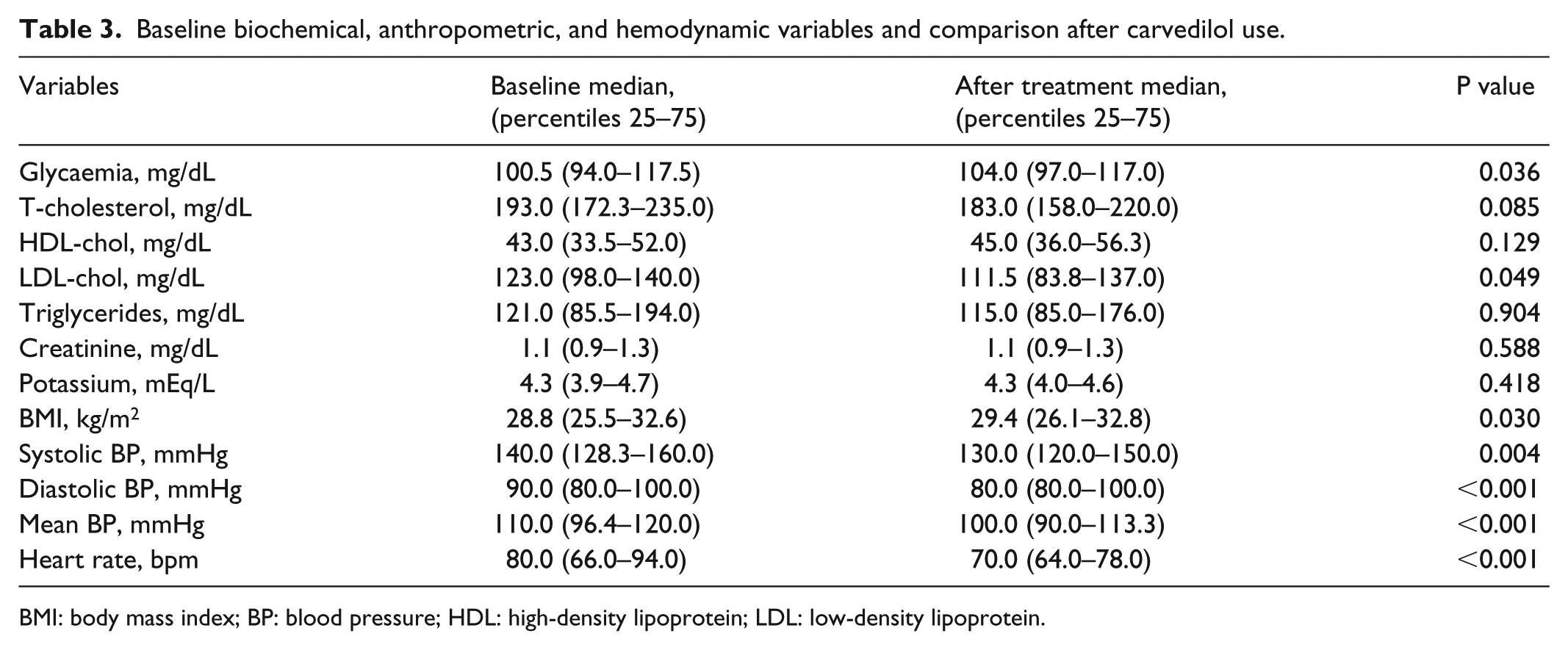
BMI: body mass index; BP: blood pressure; HDL: high-density lipoprotein; LDL: low-density lipoprotein.
Compared to baseline values, addition of carvedilol at a median dose of 50 mg/day for a median of 6 years significantly reduced systolic, diastolic, and mean BP as well as heart rate (Table 3). BMI and blood glucose increased slightly and significantly, while LVESD, LVEDD, and LVMI decreased. LVEF increased by an average of 11% after carvedilol treatment (Table 4). EF improved in 69% but did not change or decreased in 31% of patients. We found no correlation between median blood pressure and EF before and after treatment (p = 0.351, r = 0.098). In addition, no correlation was found between the change in blood pressure and the change in EF between baseline (pre-carvedilol) and carvedilol treatment periods (p = 0.808, r = –0.025).
Comparison between echocardiographic parameters before and after carvedilol use in all subjects.
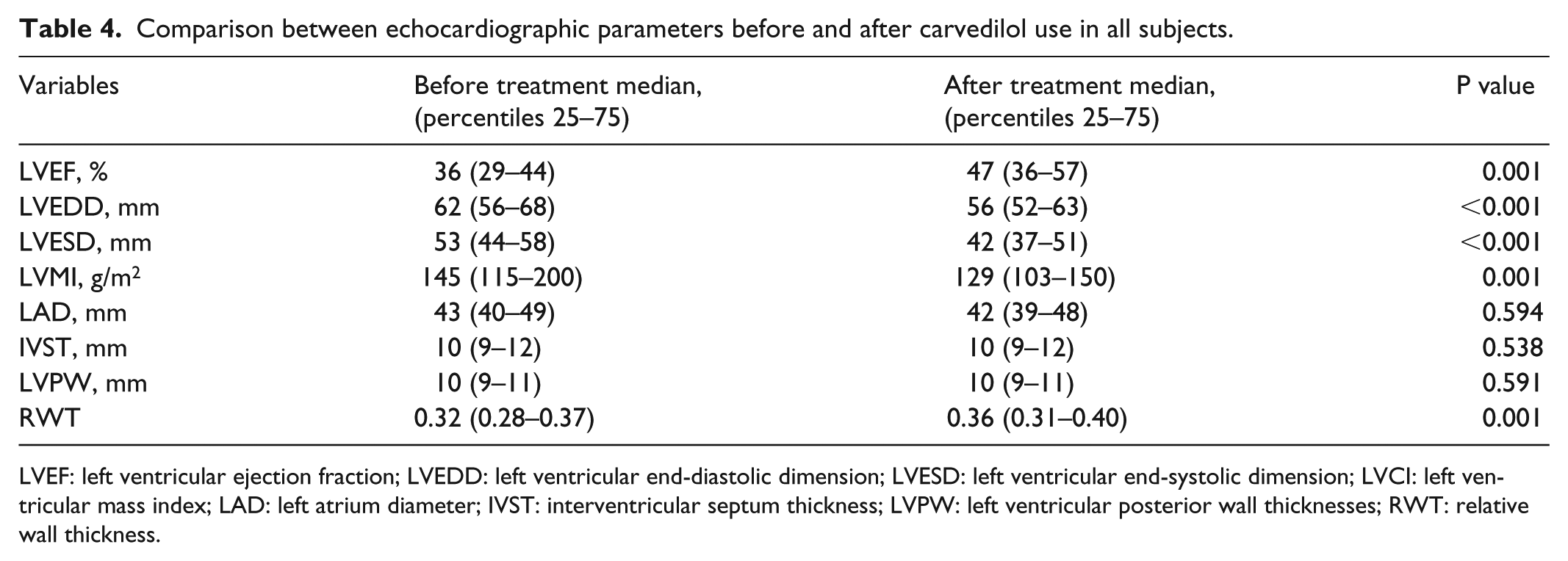
LVEF: left ventricular ejection fraction; LVEDD: left ventricular end-diastolic dimension; LVESD: left ventricular end-systolic dimension; LVCI: left ventricular mass index; LAD: left atrium diameter; IVST: interventricular septum thickness; LVPW: left ventricular posterior wall thicknesses; RWT: relative wall thickness.
Baseline demographic, hemodynamic, biochemical, and clinical characteristics were not different between the groups that improved versus those that did not improve LVEF during carvedilol treatment (Table 5). The group with improved LVEF had a non-significantly higher carvedilol dose than the group without improvement in LVEF. Moreover, the change in LVEF did not correlate with carvedilol dose (r = –0.158, p = 0.246). Nine patients died during follow-up, including six patients without improvement in EF and three patients with improvement in EF. Patients who did not have improved EF during carvedilol had nearly six-fold higher mortality (relative risk (RR): 5.7, 95% confidence interval (CI): 1.3–25, p = 0.022) during follow-up which ranged from 1 to 10 years (median: 6 years).
Comparison of the demographic, biochemical, anthropometric, and hemodynamic variables, before treatment, in the group that improved versus the one that did not improve the EF.
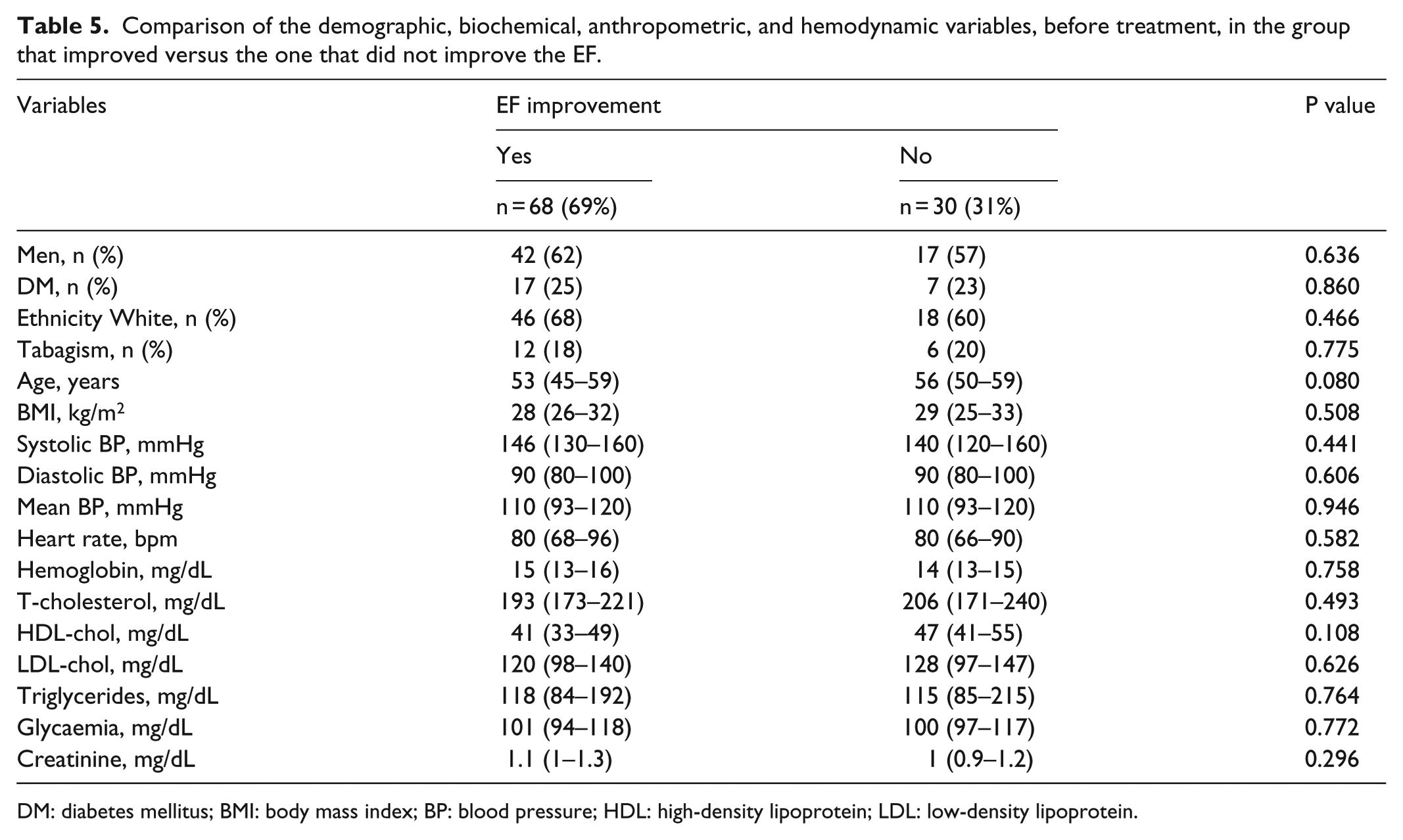
DM: diabetes mellitus; BMI: body mass index; BP: blood pressure; HDL: high-density lipoprotein; LDL: low-density lipoprotein.
Total mortality after a median 6 years follow-up of patients without improvement of LVEF was significantly higher than subjects with improvement in LVEF (41% vs 11%, log-rank and p = 0.009). The estimated 10-year survival was 89% for subjects with improved EF and only 59% for the group without improved EF. The group that improved EF had a lower baseline EF than the group that did increase during carvedilol treatment (Tables 6 and 7). The group with the best survival was the one with the highest increase in EF after treatment with carvedilol (EF median = 50.5%), and in the other group the median EF was 38%.
Comparison of medication frequency in the group that improved versus that which did not improve the EF.

EF: ejection fraction.
Comparison of the echocardiographic parameters between the group that improved and the one that did not improve the EF.

LVEF: left ventricular ejection fraction; LVEDD: left ventricular end-diastolic dimension; LVESD: left ventricular end-systolic dimension; LVCI: left ventricular mass index; LAD: left atrium diameter; IVST: interventricular septum thickness; LVPW: left ventricular posterior wall thicknesses; RWT: relative wall thickness.
Among the 98 patients included in this study, coronary angiography excluded CAD in all of the patients who submitted to this exam, that is, 36 patients (37%). The third (30%) who submitted exclusively to myocardial scintigraphy did not show any evidence of obstructive coronary disease.
Discussion
In this study, 98 consecutive HHD patients taking carvedilol were evaluated during a mean follow-up of 6 years (1–10 years). The main results include the improvement in EF, cardiac remodeling, and LV mass index. Five- and 10-year survival in subjects taking carvedilol was 98% and 83%, respectively. An improvement in EF was associated with better survival at 10 years. Mild increases in blood glucose and BMI were documented in HHD patients treated with carvedilol.
The median carvedilol dose during the treatment period was 50 mg daily, which is similar to previous reports in patients with HF.1,22 Adverse events related to carvedilol were captured in 8% of patients and included erectile dysfunction, bronchoconstriction, dizziness, lower extremity claudication, and worsening dyspnea. This frequency of adverse events was lower than that observed in MOCHA study. 22 However, in the MOCHA study, subjects had a different etiology for HF, and their mean EF was 23% compared to 36% in our study. Our data raise the possibility that carvedilol is beneficial for hypertensive patients AHA/ACC Stage B HF, that is, structural heart disease but without signs or symptoms of chronic heart HF. 23
In our study, the EF improved from a mean of 36% at baseline to 47% on carvedilol. EF improved more with carvedilol in patients with lower baseline values, which is similar to a previous study evaluating patients with EF <30%. 22 Benefit from antihypertensive drugs as diuretics and renin-angiotensin system blockers on improvement of structure and function of the heart in our sample is possible; however, all subjects were using optimized doses of diuretics and renin-angiotensin system blockers according to previous recommendations. 23 Renin-angiotensin system blockers lower blood pressure, reduce LVH, and improve cardiac remodeling.23,24
Spironolactone is recommended for HF and is related to LV reverse remodeling. 25 Half of the subjects in our study were taking spironolactone, and the dose was optimized before patients started using carvedilol.
The improvement of EF in our report is superior to that observed in the Carvedilol Post Infarction Survival Control in Left Ventricular Dysfunction substudy of HF after MI. 26 Our study excluded subjects with prior MI and/or documented ischemia. Medical records from 1184 patients were analyzed formerly, and 114 (10%) were excluded for previous MI or CAD diagnosed by cardiac catheterization or scintigraphy. The EF improved in up to 70% of patients. Sánchez et al. 12 evaluated EF in subjects with HHD and reported that EF rose in 60% of patients. However, in that study, only 52% of patients were taking beta-blockers, and blood pressure at the onset of the study was sub-optimally controlled.
In a previous study of subjects with various causes of HF and baseline EF ⩽30%, EF improved in 46% of them. 27 In our study, EF improved more in subjects with lower than higher baseline EFs. In the CAPRICORN study, 2 the authors suggested that EF improved more in subjects with lower EF because sympathetic nervous system activation is greater in those with poorer LV systolic function. We corroborate CAPRICORN study results as subjects in our study with lower rather than higher baseline EFs showed greater improvement in EF during carvedilol treatment.
The LV end-diastolic and end-systolic diameters declined significantly in subjects on carvedilol for a median of 6 years. In a previous study of 44 subjects with idiopathic dilated cardiomyopathy, Matsumura et al. 28 showed improvement in LV diameters after angiotensin-converting enzyme inhibitors/angiotensin II receptor blockers and beta-blockers use. In that report, nearly 80% of patients were taking carvedilol. In our study, 100% of patients were using diuretics, angiotensin-converting enzyme inhibitors/angiotensin II receptor blockers, beta-blockers, and all of them had HHD.
In our study, 5- and 10-year survival rates were 98% and 83%, respectively. Ten-year survival was greater among patients with improvement in LVEF than in those without improvement (89% vs 59%). De Carvalho Frimm et al. 18 reported a 73% survival in 90 patients with HHD after 4 years of follow-up. In that study, subjects were not taking beta-blockers. In our study, an association was found between the improvement in LVEF and reduction in mortality. Subjects without an improvement in LVEF had six-fold higher mortality than subjects that had an improvement in LVEF on carvedilol. Choi et al. 29 observed lower mortality among subjects with various etiologies of HF who had LV reverse remodeling with treatment. In the study by Choi et al., only 69.2% of patients were taking beta-blockers, with 91% on carvedilol and 9% on metoprolol. In our study, all patients who had hypertensive cardiomyopathy, were taking diuretics, RAS blockers, and were evaluated after optimization of carvedilol dose. The US carvedilol study 1 showed an association between systolic dysfunction and poor prognosis in HF patients. The US carvedilol trial was one of the first studies to test the safety of carvedilol in 131 patients with HF of different etiologies.
Previous studies reported a reduction of morbidity and mortality in HF subjects with reduced ejection fraction (HFrEF) when they were treated with renin-angiotensin system blockers, β-adrenergic blockers, aldosterone blockers, and cardiac resynchronization. 30 There is growing interest in morbidity and mortality among patients with HF and preserved ejection fraction (HFpEF) patients. No studies have clearly documented improved outcomes with pharmacologic intervention in the HFpEF population. 31 In the I-preserve trial, 32 an angiotensin II blocker failed to prevent cardiovascular death in HFpEF patients. We evaluated HFrEF and HFpEF patients and cardiovascular mortality was higher in HFpEF patients. In our study, we showed improvement in LVEF, reverse remodeling of the LV, and improved survival in subjects with HHD taking antihypertensive medications and carvedilol; and, Stage B HFrEF patients had lower mortality than Stage B HFpEF patients. Of note, the separation in survival between Stage B HFrEF and HFpEF patients did not begin until the fifth year. Thus, studies with 5 years or less follow-up in patients with HDD may not distinguish between these two groups. To our knowledge, this is the first study reporting improved outcomes in HHD using carvedilol.
The absence of a relationship between blood pressure and improved LVEF suggests that other actions of carvedilol, for example, α,β-blockade played a role. 17 Heart rate decreased 10 beats after carvedilol use, as reported previously. 25 However, the change in heart rate was not related to the change in EF.
We observed a mild increase of blood glucose and BMI after treatment with carvedilol. Previous data showed a neutral effect of carvedilol in blood glucose. 33 In the GEMINI study, 33 body weight did not increase in diabetic hypertensive patients using carvedilol and increased slightly in subjects using metoprolol. In our study, there was a tendency of BMI to increase during a median follow-up period of 6 years, which is a longer follow-up period than GEMINI. Total cholesterol and HDL-fraction levels did not differ in our study after carvedilol use, which is similar to reports from the GEMINI study. 33 On the other hand, LDL-cholesterol tended to fall and may have reflected a r neutral or benefic effect of carvedilol on lipoprotein lipase. 34
CAD was not found in all 66 patients who underwent invasive and noninvasive evaluation. CAD and hypertension impact LV function and risk for HF, which makes it difficult to separate the independent effects in patients with both hypertension and CAD. 35
Study limitations include the absence of a control group. However, all patients had some degree of systolic dysfunction on the echocardiogram. As the efficacy of beta-blockers such as metoprolol, bisoprolol, and carvedilol has been demonstrated in patients with systolic dysfunction due to other etiologies, especially ischemic, the ethics of withholding beta-blockers from patients with systolic dysfunction due to HHD are questionable. In our pre-post study design, each patient served as their own control. Potential confounding factors included baseline blood pressure and EF as well as the carvedilol dose during the treatment period. However, baseline blood pressure was similar in the group that improved and the group that did not improve EF on carvedilol (see Table 5). Moreover, no correlations were found between baseline or treatment blood pressure or change of blood pressure between the baseline and carvedilol treatment period and changes in LVEF. The dose of carvedilol was also similar in both groups. Although one criterion for entering the study was optimized treatment of hypertension before carvedilol, some patients has sub-optimal blood pressure since baseline BP averaged 140/90 mmHg. The baseline antihypertensive therapy, which included diuretics and renin-angiotensin system blockers, may have contributed to the improvements in LV structure and function. However, all subjects were using optimized doses of diuretics and renin-angiotensin system blockers at baseline according to previous recommendations. Another limitation of our study is that we could not identify factors, other than differences in baseline EF, which were related to higher survival of the group that had an improvement in EF and better survival. Of note, our study was designed to delineate the mechanisms by which carvedilol improved LVEF and survival in patients with HHD.
Our findings suggest that addition of carvedilol to the treatment of hypertensive patients with HHD may confer additional outcome benefits. The beneficial effects of carvedilol may be mediated through various mechanisms: (a) for beta 1 and 2 blockade in the heart, through the renal beta 1 effect, and consequent inhibition of renin production; which possibly potentiates the action of inhibitors of the renin-angiotensin system; (b) by alpha 1 blockade, which improves the afterload. Thus, carvedilol may contribute significantly to LV reverse remodeling through mechanical (post-load reduction) and neurohormonal (inhibiting renin production and adverse cardiac effects of catecholamines) effects.
Although subjects had relatively well-controlled hypertension at baseline, blood pressure control improved after carvedilol initiation. Median baseline BP was 140/90 mmHg and fell to 130/80 mmHg during carvedilol treatment. As noted previously, no correlation was found between baseline or treatment BP or changes in BP with treatment and improvement of LVEF. Moreover, patients were receiving effective doses of diuretics and renin-angiotensin system blockers at baseline, which were continued during carvedilol treatment.
In conclusion, our study results indicate that carvedilol, when added to antihypertensive treatment in patients with HHD, reversed parameters of remodeling of the left ventricle and increased EF, especially in those with baseline EF <34%. Carvedilol also improved survival in HHD. The survival was better in patients who began the study with stage B HFrEF who had improved LVEF than in patients with stage B HFpEF who did not improve LVEF during carvedilol therapy.
Footnotes
Acknowledgements
The authors of this project acknowledge the technical and scientific support of the institution Universidade Nove de Julho—UNINOVE.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from Ethic Committee from Universidade Nove de Julho —UNINOVE (number 665293).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from all subjects before the study.
