Abstract
Objectives:
Symptoms of sore throat result from oropharyngeal inflammation, for which prostaglandin E2 is a key mediator. Flurbiprofen is a non-steroidal anti-inflammatory that provides sore throat relief. The preliminary objective of this study was to develop an in vitro model for assessing prostaglandin E2 stimulation by viral and bacterial triggers. The primary objective was to investigate the effect of diluted flurbiprofen-containing lozenges on prostaglandin E2 concentrations in stimulated cells.
Methods:
Prostaglandin E2 production was stimulated in three epithelial cell lines (A549, HEp2, and clonetics bronchial/tracheal epithelial) with influenza A virus (4.5 log10 tissue culture infectious dose50/mL), or bacterial lipopolysaccharide (10µ g/mL) and peptidoglycan (3µ g/mL) and incubated overnight. Prostaglandin E2 levels were assessed by enzyme-linked immunosorbent assay up to 24 h after stimulation. The effect of flurbiprofen 8.75 mg lozenges (diluted to 0.44 mg/mL) on PGE2 production in stimulated cells was assessed in parallel; prior to viral/LPS/PEP stimulation of cells, 300 μL of test product or control was added and incubated for 30 s, 2 and 5 min (and 10 min for bacterial trigger). Prostaglandin E2 levels were measured following stimulation.
Results:
Viral and lipopolysaccharide/peptidoglycan infection did not consistently stimulate HEp2 cells and bronchial/tracheal epithelial cells to produce prostaglandin E2. Influenza virus, and lipopolysaccharide/peptidoglycan stimulated high prostaglandin E2 concentrations in A549: mean prostaglandin E2 concentration 106.48 pg/mL with viral stimulation vs 33.82 pg/mL for uninfected cells; 83.84 pg/mL with lipopolysaccharide/peptidoglycan vs 71.96 pg/mL for uninfected cells. Flurbiprofen produced significant reductions in virus-stimulated prostaglandin E2 vs stimulated untreated cells at 2 min (p = 0.03). Flurbiprofen produced significant reductions in lipopolysaccharide/peptidoglycan-stimulated prostaglandin E2 concentrations from 30 s (p = 0.02), and at 2, 5 and 10 min (all p < 0.005) vs stimulated untreated cells.
Conclusions:
A549 cells provide a suitable model for assessment of prostaglandin E2 stimulation by viral and bacterial triggers. Diluted flurbiprofen-containing lozenges demonstrated rapid anti-inflammatory activity in viral- and lipopolysaccharide/peptidoglycan-stimulated A549 cells.
Keywords
Introduction
Sore throat is a common, self-limiting condition caused by inflammation of mucous membranes in the oropharynx, often due to upper respiratory tract infections (URTIs).1–5 Symptoms usually resolve within 7 days;1,5 however, pain, swelling and discomfort often negatively impact quality-of-life.6–8
Up to 80% of sore throat cases in adults are due to viral infections, such as the common cold (rhinovirus) or influenza, with bacterial infections such as group A β-haemolytic streptococcus causing around 10% of adult cases.1,5 Non-infectious causes may contribute to sore throat, including smoking, snoring, shouting, tracheal intubation, medications or illness, air pollutants, temperature and humidity. 9 Throat irritation is also present in individuals with chronic cough. 10 Prostaglandins such as prostaglandin E2 (PGE2) and other inflammatory mediators released in response to URTI or non-infectious stimulants trigger a complex inflammatory cascade that contributes to sore throat symptoms.2,9,11–14 PGE2 may impact activation of the vanilloid receptor (TRPV1) in sensory neurones, which is associated with pain and cough pathways. 15
Antibiotics are ineffective in most sore throat cases due to their viral aetiology and have a limited effect on symptoms; unless there is a bacterial infection they are useless and their overuse is dangerous.3,16,17 Incorrect prescribing of antibiotics contributes to the development of antibiotic resistance, which could be an impending storm where one of the foundations of current medical treatment is lost.16,17 As such, more personalised and selective antibiotic use is required.16,17 In addition, pain relief is one of the key priorities for patients seeking physician advice for sore throat.18,19
A sore throat is something that an individual should, in the first instance, approach their community pharmacist for advice about. A pro-symptomatic approach to the management of sore throat, with evidence-based treatments like flurbiprofen lozenges, can help reduce unnecessary visits to community doctors.
Flurbiprofen is a non-steroidal anti-inflammatory drug (NSAID), locally administered as lozenge or spray, which provides rapid and long-lasting sore throat relief.7,20–23 The anti-inflammatory mechanism of flurbiprofen is thought to involve inhibition of prostaglandin biosynthesis, 24 in common with other NSAIDs.25,26 Thus, there is clear rationale for utilising prostaglandin concentrations as a model to assess anti-inflammatory activity in sore throat.
The preliminary objective of this study was to develop an in vitro model for assessing PGE2 stimulation by influenza virus or bacterial lipopolysaccharide (LPS) and peptidoglycan (PEP), evaluating three cell lines (A549 and HEp2 cell lines, and a normal human bronchial epithelial cell line (NHBE)). The primary objective was to assess the effects of commercially available flurbiprofen-containing lozenges on PGE2 concentrations in stimulated (infected) cells.
Materials and methods
This was an in vitro study utilising three cell lines: A549 adenocarcinomic lung-derived human epithelial cell line (CCL-185 lot# 3449902 (ATCC, Teddington, UK)), HEp2 cell line (CCL-23 lot# 3548462 (ATCC) and secondary culture of clonetics bronchial/tracheal epithelial system (CC-2540 lot# 2F1186 (ATCC) The A549 cell line provided consistent results with respect to PGE2 stimulation and is discussed in detail.
Test product and control
The test product was Strefen honey and lemon lozenges (Reckitt-Benckiser Ltd., Slough, UK). Each lozenge contained 8.75 mg of flurbiprofen, demonstrated to be the lowest effective dose providing symptomatic relief of sore throat in dose-ranging studies. Each lozenge was dissolved in 10 mL artificial saliva (mucin, type III: partially purified from porcine stomach (M1778, Sigma-Aldrich, Gillingham, UK), potassium dihydrogen orthophosphate (P/4800/50, Fisher Scientific, Loughborough, UK) and sodium hydrogen carbonate (S/4240/53, Fisher Scientific, Loughborough, UK) in phosphate-buffered saline (PBS)) in a 50 mL tube and placed on a shaker for approximately 45 min at room temperature. Addition of 10 mL cell culture media resulted in a flurbiprofen concentration of 0.44 mg/mL (total volume 20 mL). The positive control was citrate buffer at pH 3.5 (hVivo Ltd., London, UK).
Cell line incubation
A549 cells were plated at 1 x 104 cells/300 μL in each well within a 24-well plate, and incubated overnight at 37°C and 5% CO2. Media was removed slowly by aspiration and 300 μL test product or control was added. Test product and controls were incubated for varying contact times: 30 s, 2 min and 5 min for viral triggers; 30 s, 2 min, 5 min and 10 min for bacterial triggers.
Cell line stimulation (infection)
After the required incubation time, the test product or control was removed and the cell lines were stimulated with the influenza virus ((A/Sydney/5/97 (H3 N2)); 4.5 log10 tissue culture infectious dose (TCID)50/mL (hVivo Ltd.)) or LPS (10 μg/mL; L6529 derived from Escherichia coli (Sigma-Aldrich, Gillingham, UK)) and PEP (3 μg/mL; 77140 derived from Staphylococcus aureus (Sigma-Aldrich, Gillingham, UK)) to simulate the effects of viruses and bacteria, respectively. Cells were then incubated overnight at 37°C and 5% CO2.
PGE2 assessment (primary endpoint)
PGE2 concentrations were assessed by enzyme-linked immunosorbent assay (ELISA) using a prostaglandin E metabolite enzyme immunoassay kit (Item No. 514531, Cayman Chemical, 27 Cambridge, UK), according to the manufacturer’s methodology. Developed plates were read on a TECAN Sunrise ELISA plate reader (RV5-018; Tecan Group Ltd., Männedorf, Switzerland) at a wavelength of 405 nM.
Statistical analyses
The hypothesis tested was that Strefen honey and lemon lozenges would have a quantifiable effect on concentrations of PGE2 stimulated by viral or bacterial triggers, compared with stimulated control cells. Duplicate values were obtained for each measurement and analysed used a pairwise comparison performed with two-sample t-tests. Results are presented as p-values for the pairwise comparison between the test product and stimulated controls.
Results
It was observed that viral and bacterial LPS/PEP infection did not consistently stimulate HEp2 and NHBE cells to produce PGE2. A549 cells gave the most reproducible results and data in this cell line is reported below.
Viral infection (stimulation) of A549 cells
After infection with influenza A virus, PGE2 concentrations in A549 cells reached 106.48 pg/mL (95% confidence interval (CI): 76.92–136.04), compared with 33.82 pg/mL (95% CI: 33.04–34.59) for uninfected cells only (Figure 1).

Concentrations of PGE2 (pg/mL; 95% CI)a in A549 cells after infection with influenza virus or bacterial LPS/PEP compared with controls (unstimulated cells only or citrate buffer).
Bacterial LPS/PEP stimulation of A549 cells
Infection of the A549 cell line with bacterial LPS and PEP stimulated prostaglandin synthesis, with an average PGE2 concentration of 83.84 pg/mL (95% CI: 81.46–86.22), compared with 71.96 pg/mL (95% CI: 63.59–80.33) for uninfected cells (Figure 1).
Inhibition of viral and bacterial LPS/PEP stimulation of A549 cells by flurbiprofen
Flurbiprofen inhibited the synthesis of PGE2 in influenza A virus-infected A549 cells (Figure 2). Significant inhibition compared with infected control cells was observed at 2 min (p = 0.03), representing an 80% reduction compared with PGE2 concentrations in influenza A virus-infected control cells at this timepoint. Inhibition of PGE2 synthesis was also observed with incubation of bacterial LPS/PEP-infected A549 cells with flurbiprofen. Significant inhibition compared with LPS/PEP-infected control cells was seen from 30 s’ incubation time (p = 0.02; Figure 2). Significant inhibition of PGE2 concentrations continued to be observed at 2 min (p = 0.002), 5 min (p = 0.004), and 10 min (p = 0.003) incubation times.
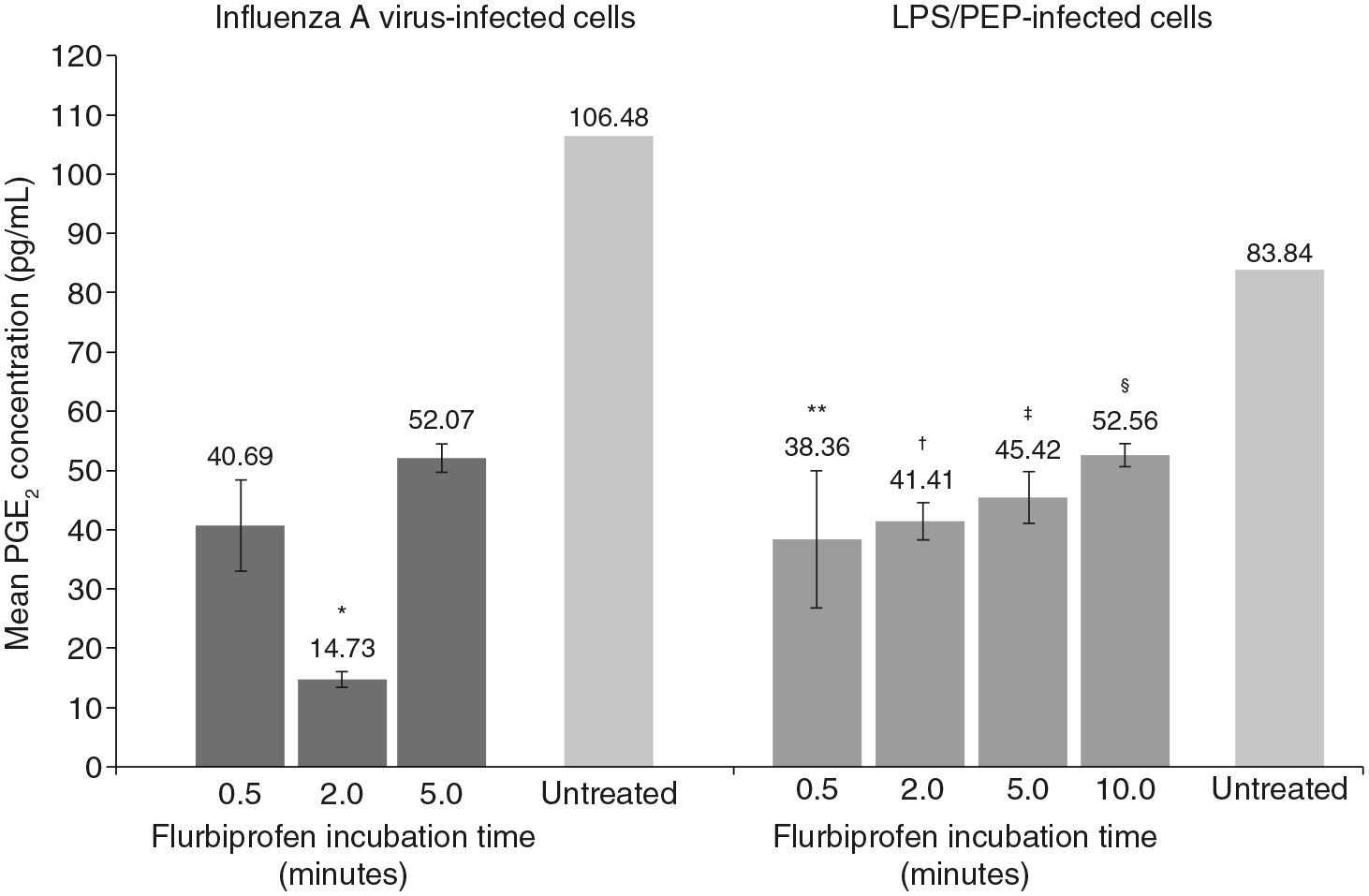
PGE2 concentrations (pg/mL; 95% CI)a for influenza virus- and LPS/PEP-infected A549 cells incubated with diluted flurbiprofen lozenges at 0.5-, 2-, 5- and 10-minb timepoints.
Discussion
Prostaglandins such as PGE2 play a key role in inflammation that causes the discomfort of sore throat.2,9,12–14 This study demonstrated that human lung-derived epithelial A549 cells can be stimulated to increase PGE2 production by influenza virus or bacterial LPS and PEP, providing a valuable model to assess the activity of pharmaceuticals that target the PGE2 pathway. Lozenges containing the NSAID flurbiprofen, at the commercially available dose of 8.75 mg, demonstrated rapid suppression of viral- and bacterial LPS/PEP-stimulated PGE2 concentrations in A549 cells; significantly lower PGE2 concentrations were observed for both viral and bacterial LPS/PEP-infected cells versus controls.
Influenza is associated with elevations in PGE2,28,29 while LPS and PEP are major components of the cell walls of Gram-negative and Gram-positive bacteria, respectively, and are known to trigger a variety of inflammatory reactions, including PGE2 production.12,13,30–33 The findings reported here indicate that flurbiprofen targets and inhibits PGE2, providing anti-inflammatory activity in response to specific viral and bacterial triggers.
The use of a human lung-derived epithelial cell line, albeit in vitro, gave the authors confidence in the data. The behaviour of A549 cells is similar to normal epithelial cells, and they are widely used as an alternative model for studying human epithelial cell behaviour; they have demonstrated release of inflammatory cytokines upon stimulation, 34 and replication of the influenza virus. 35 PGE2 release is different in other epithelial cells and the behaviour of A549 in response to stimulation (such as by influenza A virus and bacterial LPS/PEP) is an interesting and specific feature of this human-lung-derived epithelial cell line. Dissolving flurbiprofen lozenges in artificial saliva replicated the expected time for a lozenge to dissolve in the mouth, and the anticipated concentration generated in saliva during the course of sucking a lozenge. 36 Our findings indicated that flurbiprofen provides anti-inflammatory activity within the anticipated time frame for a lozenge to dissolve and using similar concentrations as would be achieved in the mouth.
Although used in previous studies investigating epithelial inflammatory responses, the A549 cell line is only a model and cannot fully represent pharyngeal epithelial cells. In addition, an in vitro model can never fully replicate the complexity of the clinical setting, where an individual’s underlying health status may affect the pathology of sore throat. In order to select the most appropriate cell lines for the study, consideration was given to: viral replication (replication does not happen in all cells), cell-line survival (maintaining non-continuous cell lines is challenging and would have introduced experimental error), and generation of inflammatory mediators (not inducible in all cell lines). The two other cell lines evaluated before A549 cells were selected (HEp2 and an NHBE cell line) produced inconsistent effects with respect to PGE2 stimulation and were therefore considered unsuitable for this study.
In addition, this small-scale study utilised few replicate assessments and timepoints, which may limit the conclusions that can be drawn. Natural differences in cell reactions were observed, with PGE2 concentrations in the unstimulated cells varying in the viral and bacterial tests. A further limitation is the focus on a limited number of infectious triggers. A range of organisms have been implicated as causes of URTIs and further studies are needed to reflect the breadth of potential pathogens.1,3,4,37–39 Furthermore, this study only assessed the effects of viral and bacterial triggers and test product on PGE2 synthesis, one element of the inflammatory process. Further studies should address their effects on other inflammatory mediators and pathways involved in the pathogenesis of sore throat.
These in vitro findings are in line with clinical experience with flurbiprofen lozenges, which have demonstrated rapid and meaningful symptomatic relief in patients with sore throat, whether the cause be viral, bacterial or other.21–23 Local/topical administration of flurbiprofen (spray or lozenge) enables direct activity and avoids systemic adverse events associated with oral NSAID use.40,41
Conclusion
This study demonstrated that it is possible to stimulate PGE2 in vitro, using a human respiratory epithelial-like cell line, with relevant viral and bacterial triggers. This provides a valuable model to assess the anti-inflammatory impact of pharmaceutical agents that may be targeting the PGE2 inflammatory pathway. Flurbiprofen lozenges provide rapid relief of sore throat symptoms in the clinical setting. The results reported here indicate that this is likely due to anti-inflammatory effects based on a clear mechanism of action on elevated PGE2 concentrations due to viral or bacterial infection.
Footnotes
Acknowledgements
The authors would like to acknowledge the contributions of Eseberuo Sefia Sedenu, Shobana Balasingam and Mike Goulder to the conduct of this study. Medical writing assistance was provided by Joanna Hulme of Elements Communications Ltd., Westerham, UK and was funded by Reckitt Benckiser Healthcare Ltd., UK.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Rob Lambkin-Williams is currently a director of VirologyConsult Ltd., and was an employee of hVivo Services Ltd. at the time of the study. Alex Mann is an employee of hVivo Services Ltd. Adrian Shephard is an employee of Reckitt Benckiser Healthcare Ltd.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Reckitt Benckiser Healthcare Ltd., UK.
