Abstract
Objective:
To determine the hypertonic saline efficacy in children with cerebral edema and raised intracranial pressure.
Method:
Studies assessing the efficacy and safety of hypertonic saline in children with cerebral edema and elevated intracranial pressure were identified using Medline, Web of Science, Scopus, and Google Scholar databases. Two reviewers independently assessed papers for inclusion. The primary outcome was a reduction of elevated intracranial pressure by the administration of hypertonic saline.
Results:
We initially evaluated 1595 potentially relevant articles, and only 7 studies met the eligibility criteria for the final analysis. Out of the seven studies, three of them were randomized controlled trials. Three of the studies found that hypertonic saline significantly reduced elevated intracranial pressure compared to control. One study reported a resolution of the comatose state as a measure of reduced intracranial pressure. It also found a significantly higher resolution of coma in the hypertonic saline group rather than the control. Three studies reported that the reduction of intracranial pressure was comparable between the groups. The random-effects model using pooled estimates from four studies showed no difference in hypertonic saline and conventional therapy mortality outcomes. Hypertonic saline was administered as bolus-only therapy at a rate of 1–10 mL/kg/dose over 5 min to 2 h and or bolus followed by infusion therapy (0.5–2 mL/kg/h). One study reported a twofold faster resolution of high intracranial pressure following hypertonic saline administration compared to controls. The re-dosing schedule varied greatly in all included studies. However, three studies reported adverse events but not methodically, and there were no reports on neurological sequelae.
Conclusion:
Hypertonic saline appears to reduce intracranial pressure in children with cerebral edema. However, we cannot draw a firm conclusion regarding the safest dose regimens of hypertonic saline, including the safe and effective therapeutic hypernatremia threshold in the management of raised intracranial pressure with cerebral edema. Future clinical trials should focus on the appropriate concentration, dose, duration, mode of administration, and adverse effects of hypertonic saline to standardize the treatment.
Introduction
Intracranial pressure (ICP) is one of the most crucial pathophysiological processes which complicates several neurologic conditions, including traumatic brain injury, diabetic ketoacidosis, brain tumors, intracranial infections, ischemic or hemorrhagic stroke, sepsis, and toxic or metabolic encephalopathies in children. The reported mortality from cerebral edema ranges from 20% to 90%. Even survivors from such an episode often experience significant neurological sequelae resulting in a major socio-economic burden.1,2 To improve cerebral perfusion, prompt management of cerebral edema with elevated ICP is recommended. Early intervention also reduces mechanical damage caused by compartmental shifts and local brain tissue compression. 3
Hyperosmolar therapy, hypertonic saline (HTS) or mannitol, is the cornerstone of management for intracranial hypertension with cerebral edema. 4 An ideal hyperosmolar agent should lower ICP whereas preserving cerebral perfusion pressure (CPP). Increasingly, HTS has come forward as a surrogate hyperosmolar agent after several trials reported that the intravenous bolus administration of HTS resulted in a sustained decrease of ICP in patients with cerebral edema, even when elevated ICP is resistant to other therapeutic agents, including mannitol.5,6 The use of mannitol as a sole agent to treat raised ICP has declined from 98% to 52% over a decade because of its many clinically significant adverse effects, including renal failure, electrolyte disorders, rebound aggravation of ICP, and hypovolemia. 7 Simultaneously, the use of HTS alone has increased from 2% to 39%. 8 However, the treatment regimen might be variable depending on underlying mechanism of cerebral edema. There is a loss of cerebral autoregulation and cerebral vasospasm in traumatic brain injury, leading to regional ischemia and intracellular swelling, initiating a complex series of pathophysiologic changes contributing to cerebral edema and raised ICP. 9 Conversely, in metabolic etiologies, there is homogeneous edema without focal ischemia or injury.
The recent consensus recommends the use of HTS or mannitol for reducing ICP in children with cerebral edema. 10 Some authors have recommended mannitol to be the “gold standard” for the treatment of cerebral edema, mainly due to its extended history. 11
However, the use of HTS can still be controversial in children with cerebral edema due to various medical or surgical conditions in neuro-critical care. There is minimal evidence regarding optimal concentration, dose, infusion rate, and frequency of HTS, particularly in children. Therefore, we aimed to conduct a systematic review and meta-analysis to determine the existing evidence on HTS to lower elevated ICP in children with cerebral edema. We also sought to determine the HTS doses, concentration, frequency of redosing, neurological sequelae, and fatality outcomes in children with cerebral edema.
Methods
Study selection
We registered our protocol before conducting this review with PROSPERO, an international database of prospectively registered systematic reviews in health and social care (ID CRD42017072694). We conducted the systematic review following the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines. Our search strategy was designed as a priori for studies’ eligibility. We searched Medline (PubMed), Web of Science, Scopus, and Google Scholar databases. We searched existing literature on the English language and applied no restriction on the year. We also screened reference lists of all relevant studies for additional applicable articles. We used MeSH headings where applicable. We included all trials on children aged 0–18 years who had cerebral edema with elevated ICP and treated with intravenous HTS.
We included experimental/quasi-experimental trials or observational studies (cohort/case-control/cross-sectional) comparing HTS with any other osmotic or therapeutic agents, such as mannitol or any other standard treatment for raised ICP. We evaluated children having both infectious and non-infectious etiology of cerebral edema. We excluded review articles, case reports and case series, research on animal models, and studies that did not use HTS as an intervention to reduce ICP. We also excluded studies without any comparator group.
Outcome and search strategy
Our primary outcome, selected by our reviewers a priori, was reduction of elevated ICP in children with cerebral edema. Our secondary outcomes were case fatality, including the comparison of HTS and other therapeutic agents for cerebral edema, HTS concentration, administration and dosage, and adverse events, as well as neurological outcomes. We defined elevated ICP as a sustained elevation of ICP > 20 mm Hg (27 cmH2O) for longer than 5 min, measured by an external ventricular drain or intraparenchymal ICP monitor and or Glasgow coma scale (GCS) < 8 with signs of brainstem dysfunction; and abnormal results consistent with cerebral edema in neuroimaging including computed tomography and magnetic resonance imaging. 12
An experienced librarian (M.A.M.) developed database-specific search strategies. We have used a comprehensive search strategy according to the PRISMA 13 guidelines on the management of raised ICP with HTS. Following key words were used: “cerebral edema,” “intracranial hypertension or elevated ICP,” “hypertonic saline,” and “children” (Full search strategy is available from authors). Four teams of paired reviewers (F.A., N.J.S., L.S., I.P., M.S., H.S., S.N., M.B.S.) with medical expertise in pediatrics and training in the systematic review and meta-analysis independently reviewed the title and abstract to include articles that met the inclusion criteria as determined by the Population, Intervention, Comparison, Outcome and Study design (PICOS) outline. We resolved disagreements through discussions with a third reviewer (M.J.C./T.A./C.A.K.).
Data extraction
We used a standardized data extraction template to collect information from each eligible study. We extracted information regarding study characteristics such as the first author, publication year, study site, duration and research design, number of participants and participant’s characteristics, inclusion and exclusion criteria, invasive or non-invasive ICP monitoring methods and rate of ICP reduction, indication, dose, frequency and concentration of HTS, description of the comparator, and outcomes of interest with mortality.
Quality assessment of included studies
We used “Risk Of Bias In Non-randomised Studies—of Interventions” (ROBINS-I) tool to assess methodological quality of non-randomized studies of effect Interventions. 14 We assessed seven domains through which bias might be introduced before or after the intervention. In the first three domains, we addressed bias due to confounding, selection of participants into the study, and bias in the classification of interventions. The other four domains covered biases due to deviations from intended interventions, missing data, measurement of outcomes, and selection of the reported result. The judgment of risk of bias in each domain was based on the answers to the broadly factual signaling questions, which then provided the basis for an overall risk of bias judgment for a particular outcome. A study with low risk of bias for all domains was judged as “low risk” category, and low or moderate risk of bias for all domains as “moderate risk.” We categorized the study at “serious risk” if we found any important problem informed by answers to the signaling questions in at least one domain. If the study was too problematic in at least a domain to provide any useful evidence on the effects of intervention, we judged the study at “critical risk.” We assessed risk of bias of included randomized trial using the Cochrane collaboration’s tool. 15
Any disagreement was resolved by discussion among reviewers and by consultation with a third reviewer if necessary.
Data synthesis and analysis
The authors intended to perform meta-analysis for all outcomes, particularly ICP reduction rate, but because of marked heterogeneity of study design and outcome, we were not capable of pooling results for our primary outcome. Therefore, we carried out a narrative synthesis to summarize the results.
However, we performed meta-analysis to pool fatality outcomes of included studies using Rev Man 5.3. We expressed summary estimates for mortality as odds ratio (OR) and 95% confidence limits using a random-effects model analysis. We used I-square (I2) statistics to quantify the proportion of statistical heterogeneity. To detect substantial heterogeneity, we considered I2 statistics above 50%.
Results
Identification and selection of studies
The comprehensive search identified 1595 potentially relevant articles after the removal of duplicates. Initial title and abstract screening identified 13 articles relevant to the effects of HTS in treating cerebral edema in children (Figure 1). An additional seven studies were excluded. The reasons for exclusion were lack of comparison group,16–18 the effect of HTS on ICP was not reported,8,19,20 and HTS was used during critical care transport 21 (Figure 1).
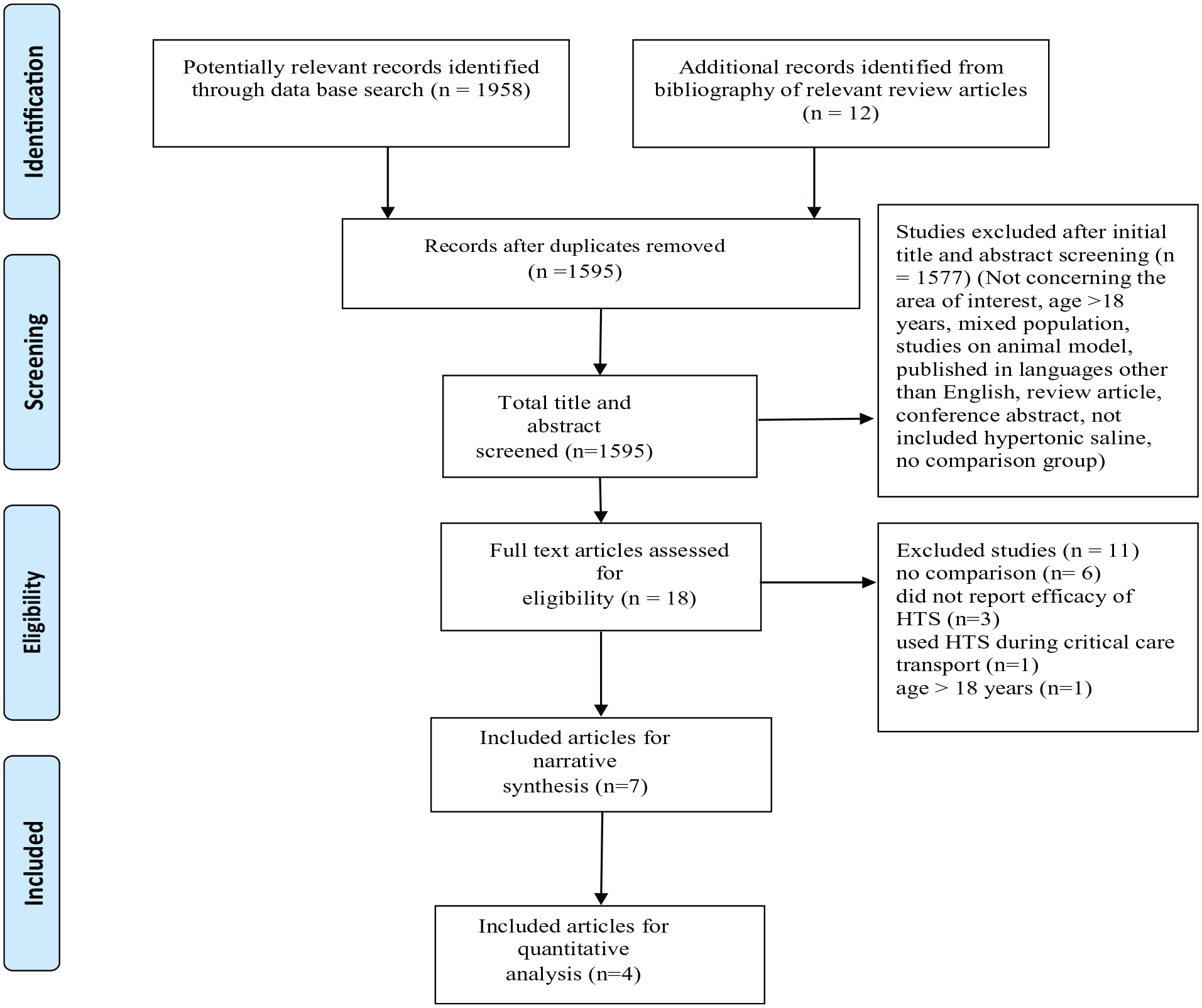
Flow diagram for study selection and exclusion.
We performed a top-up hand search in December 2020 by reviewing the reference list of newly published articles and found additional five articles potentially relevant to our study. We included one article 22 after reviewing the full text and excluded four studies, three due to lack of comparison group23–25 and the remaining one included participants older than 18 years. 26 Thus, this systematic review contains a total of seven studies.
Characteristics of included studies
In this review, we have included seven studies after full-text screening: three were randomized controlled trials (RCTs)27–29 and the remaining four were retrospective non-randomized study of intervention.22,30–32 Of the seven studies, only one open-labeled RCT compared the effect of equiosmolar doses of HTS and mannitol in lowering ICP in children. 29
A total of 202 children aged a month to 18 years were included in these studies. The characteristics of the included studies are summarized in Table 1. In total, 56% of children had a traumatic brain injury.
Characteristics of studies included in systematic review.

TBI: traumatic brain injury; RCT: randomized controlled trial; IC: intracranial; FHF: fulminant hepatic failure; HIE: hypoxic ischemic encephalopathy; CSF: cerebrospinal fluid.
Risk of bias assessment
Our included randomized trials had fair quality in terms of risk of bias. However, there were no high-risk criteria in any domain. Two criteria remained unclear (allocation concealment, selective reporting), and our reviewers assessed that this was unlikely to have biased the outcome (Supp file 2, 3).
We evaluated the risk of bias of included observational studies using the ROBINS-I tool (Supp file 4). All included observational studies were judged to be at serious risk of bias because the intervention status was not well defined, the bias in selecting participants into the study, and bias due to confounding and lack of information regarding deviations from intended interventions (Supp file 4).
ICP
All studies examined the reduction of ICP as an outcome measure. Three studies, including one randomized cross-over trial, found a significant reduction of ICP in the HTS group compared to the control group. Fisher et al. reported that the baseline ICP in the intervention group was 19.9 mm Hg, significantly decreased to a mean of 15.8 mm Hg 2 h following HTS (p = 0.003). Vats et al. showed following administration of HTS, ICP significantly reduced as early as 30 min from the baseline and continued to be reduced at 60 and 120 min. Conversely, mannitol causes a significant reduction of ICP from baseline at 60 and 120 min. Shein et al. found that 3% HTS significantly reduced ICP within 5 min of therapy completion (p < 0.001). They also found HTS was associated with a faster (twofold) resolution of ICP compared to both pentobarbital (hazard ratio (HR) = 2.171 (1.062–4.439)) and fentanyl (HR = 1.848 (1.086–3.155)) after adjusting for confounders such as age, GCS score, injury type, baseline ICP and concurrent infusions of pentobarbital, HTS, and neuromuscular blockers (Table 2). The authors also reported significant improvement of CPP following HTS therapy (p < 0.001) and significant deterioration of CPP following fentanyl administration (p < 0.001).
The effect of HTS (hypertonic saline) on elevated intracranial pressure (ICP), cerebral perfusion pressure (CPP), and overall study results.
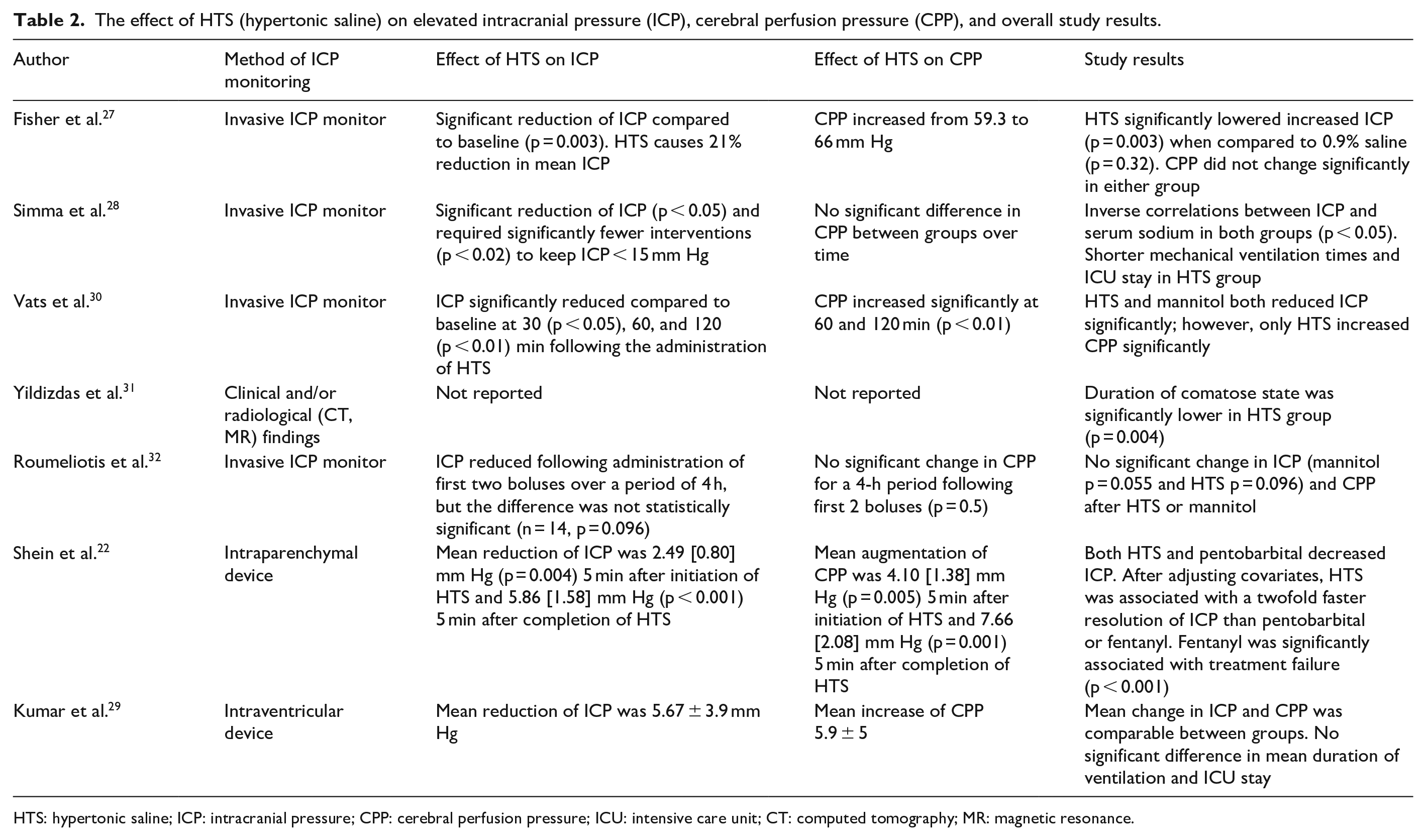
HTS: hypertonic saline; ICP: intracranial pressure; CPP: cerebral perfusion pressure; ICU: intensive care unit; CT: computed tomography; MR: magnetic resonance.
Three studies reported that both hyperosmolar agents decreased ICP adequately, and the differences were comparable between the groups (Table 2). Kumar et al. compared the efficacy of equiosmolar dose of HTS and mannitol in reducing ICP. The authors initially performed cerebrospinal fluid drainage to control episodes of elevated ICP. Hyperosmolar agents were administered if episodes of elevated ICP were not controlled by cerebrospinal fluid drainage. Authors reported that the mean reduction of ICP was 7.13 ± 2.9 and 5.67 ± 3.9 mm Hg following administration of mannitol and HTS, although the difference did not achieve statistical significance. However, the CPP showed a trend of early improvement in the HTS group compared to the mannitol group. 29 Roumeliotis et al. examined the impact of hyperosmolar therapies on ICP following administration of first two boluses (ICP > 20 mm Hg) only for 4 h, and the impact of further boluses was not evaluated. The authors reported that although both HTS and mannitol decreased ICP compared to baseline, the difference was not statistically significant for the initial 4 h. 32 Simma et al. compared HTS to Ringer’s Lactate solution and found no significant difference in ICP and cerebral perfusion pressure between the groups. However, to keep ICP below 15 mm Hg, HTS group required significantly fewer interventions (p < 0.02) and had significantly shorter intensive care unit (ICU) stay time (p = 0.4) compared to the control group. 28
Only one study comparing HTS and mannitol reported a comatose state resolution as a measure of reduced ICP. 31 The coma’s resolution was significantly higher in the intervention group compared to the control group, 89 ± 42 h and 123 ± 48 h, respectively (p = 0.004).
Mortality
Five studies reported on case fatality. Fisher et al. reported five deaths during the hospital stay, but we excluded this result from the meta-analysis as the trial had a cross-over design. Shein et al. reported a 32% in-hospital case fatality rate, and we failed to pool this data because all patients sequentially received fentanyl, HTS, and pentobarbital. Meta-analysis using the results pooled from two randomized trials and two non-randomized studies of intervention revealed no statistically significant difference between treatment groups in terms of mortality (Figure 2). Considering methodological difference, if we exclude observational studies from the meta-analysis, we did not observe any significant difference in the outcome.

Results from meta-analysis of included studies on the effects of hypertonic saline on case fatality in children with cerebral edema and intracranial hypertension. CI: confidence interval.
HTS dosage, administration, and concentration
The concentration of HTS ranged from 1.5% to 3% in all studies (Table 3). The most commonly used concentration of HTS was 3%. The administration volume and type varied greatly. HTS was administered intravenously as bolus-only therapy at a rate of 1–10 mL/kg/dose and or bolus followed by infusion therapy (0.5–2 mL/kg/h). Kumar et al. administered 3% HTS as a bolus of 2.5 mL/kg for 5 min and required a mean of 4.5 doses to control episodes of elevated ICP. The number of repeat doses also varied considerably. The duration of bolus therapy ranged from 5 min to 2 h. Roumeliotis et al. reported a mean bolus dose of HTS was 1.8 mL/kg, and 50% of participants received a continuous infusion for the first 48 h of admission. Shein et al. administered 3% HTS over 10–21 min. Significant improvement of ICP was noted within 5 min of initiation of bolus (p = 0.004) and 5 min after completion of therapy (p < 0.001) compared to controls. Simma et al. 28 administered 1.5% HTS as a maintenance fluid for 72 h (Table 3).
Concentration, dose, frequency, and adverse effects of hypertonic saline and effect on serum chemistry.

HTS: hypertonic saline; ICP: intracranial pressure; CPP: cerebral perfusion pressure; BSA: body surface area; IQR: interquartile range.
Adverse events and neurological sequelae
Two randomized trials and one retrospective study reported adverse events (Table 3), but none of the studies reported the events systematically, and there were no reports on neurological sequelae. A randomized trial reported an average 7 mEq/L elevation of serum sodium level after a bolus therapy of 10 mL/kg 3% HTS therapy, but no significant changes of serum creatinine and blood urea nitrogen (BUN) measurements; 27 there was no protracted or rebound increase of ICP throughout the study period. The average serum sodium ranged from 149 to 157 mosm/L (three studies), and an RCT found an inverse correlation between serum sodium and ICP (p < 0.001). 28 Roumeliotis et al. 32 found that serum sodium did not change significantly from baseline following administration of HTS (pre-dose sodium = 142 ± 7 vs post-dose = 142 ± 6, p = 0.1).
Simma et al. 28 reported that in the HTS group, 40% of participants developed a complication, 7% developed two complications, 25% had pneumonia, and 7% had acute renal failure, but there was no significant difference between the groups in terms of complications. In a retrospective analysis, Yildizdas et al. 31 found that children treated with HTS developed hyperchloremic metabolic acidosis (2/25, 8%) and diabetes insipidus (1/25, 4%). No acute renal failure, pulmonary edema, or subarachnoid hemorrhage was detected.
Discussion
We include seven studies involving a total of 202 children with cerebral edema. For the outcome of “ICP,” no meta-analysis was possible due to inconsistencies in the definition of outcome measure and heterogeneity in reporting ICP changes. Therefore, we reported results by narrative synthesis. In essence, included study results indicated that ICP was reduced significantly by administering HTS compared with baseline, and HTS therapy is as effective as mannitol or other ICP lowering agents. Moreover, HTS has some additional benefits, such as the faster resolution of ICP and augmentation of CPP. However, treatment selection might be individualized based on sodium level and cerebral hemodynamic. Meta-analysis using the pooled estimates for “mortality” outcome showed comparable results between treatment groups. There was no consensus between the included studies regarding dose volumes, frequencies, timing, and indications of redosing; and use of bolus and or infusion therapy. Most studies used 3% HTS, and this concentration appears safe and effective at reducing ICP in children with cerebral edema. However, due to the heterogeneity of data and limited numbers, we are unable to make any recommendation regarding the dosing regimen, including dose volumes. None of the studies systematically reported data on adverse effects, including neurological sequelae.
The mechanism of action behind ICP lowering effect of HTS is yet to be well implicated. However, existing evidence depicts that HTS generates an osmotic gradient from the intracellular to the interstitial space, leading to shrinkage of brain tissue and therefore a reduction in ICP. HTS also enhances volume resuscitation and upsurges circulating blood volume, mean arterial blood pressure, and CPP. 5
In a meta-analysis of five randomized clinical trials on the adult population, Kamel et al. compared equiosmolar doses of HTS to mannitol and revealed HTS was more effective in lowering ICP than other measures such as mannitol. In Kamel et al.’s 7 meta-analysis, there was also a trend toward more significant quantitative ICP reduction with HTS therapy. Feng et al. 33 in their meta-analysis showed that HTS had same effectiveness in the reduction of ICP as other conventional therapies in the Chinese mainland adult population. There was no significant difference in mean arterial pressure and mean venous pressure between the two treatment groups. Stopa et al., 34 in a recent systematic review, found that both HTS and mannitol effectively lower ICP and improve clinical outcomes in children with TBI. Authors failed to identify which agent is better or what treatment protocol to follow due to a lack of high-quality data. Although we included children with cerebral edema due to various etiologies, including TBI, our conclusions are nearly similar due to lack of high-quality RCTs. However, HTS has demonstrated efficacy and favorable cerebral hemodynamic and thus deserve consideration as a potential first-line therapy in children with cerebral edema.
Our review had some limitations. Because we tried to find the effect of HTS in children with cerebral edema with elevated ICP, very few studies met our eligibility criteria; furthermore, we did not include unpublished studies and studies published in other languages due to lack of professional expert translator, so our review might have publication bias. Marked heterogeneity of included studies is another key limitation of this review. There was heterogeneity between study population and methodologies, variations in the comparison group, and little consistency regarding HTS infusion rate and redosing schedule. Markedly, heterogeneous primary outcome measures of included studies forbade us for a meta-analysis for the rate of ICP reduction and other estimates. However, based on clinical homogeneity, we have included four studies in the pooled analysis for mortality outcome even though they are methodologically different and reported various outcome measures. Nevertheless, the review has some strength. Randomized controlled studies and non-randomized trials of interventions (retrospective study) were all included. We included studies that compared HTS with mannitol or other ICP lowering agents to comprehend better potential therapy in providing an improved outcome for children with cerebral edema. Besides, all studies about HTS therapy in children with multiple etiologies of cerebral edema were included; there were no restrictions on the date of publication year.
Conclusion
The current evidence suggests that HTS reduces ICP, improves CPP, and is materialized to be as effective as mannitol in children with cerebral edema. HTS may deserve consideration as the first-line therapy for elevated ICP in such children, as it exhibited favorable cerebral hemodynamic. However, we are unable to draw firm conclusion regarding the safest dose regimens of HTS, repeat dosing, and safe and effective threshold of therapeutic hypernatremia in the management of cerebral edema.
Randomized clinical trials comparing equiosmolar and equivolumic HTS with conventional mannitol therapy are crucial for children with raised ICP. More extensive clinical trials with adequate sample size will guide the most suitable first-line treatment regimen for hyperosmolar agents in the pediatric population and optimal dose, duration, and administration mode (multiple boluses vs continuous infusion) of HTS, including the safe and effective threshold of therapeutic hypernatremia. Furthermore, future trials should consider testing the adverse effect of HTS, including fatality and long-term neurological sequelae for children with cerebral edema.
Supplemental Material
sj-doc-5-smo-10.1177_20503121211004825 – Supplemental material for Effect of hypertonic saline in the management of elevated intracranial pressure in children with cerebral edema: A systematic review and meta-analysis
Supplemental material, sj-doc-5-smo-10.1177_20503121211004825 for Effect of hypertonic saline in the management of elevated intracranial pressure in children with cerebral edema: A systematic review and meta-analysis by Farzana Afroze, Monira Sarmin, CA Kawser, Sharika Nuzhat, Lubaba Shahrin, Haimanti Saha, Nusrat Jahan Shaly, Irin Parvin, Mohsena Bint-e Sharif, M Al Mamun, Tahmeed Ahmed and Mohammod Jobayer Chisti in SAGE Open Medicine
Supplemental Material
sj-docx-1-smo-10.1177_20503121211004825 – Supplemental material for Effect of hypertonic saline in the management of elevated intracranial pressure in children with cerebral edema: A systematic review and meta-analysis
Supplemental material, sj-docx-1-smo-10.1177_20503121211004825 for Effect of hypertonic saline in the management of elevated intracranial pressure in children with cerebral edema: A systematic review and meta-analysis by Farzana Afroze, Monira Sarmin, CA Kawser, Sharika Nuzhat, Lubaba Shahrin, Haimanti Saha, Nusrat Jahan Shaly, Irin Parvin, Mohsena Bint-e Sharif, M Al Mamun, Tahmeed Ahmed and Mohammod Jobayer Chisti in SAGE Open Medicine
Supplemental Material
sj-docx-4-smo-10.1177_20503121211004825 – Supplemental material for Effect of hypertonic saline in the management of elevated intracranial pressure in children with cerebral edema: A systematic review and meta-analysis
Supplemental material, sj-docx-4-smo-10.1177_20503121211004825 for Effect of hypertonic saline in the management of elevated intracranial pressure in children with cerebral edema: A systematic review and meta-analysis by Farzana Afroze, Monira Sarmin, CA Kawser, Sharika Nuzhat, Lubaba Shahrin, Haimanti Saha, Nusrat Jahan Shaly, Irin Parvin, Mohsena Bint-e Sharif, M Al Mamun, Tahmeed Ahmed and Mohammod Jobayer Chisti in SAGE Open Medicine
Supplemental Material
sj-docx-6-smo-10.1177_20503121211004825 – Supplemental material for Effect of hypertonic saline in the management of elevated intracranial pressure in children with cerebral edema: A systematic review and meta-analysis
Supplemental material, sj-docx-6-smo-10.1177_20503121211004825 for Effect of hypertonic saline in the management of elevated intracranial pressure in children with cerebral edema: A systematic review and meta-analysis by Farzana Afroze, Monira Sarmin, CA Kawser, Sharika Nuzhat, Lubaba Shahrin, Haimanti Saha, Nusrat Jahan Shaly, Irin Parvin, Mohsena Bint-e Sharif, M Al Mamun, Tahmeed Ahmed and Mohammod Jobayer Chisti in SAGE Open Medicine
Supplemental Material
sj-tif-2-smo-10.1177_20503121211004825 – Supplemental material for Effect of hypertonic saline in the management of elevated intracranial pressure in children with cerebral edema: A systematic review and meta-analysis
Supplemental material, sj-tif-2-smo-10.1177_20503121211004825 for Effect of hypertonic saline in the management of elevated intracranial pressure in children with cerebral edema: A systematic review and meta-analysis by Farzana Afroze, Monira Sarmin, CA Kawser, Sharika Nuzhat, Lubaba Shahrin, Haimanti Saha, Nusrat Jahan Shaly, Irin Parvin, Mohsena Bint-e Sharif, M Al Mamun, Tahmeed Ahmed and Mohammod Jobayer Chisti in SAGE Open Medicine
Supplemental Material
sj-tif-3-smo-10.1177_20503121211004825 – Supplemental material for Effect of hypertonic saline in the management of elevated intracranial pressure in children with cerebral edema: A systematic review and meta-analysis
Supplemental material, sj-tif-3-smo-10.1177_20503121211004825 for Effect of hypertonic saline in the management of elevated intracranial pressure in children with cerebral edema: A systematic review and meta-analysis by Farzana Afroze, Monira Sarmin, CA Kawser, Sharika Nuzhat, Lubaba Shahrin, Haimanti Saha, Nusrat Jahan Shaly, Irin Parvin, Mohsena Bint-e Sharif, M Al Mamun, Tahmeed Ahmed and Mohammod Jobayer Chisti in SAGE Open Medicine
Footnotes
Acknowledgements
We gratefully acknowledge the donors for their support and commitment to icddr,b’s research efforts. We would like to express our sincere thanks to all clinical fellows, nurses, members of feeding team, and cleaners of the hospital for their invaluable support and contribution to patient care.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
We have conducted a systematic review and meta-analysis, so ethical approval is not applicable. We registered our protocol prior to conducting this review with PROSPERO, an international database of prospectively registered systematic reviews in health and social care (ID CRD42017072694). We conducted the systematic review following the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: icddr,b, and its donors supported this work, which provides unrestricted support to the institution for its operations and research. Current donors providing unrestricted support include Government of the People’s Republic of Bangladesh, Global Affairs Canada (GAC), Swedish International Development Cooperation Agency (Sida), and the Department for International Development, UK (UKaid).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
