Abstract
Objective:
The geriatric population suffers from a predisposition to cardiac events due to physiologic changes commonly associated with aging. The majority of the trauma population seen at our facility is within the geriatric population (greater than 65 years old). Therefore, this study was aimed to determine which of those preexisting factors were associated with an increased risk for developing cardiac event. By assessing those risks, we hoped to determine a timeline for the highest risk of cardiac event occurrence, in order to identify a safe period of when cardiac monitoring was indicated.
Methods:
A retrospective study performed over 6 months reviewing geriatric trauma patients with hip, pelvis, or femur fractures, n = 125. A list of predetermined risk factors including comorbidities, pathologies, laboratory values, electrocardiogram findings, and surgery was crossed with the patient’s records in order to identify factors for increased risk of cardiac event. Once patients who had documented cardiac events were identified, a temporal pattern of cardiac event occurrence was analyzed in order to determine a period when noninvasive cardiac monitoring should remain in place.
Results:
In 125 patients, 40 cardiac events occurred in 30 patients. The analyzed variables with statistically significant associations for having a cardiac event were comorbidities (p = 0.019), elevated body mass index (p = 0.001), abnormal initial phosphorus (p = 0.002), and an electrocardiogram finding of other than normal sinus rhythm (p = 0.020). Of the identified cardiac events, we found that by hospital day 3 68% of cardiac event had occurred, with 85% by hospital day 4, 95% by day 5, and 100% within the first 7 days of admission.
Conclusion:
Patient history of cardiac comorbidities, elevated body mass index, abnormal phosphorus, and abnormal electrocardiogram findings were found to be significant risk factors for cardiac event development in geriatric trauma. All recorded events in our study occurred within 7 days of the initial trauma.
Introduction
It is well known and has been reported by several resources that the elderly population, in comparison to their nongeriatric counterparts, has an appreciable decrease in the cardiac reserve needed to mount a compensatory response to injury.1–3 This described reduction can be attributed to multiple factors that specifically target the geriatric population, including replacement of myocytes with fatty and fibrous tissues, impaired ventricular performance, decreased cardiac output, and decreased left ventricular ejection fraction. 1 Recognizing the impact and significance of this information on our trauma institution’s main demographic, this retrospective study was conducted as a quality improvement project with the aim to identify comorbidities and risk factors that are more likely to increase the threat of cardiac injury and the timeline of when it occurs.
Upon review of our institution’s trauma database, it was noted that a strikingly significant number of trauma admissions were patients who met the criteria to be included in our Geriatric Trauma Institute (GTI), defined as age 65 or older. Furthermore, a significant amount of our GTI patients were those who were admitted after sustaining hip, pelvic, or femur fractures. With further analysis of this patient population and their medical histories, it was obviously noted that a substantial amount of these patients also had a history of cardiac pathologies or harbored major risk factors for cardiac abnormalities. The majority of our geriatric trauma patients are blindly placed on telemetry monitoring at hospital admission which remains throughout their entire hospital course without evaluation of risks or duration. Due to the vast amount of patients on telemetry monitoring, resources at our institution are strained and are often unable to provide coverage for all patients on whom telemetry is ordered. This issue of resource management, specifically monitor allocation, is a factor that plagues many institutions, thus the findings presented within can help additional trauma centers to establish appropriate resource distribution and cardiac monitoring protocols.
This review was conducted in order to identify risk factors and timing of cardiac events (CEs) in the immediate post-trauma geriatric population. For the purpose of this study, we defined CE as any change from a previously established cardiac rhythm baseline or any change from a previously recorded normal sinus rhythm. The data presented within reflect only the CE types and amounts witnessed through the course of the study and did not evaluate for other types of CEs that were not observed in our patient population. By determining the impactful risks and pertinent timelines, it was our intention that when indicated, noninvasive cardiac telemetry could commence at hospital admission. With the immediate implementation of telemetry, we theorized that any cardiac rhythm changes could then be rapidly identified and were hopeful that any necessary interventions could then be provided. Furthermore, in our determined study population, we retrospectively assessed the time from admission to CE occurrence. This was done in order to identify a significant time interval in which changes from cardiac baseline were recognized on telemetry. It was our intention to not only identify a safe window in which telemetry monitoring was mandated but also determine a point after this distinguished duration in which monitoring could reliably be discontinued.
Objectives
The study was performed as a retrospective quality improvement project in our geriatric trauma population. This information was analyzed in order to identify which factors in the post-traumatic period were associated with an increased risk of CEs. As a result of population size and risk occurrence, our factors were assessed based on the amount of associated risks a patient harbored per category rather than individual risk factor quality (Figure 1). We hypothesized that patients found to have significant abnormalities in any of these groups would be at an increased risk for sustaining a CE and would require noninvasive cardiac monitoring. Furthermore, we theorized that the more factors a patient had, the more at risk for CE they would be. Finally, within the study population, we attempted to determine the temporal distribution of CEs in order to determine an appropriate interval for cardiac monitoring.
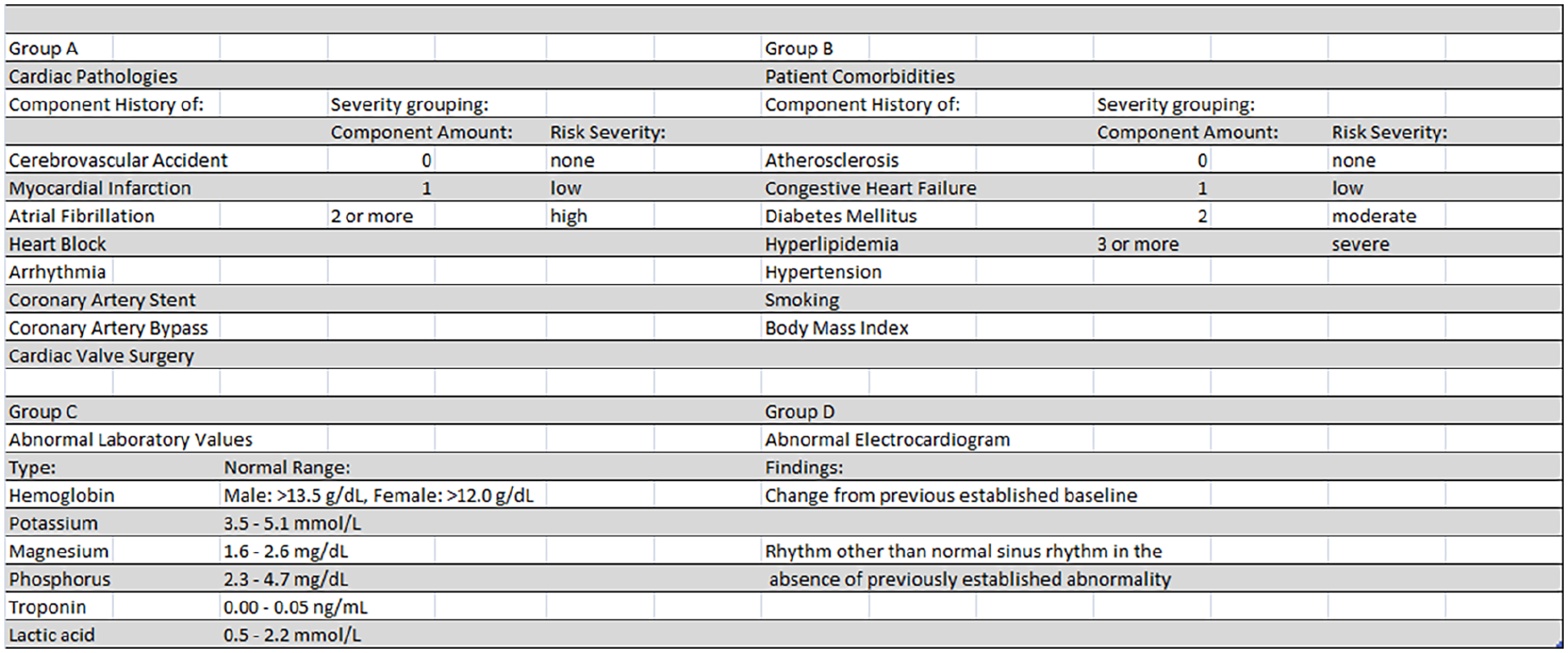
Assessed categories, components, and grouping for cardiac event risk factor analysis.
Methods
The study was a retrospective review performed at a Level 1 Trauma Center over a 6-month period, from November 2016 through April 2017, which identified 125 applicable patients. The study period was determined by the availability of electronic medical record (EMR) data which became hospital wide beginning at that time. The authors received no funding or specific grant from any funding agency in the public, commercial, or not-for-profit sectors for the production of this study or the preparation of this manuscript. The study was performed after approval was obtained from our hospital’s Institutional Review Board.
The inclusion criteria comprised two factors. First, the patients must be part of the geriatric population, established as age 65 or older, and second, their injury complex must contain hip, pelvic, or femur fractures. The “hip” fracture locations were defined as: femoral head, subcapital neck, transcervical neck, intertrochanter, subtrochanter, greater trochanter, or lesser trochanter. The “pelvic” fractures included ramus, tubercle, iliac, and ischial. Finally, per our study, those recognized as “femur” fractures were those considered to be confined within any part of the femoral shaft or the distal femur.
The analyzed geriatric demographic has already been described as having an increased level of cardiac stress; however, in addition to the inclusion criteria, preexisting conditions were reviewed to determine factors that could potentially further increase the risk. For the purpose of this study, a CE was defined as any change from a previously established cardiac baseline throughout the patient stay. Our definition of “cardiac baseline” specifically focused on documentation of prior rhythms, cardiac pathologies, or other abnormalities found in the patient’s medical record. If the patient did not have a prior medical record or was unknown to our hospital system, and a previously established baseline could not be identified or obtained from the patient at the time of initial assessment, any rhythm abnormality other than normal sinus rhythm was considered a CE until proven otherwise by subsequently obtained information. As telemetry was the main form of noninvasive cardiac monitoring employed by this study, any abnormality or change from baseline identified on telemetry was considered a CE and further evaluation and treatment was implemented at that time. Additionally, as this study was performed in a retrospective manner, a selected type of noninvasive monitoring and a mandated time period for monitoring was unable to be employed. Instead, as our hospital system routinely uses electrocardiogram (EKG) telemetry, this was the only form of noninvasive cardiac monitoring that could be evaluated. As well as, the reviewed data and capture period were based on the clinical scenario at the time of the patient’s admission and did not have a set clinical duration for the study.
The evaluated conditions considered as possible “risk factors” included cardiac pathologies (history of cerebrovascular accident, myocardial infarction, atrial fibrillation (AF), heart block, arrhythmia, history of coronary artery stenting, history of coronary artery bypass grafting, history of cardiac valve surgery), cardiac comorbidities (atherosclerosis, congestive heart failure, diabetes mellitus, hyperlipidemia, hypertension, smoking, body mass index (BMI)), laboratory values, EKG abnormalities, and need for surgery. Given the patient population, the severity of each individual diagnosis as a risk factor compared to another was unable to be determined; therefore, the pathologies and comorbidities were grouped into separate categories. These categories were then grouped by severity based on the amount of risk factors, rather than each factor individually. The cardiac pathologies were grouped such that having zero pathology classified as “none,” while one pathology was recorded as “low,” and a history of two or more pathologies was considered “high.” Cardiac comorbidities were similarly grouped. Whereas, having zero comorbidities was again classified as “none,” one comorbidity was recognized as “low,” two comorbidities was considered “moderate,” and finally three or more comorbidities was classified as “severe” (Figure 1).
Patient suspected pertinent laboratory values were also assessed, including hemoglobin, potassium, magnesium, phosphorus, troponins, and lactic acid. This study was performed after the establishment of the EMR at our facility, and therefore, the normal laboratory range was determined by the current values computed by our EMR. Any value that did not lie within this established range was therefore considered an abnormality in our data calculations. The range of values is included in Figure 1 for reference. Furthermore, the occurrence of CE in the patient population requiring surgery was compared to those patients who did not undergo an operative procedure. The last analyzed potential cardiac factor was the patient admission EKG. Per the frame of this study, the EKG was considered either normal or abnormal. The definition of “abnormal” in this study was defined as anything differing from a previously reported patient baseline or from normal sinus rhythm. This determination was based on the patient’s admitting EKG. If the patient had a previously underlying abnormal rhythm that was present and unchanged on the admitting EKG, this was still classified as normal.
Additionally, this study aimed to analyze the occurrence of CEs throughout patient admission. By doing this, it was our intent to identify a safe duration where cardiac monitoring would identify the majority of abnormalities in rhythm and a time frame of safe discontinuation. Therefore, we retrospectively reviewed the study population, focusing on the patients who experienced a CE that was recognized by the noninvasive monitor. The first documented time of the rhythm abnormality was then recorded for each patient. The timing for each change in rhythm was then compiled and underwent statistical analysis of the temporal pattern to determine CE risk based on time.
Statistical analyses
Chi-square was utilized to investigate influential factors resulting in a CE. Where appropriate a layered chi-square was used to control for a third variable, such as operative intervention. When greater than 20% of the cells had an expected frequency of less than five, Fisher’s exact test (FET) was employed. Overall, alpha was set at 0.05. An a priori chi-square (df = 1) power analysis given a 0.2 effect size, 0.05 alpha, and desired power of 0.85 yielded a required sample size of 225. Given the potential for multiple testing, lowering alpha to a Bonferroni-corrected value of 0.005 increased the required sample size to 370. IBM SPSS version 24 was the software used to perform the statistical analyses. A longitudinal histographic analysis was applied to time from admission to first recorded CE. The intent was to identify a guideline for duration of cardiac monitoring for situations of limited monitoring resources during which the majority of rhythm abnormalities will be captured.
Results
A retrospective review of geriatric trauma patients at a single center over a 6-month period was able to identify 125 total patients meeting inclusion criteria, n = 125. Of the study population, 67% (84/125) of patients sustained hip fractures, 18% (22/125) suffered femur fractures, and 17% (21/125) patients had pelvic fractures. One patient sustained both hip and pelvic fractures, while another patient sustained both hip and femur fractures. Average age, injury severity score, and Glasgow Coma Score for the study population were 83, 9, and 14, respectively. The majority of patients in this study were female, 76%.
Patients who had at least one prior cardiac pathology experienced 63% (19/30) of CEs. Those with no prior cardiac pathology had 37% (11/30) of the observed CEs, while the low group experienced 27% of CEs, and the high group observed 37%. Furthermore, patients with at least one comorbidity suffered 97% (29/30) of the CEs. The percentage of CEs that occurred per level followed an assumed pattern, in that an increasing amount of cardiac comorbidities would increase the risk of post-traumatic CEs. (None 3% (1/30), low 20% (6/30), moderate 30% (9/30), or severe 47% (14/30)). Assessment of the laboratory values was hindered due to incomplete data collection. For example, only eight patients who sustained CEs had admitting troponins collected. Due to the small troponin data size, no significant statistical analysis could be performed. Chi-square analyses (normal/abnormal vs CE yes/no) of hemoglobin, lactic acid, potassium, and magnesium did not yield statistically significant results. Phosphorus (P) level was the only identified statistically significant laboratory value in relation to CE occurrence, p = 0.002. Finally, of the 106 patients having had an EKG performed, 27% (29/106) were found to have an abnormal EKG and sustain a CE throughout their admission. This included 97% of the patients in which a CE occurred, leaving only one patient with a CE, and 3% (1/30) without data collected via EKG.
CE occurrence was analyzed in the population, for which 40 CEs were identified in 30 patients. The types of CEs seen in our study included ST elevation myocardial infarction (STEMI), non-ST elevation myocardial infarction (NSTEMI), AF, supraventricular tachycardia (SVT), ventricular tachycardia, multifocal atrial tachycardia, heart block, atrial flutter, asystole, heart failure, cardiac tamponade, atrial thrombus, and cardiogenic shock. The CE types that we witnessed, and their amount of occurrence, were recorded in Figure 2. Given the limited total population size and event size, risk factors pertinent for individual event types were unable to be identified.

Observed cardiac events by type and number.
From our collected data, we employed either a chi-square test or FET, when greater than 20% of the cells had an expected frequency of less than 5, in order to find the variables most likely to be a predictor of having a CE. The variables with a statistically significant association with having a CE were preexisting comorbidities (FET, p = 0.019, with an approximate strength of association φ = 0.236, p = 0.008); categorized BMI (FET, p = 0.001, with an approximate strength of association φ = 0.317, p < 0.0005); abnormal initial phosphorus (FET, p = 0.002, with an approximate strength of association φ = 0.329, p < 0.0005); and abnormal EKG (χ2 (1) = 5.393, p = 0.020, with an association magnitude, Cramer’s V = 0.226, p = 0.020).
Analysis of CEs in post-trauma patients who underwent operative intervention was compared to the data for CEs in patients who sustained trauma alone without requiring surgery. Surgery was not statistically significantly associated with having a CE (χ2(1) = 0.876, p = 0.349, consequently, no association was found, φ = 0.084, p = 0.349). Of our study population, 81% (101/125) underwent a form of operative repair, while 19% (24/125) did not require surgical intervention. Of the 81% that required surgery, 35 CEs occurred in 26 patients (26% of patients), while the non-operative cohort experienced 4 CEs, occurring in 3 of 24 patients (13%). Figure 3 was compiled to display the rates of CE occurrence per category.

Cardiac event rates per risk categories.
Finally, by recognizing CEs on telemetry, we were able to identify a temporal distribution of occurrence. Of the 40 CEs recorded, by hospital day 3, 68% occurred, by day 4, 85% had taken place, through day 5, 95% of events occurred, and 100% of events occurred by post-trauma day 7 (Figure 4). In the surgical population, we found that 97% of CEs occurred within 4 days of surgery and 100% occurred by post-operative day 5.5 (Figure 5).
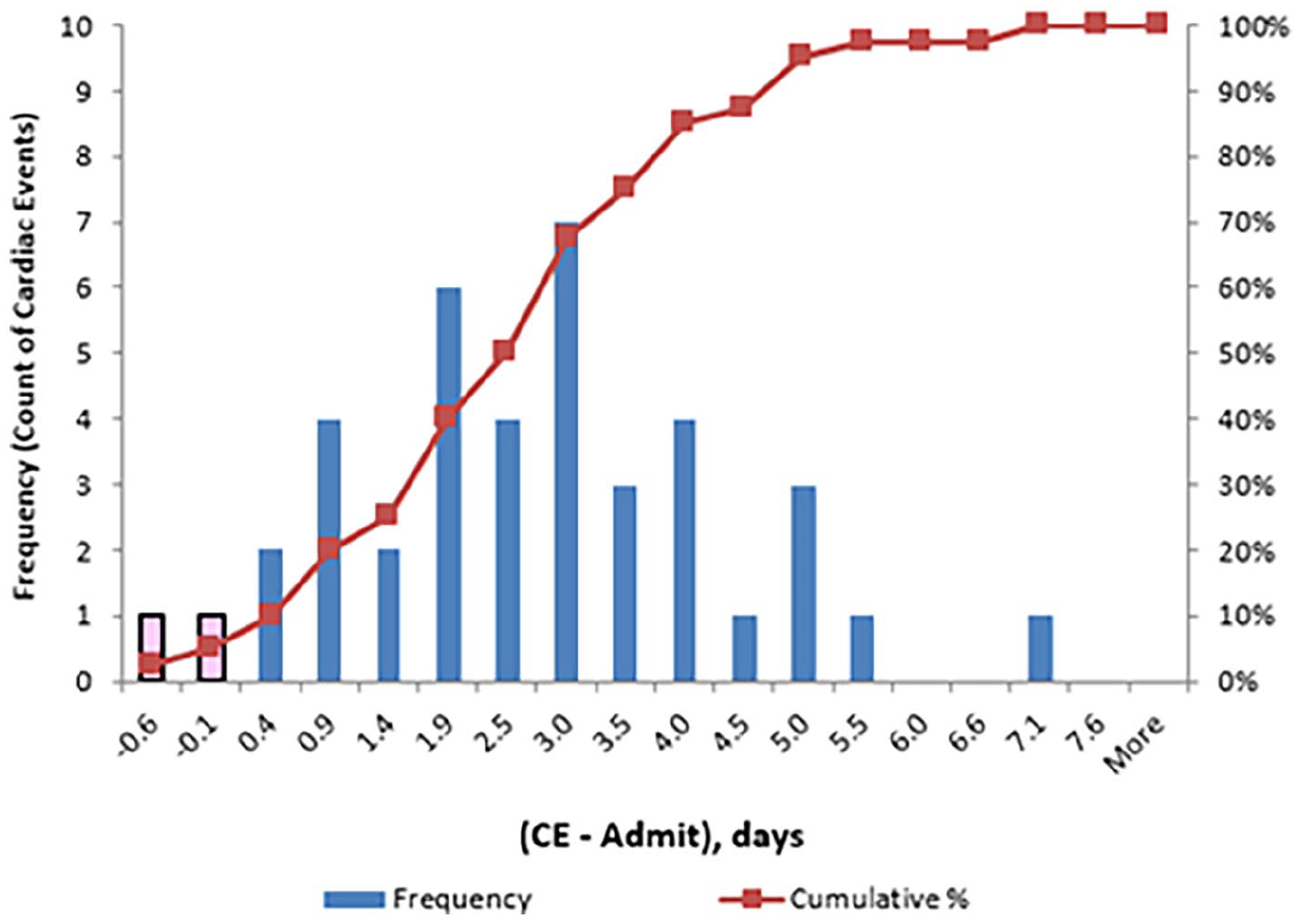
Time of cardiac event in relation to hospital admission.

Time to cardiac event following surgical procedure.
Discussion
This study was a retrospective review of 125 geriatric trauma patients who sustained hip, pelvis, or femur fractures that were evaluated at a community Level 1 Trauma Center. The purpose of this study was to determine which patients, in an already predisposed population, were at an increased risk for post-traumatic or post-operative CEs. By evaluating patient EKG findings, comorbidities, previous cardiac histories, and laboratory value abnormalities, we were able to determine important correlating factors for predicting patients who were more likely to experience a change from cardiac baseline. These factors included a history of multiple cardiac comorbidities, elevated BMI, abnormal phosphorus, and abnormal EKG rhythm. Although our attempt was to identify significant pathologies and risk factors that would be associated with increased occurrence of CEs, we could not establish a hierarchal ranking among the risk factors, and to the best of our knowledge, this has not been established in the literature. Therefore, grouping of the pathologies and comorbidities was performed and the separate groups were ranked based on severity then analyzed.
Finally, by review of the data presented, we were able to determine time periods for when the majority of CEs occurred in our population. For patients who underwent surgery, 100% of CEs occurred within 5.5 days of surgery, and for the combined surgical and nonsurgical population, 100% of events occurred within 7 days of hospital admission (Figures 4 and 5). It does need to be recognized that this study was performed from a retrospective scope for which a mandated monitoring period could not be established. The duration of monitoring in this study was based on the patient and clinical factors of that admission. However, despite the lack of designated duration, the data on CEs in our population were reviewed throughout the entire length of stay (LOS); 100% of events were identified within the first seven post-traumatic days, regardless of total hospital LOS. With this timeline, we now are able to effectively limit our monitor use to a safe 7-day window, allowing us to achieve our goal of properly allocating hospital resources based on data and identified risk factors from this study rather than physician discretion alone.
The most commonly occurring CE seen in our study was AF. AF was the cause of 15 of the 40 seen CEs (37.5%), occurring in 12% of the study population. It has previously been reported that the incidence of AF is related to increasing age. 4 There are a reported 2.2 million people in the United States of America with AF. 4 The prevalence of AF in those older than 40 years of age is 2.3%, whereas in those over 65 years of age it is 5.9%. 4 The geriatric population (over 65 years old) makes up approximately 70% of the population with AF. 4 Although our population is small, the occurrence of AF in our population is higher than the norm. This increase could be due to the patient’s trauma as a small study by Marco et al. 5 found an increase in new-onset AF after geriatric trauma, especially in male patients.
Of the significant results, the discussion of the striking difference between normal and abnormal P must be entertained. Historically, there have been multiple reports linking hyperphosphatemia to cardiovascular disease and myocardial infarction in patients with kidney disease. Additionally, recent data has also shown an increase in the risk for myocardial infarction and new-onset heart failure in patients without kidney disease and glomerular filtration rates above 60. 6 Alternatively, there has been some prior non-trauma data that has shown low phosphorus levels can be cardioprotective. However, per our data, this does not appear applicable as the majority of CE in abnormal phosphorus levels occurred in patients with hypophosphatemia. 7 Because of this discrepancy and the clinically significant amount of CEs that occurred in patients with abnormal P levels, we feel that the p value specifically must be further reviewed in the geriatric trauma population to identify its correlation with CEs.
To the best of our knowledge, in conjunction with extensive PubMed literature search, this is the first reported study with the goal of evaluating EKG telemetry rhythm changes for recognition of CEs in the geriatric trauma population. We formulated this study based on the main benefits of cardiac monitoring in the general population, while specifically applying that to our geriatric trauma patient population. According to the American Heart Association (AHA), there are four main rationales for arrhythmia monitoring. 8 These factors include the immediate recognition of sudden cardiac arrest to improve time to defibrillation, recognizing deteriorating conditions that may lead to life-threatening sustained arrhythmias and prompting treatment, facilitating management of non-life-threatening arrhythmia, and finally diagnosing arrhythmia or the cause of symptoms with subsequent management. 8 These four principles were incorporated into the stem of our hypothesis. We hoped to use telemetry in an already predisposed geriatric trauma population as a way to rapidly assess changes from the patient baseline and to quickly treat the patient if needed.
Additionally, AHA also comments on the indications and establishes duration recommendations for cardiac monitoring. They designate multiple different categories based on the patient’s history, acute status, devices, and other risk factors. Although several categories are discussed, there is no discrete mention of monitoring indications or duration recommendations for the geriatric trauma population. 8 Similarly, the Eastern Association for the Surgery of Trauma (EAST) has no discrete guideline for cardiac monitoring in the general geriatric trauma population. 9 EAST does, however, comment on certain trauma situations in which noninvasive monitoring is indicated, with the focus mainly on blunt cardiac injury. For patients who sustained blunt cardiac injury, EAST recommends continuous cardiac monitoring if a new or unexplained rhythm is identified on admission EKG. 9 Furthermore, if that abnormal rhythm is sustained, they recommended progressing to echocardiogram for additional evaluation. 9 However, this application to blunt cardiac injury was not applied generally to the geriatric trauma population and the authors of this article were unable to identify any other source with similar broad-based geriatric trauma recommendations. Similar to the recommendations by EAST for blunt cardiac injury, the pertinent data that we found via our cardiac rhythm analysis may likely also require further evaluation via echocardiogram for management planning. 9
As the question we proposed had no other identifiable answer or previously published comparison study, we aimed to determine one. We identified multiple cardiac rhythm abnormalities on EKG telemetry alone (40) in a small population size and also identified significant contributing factors including comorbidities, BMI, P, and EKG abnormalities. As the duration recommendations produced by AHA did not reflect the trauma population, we found in our study that over half of the recognized CEs occurred within just 3.5 days following the initial trauma and that 100% of our recorded CEs were seen within the first week (Figure 4).
The intent of this study was to identify risk factors and a timeline of CEs that would be recognized on EKG telemetry to prompt necessary treatment. Further research must be performed in order to identify the implications of the results of said telemetry monitoring in the geriatric trauma population. Multiple other studies have been performed weighing the benefit of cardiac monitoring in non-trauma populations, for which mixed results have been reported. Early identification of life-threatening cardiac arrhythmias is one of the main benefits of telemetry and why it is indicated per AHA. Per the study produced by Cleverly et al., 10 multiple benefits of telemetry were seen, including higher survival rates immediately following cardiac arrest, higher survival rates to hospital discharge, and greater benefit of telemetry recognition in events that occurred over night or early in the morning. An additional study analyzed patient events per their AHA classifications. Their analysis showed that 43% of patients in which monitoring was indicated sustained a CE, and a subsequent change in management occurred in 25% of those patients. 11 However, in contrast, there have been additional studies questioning the benefit of telemetry in patients with respiratory illness. 12 One study found that in patients with respiratory illness the use of telemetry showed no difference in 30-day readmission rates, hospital mortality, or 90-day mortality rates when compared to non-monitored patients. 12 Additionally, the report also found that LOS was actually increased in patients on telemetry compared to those patients who were not monitored. 12 Although these referenced studies provide valuable information, no studies could be found that assessed telemetry specifically in geriatric trauma, and as such we feel that further research in this demographic is warranted.
As the authors of this article, we felt that this study was important to produce due to the lack of both previously published data and proper resource allocation. However, we do acknowledge several study limitations, namely, performance at a single center, insufficient sample size, incomplete data (e.g. admission EKGs and substantial amount of missing lab values), and selection bias, as injuries were confined to the hip, pelvis, or femur. Holding the desired 0.85 power constant, sample size insufficiency increased the a priori 0.2 effect size by 35% to 0.269 for 0.05 alpha and by 73% to 0.345 for the Bonferroni-adjusted alpha of 0.005; or, in terms of statistical power (at 0.2 effect size), a reduction from a power of 0.85 to 0.608 and 0.283, respectively. The sample size prevented performance of meaningful inferential multivariable comparative analyses of individual comorbidities and previous cardiac conditions against each other for severity per specific pathology. Additionally, analyses of and involving laboratory values were further restricted as labs of interest were inconsistently collected, which further decreased the useful sample size for those analyses. A three- to fivefold sample size increase would accommodate more powerful and robust statistical methods and analysis of the pathologies separately. This would have the potential to determine increased severity for certain conditions in comparison to others and provide evidence for or against certain laboratory abnormalities. Finally, this larger sample size would allow investigation of additional potential factors not accounted for in the study that may influence CE occurrence, including but not limited to: underlying cardiac histories, comorbidities, lab values, admitting vitals, medications, and specific operative procedures.
Conclusion
The main hypothesis of this study was to identify underlying factors that would further increase the risk of an already predisposed population to CEs. We were able to establish a secondary goal of this study to create a protocol in which hospital resources like telemetry monitoring could be properly allocated and used for a proper duration. Through the significant risk factors identified by this study (cardiac comorbidities, elevated BMI, abnormal phosphorus, and abnormal EKGs), patients harboring these factors are started on cardiac monitoring immediately. As we are attempting to limit the superfluous use of hospital resource, those in our population who do not meet our criteria do not undergo monitoring. The duration then lasts the full 7 days, or until patient discharge, to capture our goal of 100% of events. With our new recommendations, we will review the accuracy of our data compared to our data prior to implementation of the guidelines. As there appears to be no other similar recommendations from other institutions or our governing bodies, it is our hope to identify further significant factors and devise potential recommendations that are applicable to other institutions in an effort to help guide their geriatric trauma management and resource allocation.
Footnotes
Author contributions
K.R.C., D.S.U., B.D.D., and T.J.S. contributed to conception and design, acquisition of data, and interpretation of data. K.R.C., T.J.S., B.D.D., and S.L.M. contributed to manuscript writing and drafting. K.R.C., D.S.U., T.J.S., B.D.D., and S.L.M. revised the manuscript critically for important intellectual content. K.R.C., D.S.U., B.D.D., T.J.S., and S.L.M. approved the final version of the manuscript to be published.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Disclosure
We would like to report that a condensed poster presentation of this study was displayed at The American College of Surgeons Trauma Quality Improvement Program in Chicago, IL, on 12 November 2017.
Ethical approval
Ethical approval for this study was obtained from Duke LifePoint Conemaugh Memorial Medical Center Institutional Review Board, 17-21.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Informed consent was not sought for the present study because this study was performed in a retrospective manner for which patients had already been treated and discharged from the hospital prior to review of information. No patient specific information is included in the study. Only authors approved by IRB 17-21 were able to access patient data to produce information pertinent to this study and then patient-specific information was immediately deleted and none was saved.
