Abstract
Background:
Treatment of patients with traumatic axonotmesis presents challenges. Processed human umbilical cord membrane has been recently developed with improved handling and resorption time compared to other amniotic membrane wraps, and may be beneficial in nerve reconstruction. This study evaluates postoperative outcomes after traumatic peripheral nerve injury after placement of commercially available processed human umbilical cord membrane.
Methods:
We performed a prospective, single-center pilot study of patients undergoing multi-level surgical reconstruction for exposed, non-transected peripheral nerve. Functional outcomes including pain, range of motion, pinch and grip strength, and the QuickDASH and SF-36 patient-reported outcome measures were recorded, when possible, at the 1-week and 3, 6, and 9 months postop visit. One-tailed paired t-tests were performed to evaluate outcome improvement at final follow-up.
Results:
Twenty patients had processed human umbilical cord membrane placement without surgical complications. Mean follow-up was 7.5 months (range: 3–10 months) and mean age was 39 years (range: 15–65). Twelve (67%) patients were male, and the majority of placement sites were in the upper extremity (85%). Mean preoperative visual analog scale pain score was significantly reduced at most recent follow-up, as were QuickDASH scores. All patients had improved functional outcomes at the 9-month follow-up, and SF-36 outcomes at 9 months showed improvement across all dimensions.
Conclusion:
This study indicates that processed human umbilical cord membrane may be a useful adjunct in nerve surgery with noted improvements in postoperative function, pain, and patient-reported outcome measures. Future studies are needed to assess long-term outcomes after traumatic nerve injury treated with processed human umbilical cord membrane.
Background
Acute peripheral nerve injuries occur in 3%–10% of all traumatic extremity injuries, with the majority occurring in the upper extremity. 1 Often, these injuries to peripheral nerves are associated with multiple other tissue types, including bone, tendon, and vascular damage, and are associated with suboptimal outcomes.1,2 Nerve defects typically fall into one of two categories: neurotmesis, in which the nerve and the nerve sheath are disrupted, and axonotmesis, in which the axons and their myelin sheath are damaged but the endoneurium, perineurium, and epineurium remain intact. 3 Neurotmesis, which is the most common type of nerve defect in multilevel traumatic injuries, is relatively straightforward to treat with exploration and repair. However, the treatment of patients with axonotmesis presents a greater challenge to the surgeon as damage to the nerve is not always visible. 3
In axonotmetic injuries, as in neurotmesis, the nerve will undergo Wallerian degeneration and subsequent axonal growth. While Wallerian degeneration is necessary for nerve regeneration, 4 it is associated with inflammatory reaction involving macrophages. 5 This inflammatory process, along with the disrupted microvascular blood flow from the original injury can lead to edema, increasing intraneural pressure and causing additional compression and damage to the myelin sheath. 6 Elevated intraneural pressure can further restrict blood flow, leading to additional inflammation, in a so-called “cumulative injury cycle.” Neuritis, defined as inflammation of a nerve causing pain or disability, has been described as a common occurrence following hand surgery or traumatic injuries including lacerations, blunt injuries, and chronic compression. 7
Even if the nerve is not significantly damaged by the initial trauma, subsequent edema, inflammation, and fibrosis in the surrounding soft tissue may cause compression and impingement of nerve gliding, ultimately leading to ischemia, inflammation, demyelination, and impaired nerve function. Thus, it has been speculated that modulation of the inflammatory response both within and surrounding the nerve may help to alleviate symptoms such as thermal hyperalgesia and mechanical allodynia experienced by patients with neurotmetic injuries. 5
One potential avenue for immunomodulation may be through the use of amniotic membranes. Amniotic membranes have become increasingly popular as a biologic dressing as they are known to contain cytokines that polarize macrophages toward the M2 phenotype and have demonstrated the ability to modulate the inflammatory response. 8 Multiple amniotic membrane–derived products (e.g. AmnioFix, EpiFix, AmnioGuard, Acelagraft, XWrap Hydro Plus, Clarix, Nucell) have been used in a variety of procedures to prevent scarring, reduce inflammation, and promote regeneration of native tissue.9–11 While most amniotic membrane products were developed for wound care, some have recently demonstrated utility in nerve repair as a wrap to reduce adhesion formation and scarring.9,10 Early reports of amniotic membrane as an adjunct to nerve repair are promising, however, the literature on its clinical use in this context remains limited. 10
Processed human umbilical cord membrane (PUCM) has recently been developed to provide the known benefits of amniotic membrane–derived products with improved handling, resorption time, and neuroregenerative properties. Given the shortcomings of current treatment algorithms to address axonotmetic injuries, the authors designed a study to evaluate postoperative outcomes of multi-level surgical reconstructions in which a commercially available PUCM (Avive® Soft Tissue Membrane, Axogen Inc, Alachua, FL) was used to cover nerves exposed by trauma to the upper and lower extremities. No published studies have described the use of PUCM in traumatic peripheral nerve injury.
Methods
Approval was obtained from our Institutional Review Board, and we performed a prospective, single-center pilot study of patients undergoing multi-level surgical reconstruction following trauma with exposure of a non-transected peripheral nerve. The objective of this study was to determine whether the use of PUCM in these procedures may lead to clinically significant improvements in outcomes compared to similar injuries reported in the literature, as well as an internal control group. Written informed consent was obtained from all subjects included in our study.
Treatment group
Patients were prospectively included in the study if they were 13–70 years of age, sustained an injury to the upper or lower extremity with concurrent damage to bone or soft tissue, and had at least one exposed nerve within the zone of injury. Patients were excluded if they had a complete nerve transection, initial surgical intervention occurred > 21 days after initial injury, and if the patient had chronic diseases that affected nerve repair (such as diabetes mellitus type I or II) or received therapy for their injury, which affects nerve repair. Patients with a history of a chronic ischemic condition of the repaired extremity or who had injuries with significant vascular damage resulting in inadequate perfusion despite repair were also excluded from the study. Patients meeting the study inclusion and exclusion criteria were consented to participate in prospective follow-up assessments. Demographic information, mechanism of injury, affected tissue, concomitant injuries, and medical history were recorded preoperatively. PUCM usage, as well as reconstruction procedures, surgical time, and adverse effects or complications were recorded intraoperatively.
Given the high variability of traumatic injuries and the lack of published studies on non-transected nerve injury outcomes, sample size calculations determined by power analysis were untenable. The decision to enroll 20 patients in our study was made with regard to the number of similar injuries seen in our practice over a 1-year period (the year prior to beginning our study).
The following outcome measurements were collected where possible: postoperative Visual Analog Scale (VAS) pain,12,13 Tinel’s sign, 14 protective sensation, static two-point discrimination (s2PD) assessment,15,16 moving two-point discrimination (m2PD) assessment, 17 Semmes–Weinstein monofilament assessment, 18 range of motion (ROM) assessments, pinch/grip test, 19 muscle strength, 19 Medical Research Council Classification (MRCC) for sensory/motor function, 20 and electromyography (EMG) results. 21
The QuickDASH 17 and LEFS PROMs 22 were utilized for upper and lower extremity injuries, respectively, along with the SF-36 23 for quality-of-life assessment. Postoperative medication, economic data, and revision procedures were also collected at follow-up visits. Follow-up data were collected at 1-week and 3, 6, and 9 months postop, when possible.
Retrospective control group
Using the same inclusion/exclusion criteria, data were collected retrospectively for patients treated in the year prior to PUCM usage at our center.
Statistical analysis
One-tailed paired t-tests were performed to determine whether there was significant improvement from the first measurement to final follow-up. One-tailed Welch t-tests were performed to compare our treatment cohort with our internal control group, as well as outcomes of similar nerve procedures reported in the literature. Post hoc power analysis was performed with α = 0.05.
Results
A total of 20 patients with PUCM placement to cover exposed nerves after traumatic injury and subsequent neurolysis and/or nerve transposition were included in our study (Table 1). In cases where comorbidities necessitated additional surgery after placement, PUCM was observed to stay in the desired position without visible degradation. Our study found no complications up to 9 months following traumatic injury with exposed nerve.
Summary of clinical cases using processed umbilical cord membrane (PUCM).

ATV: all-terrain vehicle.
Nineteen patients (95%) had a follow-up of ⩾ 3 months, 90% ⩾ 6 months, and 65% ⩾ 9 months. All patients had at least 3 months of follow-up, with mean follow-up of 7.5 months (range: 3–10 months). The mean age was 39 years (range: 15–65). Twelve (67%) patients were male, and the majority of placement sites were in the upper extremity (85%). The most common mechanism of injury was motor vehicle collision, and the ulnar nerve was most commonly injured (Table 1). Mean age of our internal control group was 25.5 years (range: 15–50), and 63.6% (7/11) of control patients were male. Control group injury characteristics are shown in Table 2.
Summary of internal control cases (not using processed umbilical cord membrane).

Mean preoperative VAS pain score was 4.9 (n = 19, range: 0–8). At 3, 6, and 9 months postoperatively, scores were significantly reduced to 1.7 (n = 19, range: 0–7), 1.3 (n = 18, range: 0–4), and 1.7 (n = 13, range: 0–5), respectively (Table 3). Mean pre- and postoperative (average follow-up: 8.3 months) VAS pain scores in the retrospective internal control group were 3.7 (n = 11, range: 0–8) and 1.8 (n = 11, range: 0–5), respectively (Table 3).
Pain scores in our cohort before surgery, and at 3, 6, and 9 months postop; pain scores from our internal control group; and pain scores from similar procedures in the literature.
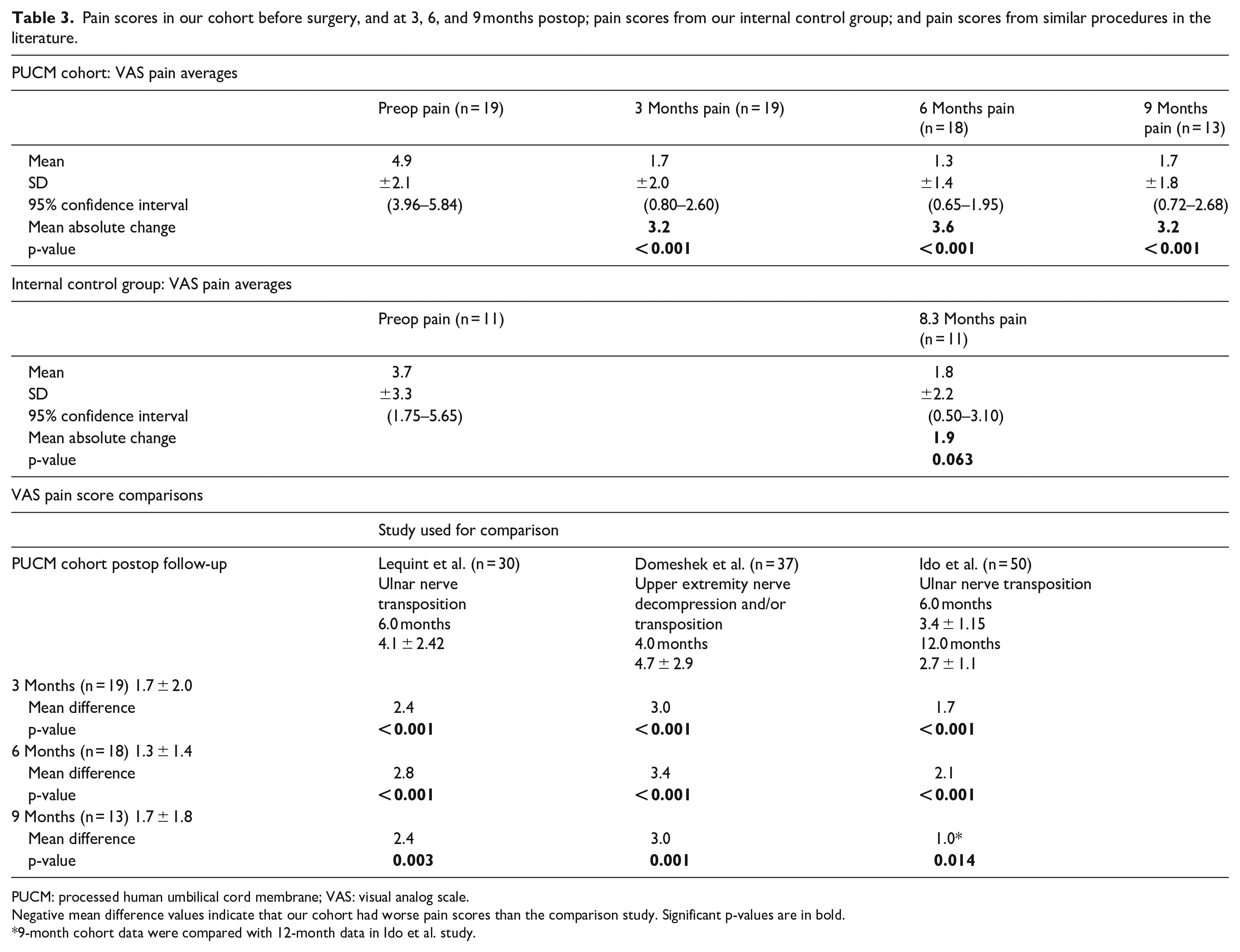
PUCM: processed human umbilical cord membrane; VAS: visual analog scale.
Negative mean difference values indicate that our cohort had worse pain scores than the comparison study. Significant p-values are in bold.
9-month cohort data were compared with 12-month data in Ido et al. study.
Outcomes were classified according to the British Medical Research Council Scale of Sensory Recovery modified by MacKinnon and Dellon (Table 4). 17 At 3 months postop (n = 5), s2PD and/or m2PD outcomes were excellent in 60% (3/5) of patients and very good in 40% (2/5). At 6 months (n = 3), 33% (1/3) had excellent, 33% (1/3) very good, and 33% (1/3) had good sensory outcomes. At 9 months (n = 10), 40% (4/10) had excellent, 50% (5/10) very good, and 10% (1/10) good sensory outcomes (Table 5). 18
ASSH: American Society for Surgery of the Hand.
Semmes–Weinstein monofilament testing 18 outcomes.

At 9-month follow-up, measured as a percentage of the non-operative side, patients had regained an average of 60.6% grip strength (n = 10, range: 10.8–112.5%), 84.6 % key pinch strength (n = 9, range: 15.8–184.0%), 90.5% three-jaw pinch strength (n = 6, range: 15.0–157.7%), and 87.9% tip pinch strength (n = 6, range: 15.4–200.0%).
Nineteen patients (95%) reported improved active ROM (AROM) compared to their preoperative condition. Total active motion results were categorized using the American Society for Surgery of the Hand (ASSH) classification system (Table 4). 24 Of those that were objectively measured, 75.1% had good–excellent outcomes (n = 7 excellent, 5 good), 18.8% had fair outcomes (n = 3), and 12.5% had poor AROM outcomes (n = 2).
QuickDASH scores improved from 48.9 at 3 months postoperatively to 39.0 at 6 months (n = 5 range: 23.3–56.0, p-value: 0.098), and 22.9 at 9 months (n = 8, range: 1–62.5, p-value: 0.004; Table 6).
SF-36 and DASH scores reported for our cohort and the relevant literature.

SF-36 forms were not completed prior to surgery, as the injuries in this study were traumatic in nature. Given this constraint, the decision was made to compare our 9-month postoperative scores to those reported by Novak et al. 25 in a study assessing unrepaired peripheral nerve injuries at 6 months. While our results showed improvements across all dimensions of the SF-36 (Tables 6 and 7), only pain showed significant improvement (Table 7).
Statistical comparisons of final SF-36 and DASH scores in our cohort versus those reported in the literature.
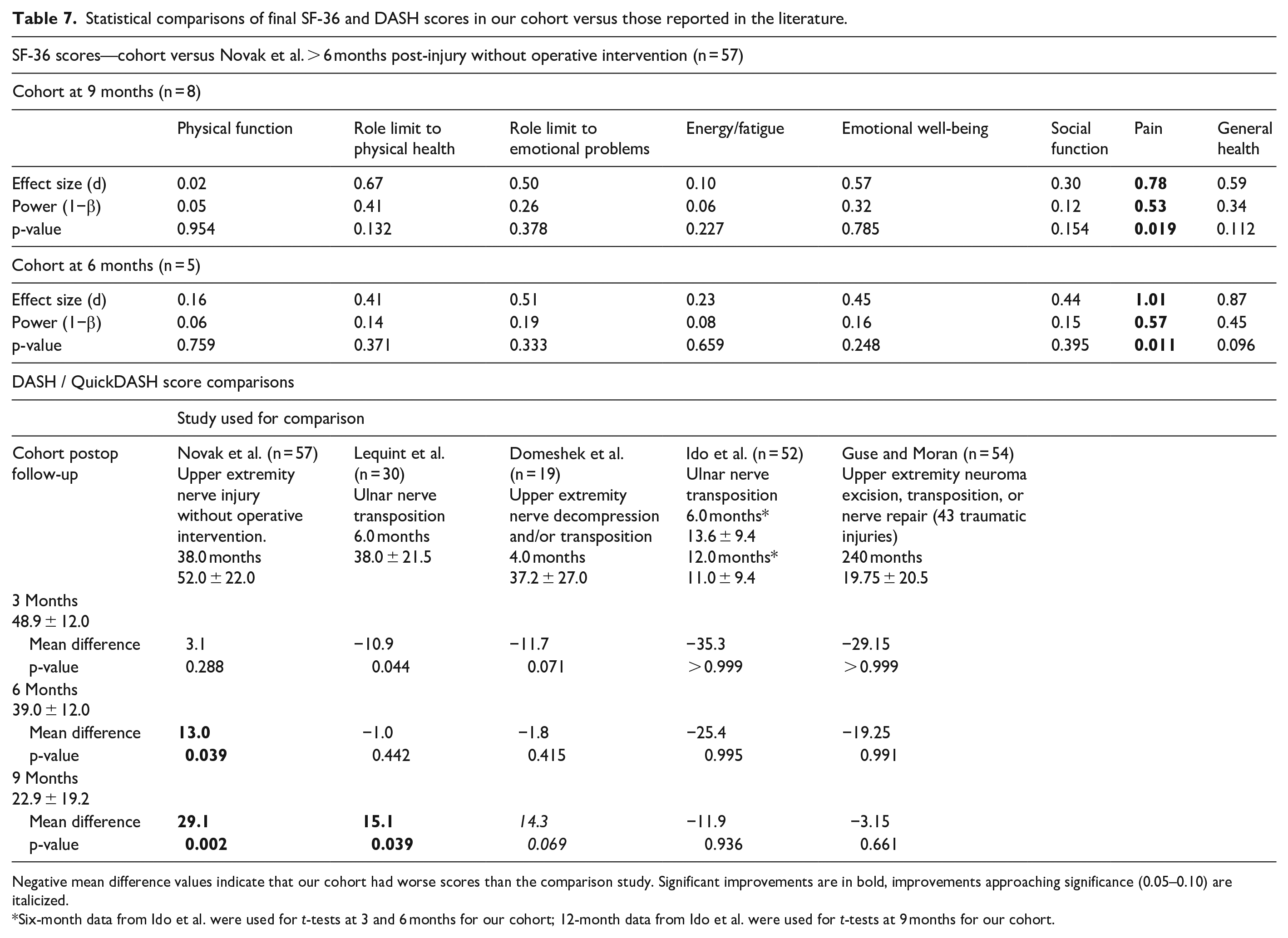
Negative mean difference values indicate that our cohort had worse scores than the comparison study. Significant improvements are in bold, improvements approaching significance (0.05–0.10) are italicized.
Six-month data from Ido et al. were used for t-tests at 3 and 6 months for our cohort; 12-month data from Ido et al. were used for t-tests at 9 months for our cohort.
Discussion
Untreated nerve injuries rarely result in total functional recovery, even when the epineurium remains intact. 26 Often, scarring and adhesion formation between the nerve and the surrounding tissue can interfere with normal axon regeneration and cause painful traction. 26 In addition, the activation of immune cells such as neutrophils, lymphocytes, and macrophages at the site of trauma can exacerbate the inflammation associated with Wallerian degeneration and interferes with normal axonal regeneration. 26
Traumatic peripheral nerve injuries are notoriously difficult to treat and are frequently associated with suboptimal outcomes.17,27 This can be especially true for injuries in which the nerve has been impacted, but not transected, making it difficult for the surgeon to visualize the full extent of the injury. Full recovery to pre-trauma condition is rare for these patients, 2 and many continue to have functional limitations resulting from pain, diminished sensation, scarring, adhesions, muscle weakness, and the need for secondary procedures. 28 In 2015, Emamhadi et al. 29 reported that only 13.1% of patients who underwent ulnar nerve neurolysis between 3 days and 6 months after traumatic injury had good functional outcomes.
Many solutions have been proposed to improve outcomes following peripheral nerve surgery (Supplemental Table 1).28,30–48 In the early 2000s, surgeons began to apply the concept of wrapping nerve lesions to prevent unorganized proliferation of tissue, neural fibrosis, and changes in metabolism using allograft materials. 26 Inflammatory reactions and immune response issues with these early allograft wraps led to the use of nerve wraps comprised of autologous tissues including: vascular grafts, dermofascial fat grafts, and muscle flaps. 49 Though these did help to separate tissues and prevent adhesion, they did little to address the issue of impaired regeneration. Later, synthetic materials were developed for wrapping injured nerves (Supplemental Table 1).30–32 These aimed to address disadvantages of biologic wraps and isolate neurotrophic/neurotropic factors to the site of injury. 50 While synthetic options offer some structural advantages, they are often limited by issues related to their resorption. If materials fail to degrade, they can compress the nerve as it regenerates. Other synthetics may produce harmful products in the degradation process. 50 Unfortunately, synthetic alternatives again failed to sufficiently aid in regenerative processes following repair. More recently, wraps containing absorbable, acellular collagen matrices (ACM) have been used in cases of axonotmesis (Supplemental Table 1).30–32,51 These wraps have been derived from both human and animal tissue, and commercially available products include human amniotic membrane, porcine intestinal submucosa, and collagen type I. 51 These ACM wraps stimulate neovascularization and proliferation of axons with growth factors and cytokines. 52 While many of these synthetic and biological materials have shown some degree of benefit in pre-clinical models, a gold-standard for use in human subjects has not emerged. 50
As surgical use of amniotic membranes increases, interest has evolved to include human umbilical cord membranes (hUCM) due to their thickness, which makes them easier to handle in surgical situations and slower to resorb. In addition to providing all the benefits of amniotic membrane, hUCM also contains factors such as Wharton’s jelly that could confer additional benefits to the neuroregenerative process. Unfortunately, clinical evidence is limited, and there is little data describing the use of novel technologies, such as PUCM, in non-transected nerve injuries resulting from traumatic injuries 32 (Supplemental Table 1). Therefore, our results using PUCM in traumatic, non-transected peripheral nerve injuries were compared to similar procedures using traditional repair algorithms.
Mechanisms of PUCM
While the driving mechanisms behind PUCM’s neurogenerative properties are still being elucidated, the results of our study indicate that it may be a useful adjunct in treating peripheral nerve injuries. In a 2019 review of emerging trends in peripheral nerve repair, Carvalho et al. 53 pointed out that novel products must not only act as a barrier, but support host regenerative processes.
Amniotic membrane–derived products are known to reduce scar formation by utilizing hyaluronic acid present in the mesenchymal layer to inhibit fibroblast activation.9,11 Interleukin-10 (IL-10) in the epithelial cells of amniotic tissue suppresses the expression of pro-inflammatory cytokines and antigens to reduce inflammation. 10 Amniotic membrane wraps have been used successfully to wrap the neurovascular bundle in prostectomy, accelerating return to continence and potency compared to controls. 54 In a rat model of peripheral nerve injury, Echeverry et al. 5 showed that selective modulation of pro-inflammatory processes may alleviate neuropathic pain without delaying nerve regeneration. In addition to their anti-scarring and anti-inflammatory properties, growth factors within amniotic mesenchymal and epithelial cells promote the proliferation and differentiation of epithelial cells.10,11
PUCM is minimally processed for maximum retention of beneficial proteins in umbilical cord tissue such as IL-4, IL-10, and growth factors including epidermal growth factor (EGF), fibroblast growth factor (FGF), platelet-derived growth factor (PDGF), vascular endothelial growth factor (VEGF), and transforming growth factor-beta (TGF-β3). 55 Damaged microcirculation is known to hinder nerve regeneration and functional recovery. 26 VEGF has been shown to accelerate regrowth of perforating vessels that feed the internal vascular structures of nerves. This increase in circulation can boost axonal regrowth and Schwann cell proliferation in regenerating nerves. 56 Furthermore, IL-4 has been shown to both inhibit neuroinflammation 57 and stimulate M2 (repair type) macrophages in vitro and in vivo,57,58 while the combination of IL-4 and IL-10 has been shown to increase eosinophil migration into surrounding tissue. 58 Eosinophils, while known more widely for their role in immune response, have the ability to produce nerve growth factor (NGF) as well as neurotrophin-3 (NT-3)—a protein that promotes peripheral nerve growth, differentiation, and survival. 53 Unfortunately, after peripheral nerve injury, levels of NT-3 appear to remain unchanged. 59 Given the lack of an endogenous response and that NT-3 is not yet commercially available, PUCM may serve an important role in upregulating NT-3 following peripheral nerve injury.
Pain and functional recovery
Pain is perhaps the most important outcome measure to consider in peripheral nerve repair given its impact on nearly every element of functional recovery. 60 In their study of peripheral nerve injuries > 6 months post-injury without operative intervention, Novak et al. 25 found that worse scores on the SF-36 bodily pain domain were significantly associated with greater injury-related disability (measured by DASH). A similar study of 124 patients with untreated peripheral nerve injuries (median of 14 months post-injury) showed that pain disability scores accounted for 57% of variance in DASH scores and 55% of variance in illness intrusiveness. 60
While the majority of studies on pain assess pharmacotherapy interventions, a reduction in patient-reported pain greater than two points or 30% of preoperative VAS has been proposed as a clinically significant reduction in neuropathic pain. 61 While there is currently no consensus on the degree of reduction that constitutes efficacy of a surgical intervention, VAS is frequently used as an objective measure to support clinical outcomes following nerve surgery. In our study, pain decreased greater than two points on average (mean reduction of 3.2, 3.6, and 3.2 at 3, 6, and 9 months, respectively) with 83.3% (n = 10) of patients reporting decreased pain at 9-month follow-up. Both 6- and 9-month postoperative pain scores were significantly lower in our cohort than those reported in the literature for similar procedures (Table 2).62–64 In addition, Welch’s t-tests revealed that our 6- and 9-month SF-36 pain scores were significantly improved compared to similar injury patterns that did not undergo surgical intervention (Table 6). 25
Sensory recovery in our cohort compared favorably with a 2007 study that included eight partially transected median nerves. 65 In this study of 52 (73.2%) epineural and 19 (26.8%) group fascicular repairs, Vordemvenne et al. 65 included reported 0% excellent, 57% very good, 29% good, and 14% poor outcomes. Our cohort had 40% excellent, 50% very good outcomes, 10% good outcomes, and 0% poor outcomes.
Motor testing in our cohort showed significant improvement in 9-month pinch strength to outcomes reported in the literature (p = 0.022). 66 However, grip strength outcomes for our cohort showed no significant difference compared to similar injuries in the literature (p = 0.149). 66 It is worth noting that our follow-up is relatively short term for this particular outcome measure. 66 Studies assessing strength outcomes often take follow-up measurements years, rather than months, after repair. 66 Given the muscular changes that can occur in the presence of a damaged nerve, it may take longer than 9 months to adequately assess strength outcomes data.
DASH and SF-36—patient-reported outcome measures
Patient-reported outcome measures have recently gained traction as a determining factor for operative success. 17 Compared to ROM and sensory tests such as two-point discrimination, these focus more on quality of life and patient-specific impact of injury and recovery.
The mean 9-month DASH score in our cohort was 22.9 (n = 8, range: 5.0–62.5). While it is difficult to match cohort demographics with published reports, our final scores and mean improvement compared favorably to similar nerve procedures in the literature62–64,67 (Tables 5 and 6). Novak et al. 25 also reported DASH scores and found a mean score of 52 ± 22 in untreated injuries, which was 29.1 points higher (p-value: 0.002) than 9-month QuickDASH scores in our cohort (Table 6). The minimal clinically important difference for the DASH form in similar procedures has been reported between 10 and 14 points, indicating that improvements in QuickDASH scores for our cohort are significantly improved compared to earlier timepoints and similar untreated injuries in the literature. 68
SF-36 data following traumatic nerve injury have also been studied to a limited degree in the literature. Novak et al. 25 reported worse scores in unrepaired peripheral nerve injuries across all but one dimension (physical function) of the SF-36 as compared to our cohort.
Limitations
Concomitant injuries likely affected VAS pain scores and AROM in some cases, making it difficult to determine the cause of functional deficits at final follow-up. This issue is inherent in all studies of traumatic nerve injury as the majority of these involve insult to surrounding structures.69,70 The small sample size of our cohort prevented adequate comparisons of SF-36 outcome measures to prior studies. In addition, our study did not feature a large control group, and baseline outcomes for patients with these injury patterns are not well-documented in the literature. However, since traumatic nerve injuries with exposed nerve are relatively rare, we believe that this preliminary study will provide a basis for further use and evaluation of products such as PUCM for nerve protection following traumatic injury. Furthermore, our data fill a significant gap in the literature, as the majority of published reports on nerve wraps do not address traumatic, non-transected nerve injuries. Despite scheduled visits, and multiple attempts to contact patients for final measurements, there was a high attrition rate after 6 months in our cohort. While this is consistent with the literature on attrition in orthopedic trauma,71–75 more patients with long-term data are needed to adequately compare 9-month data to the earlier timepoints (3 and 6 months).
Other possible limitations to our study are the inclusion of only one hospital site, and the fact that the majority of our patients were male. However, most traumatic nerve injuries occur in the male population and our study’s demographics did not differ significantly from the literature. Given the paucity of evidence on clinical use of PUCM, our data may provide a foundation for the development of future studies. These studies should aim to investigate the efficacy of PUCM and similar products in a multicenter setting to reduce homogeneity and increase sample size.
Conclusion
While our study is not without limitations, there is a need for reports evaluating the efficacy of prophylactic treatments for non-transected traumatic nerve injuries. The data we present may serve as a valuable resource when considering adjunctive treatments in this patient population. The results of this study indicate that the use of PUCM as a soft tissue covering during nerve surgery is safe and resulted in noted improvements in postoperative function (sensation and ROM), pain, and patient-reported outcome scores. In addition, it was noted that these outcomes compared favorably to those reported in the historical literature for traditional nerve repair algorithms.
Supplemental Material
sj-pdf-1-smo-10.1177_20503121211023356 – Supplemental material for Evaluation of postoperative outcomes in patients following multi-level surgical reconstructions with the use Avive™ soft tissue membrane on nerve after traumatic injury of the upper extremity and lower extremity
Supplemental material, sj-pdf-1-smo-10.1177_20503121211023356 for Evaluation of postoperative outcomes in patients following multi-level surgical reconstructions with the use Avive™ soft tissue membrane on nerve after traumatic injury of the upper extremity and lower extremity by Cameron T Cox, Joash R Suryavanshi, Bradley O Osemwengie, Sterling Rosqvist, Matthew Blue, Desirae McKee and Brendan J MacKay in SAGE Open Medicine
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors would like to disclose the following support for DM: paid teaching and consulting from AxoGen Inc, Alachua FL; general research support from the Shannon Holloway Research Chair. The authors would like to disclose the following support for BM: paid teaching and consulting from Axogen Inc, Alachua FL. The remaining authors have nothing to disclose.
Ethical approval
Ethical approval for this study was obtained from TTUHSC Lubbock/Odessa IRB, ID: L18-104.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded in part by Axogen Inc, Alachua, FL.
Statement of informed consent
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008. Written informed consent was obtained from all patients for being included in the study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

