Abstract
Introduction:
Soil-transmitted helminth infections and malnutrition are major health problems of school-age children in developing countries. Malnutrition and soil-transmitted helminth infections often co-exist with synergetic consequences.
Objective:
The aim of this study was to determine the prevalence and intensity of soil-transmitted helminths and its association with nutritional status of school-age children.
Methods:
School-based cross-sectional study was carried out from April to May 2014 among 404 elementary school-age children in Jimma Town, Southwest Ethiopia. Data on background characteristics were collected using structured interviewer administered questionnaire. Anthropometric measurements were taken according to World Health Organization standard. Fresh single stool sample was collected from each study participant and examined using direct wet mount and McMaster techniques. Anthropometric indices were generated using WHO AnthroPlus software. Multivariable logistic regression models were fitted to isolate independent predictors of intestinal parasitic infection and nutritional status using STATA-MP software. All tests were two-sided and P < 0.05 was used to declare statistical significance.
Results:
The overall prevalence of intestinal parasites and soil-transmitted helminths were 68.6% (n = 277) and 55.0% (n = 222), respectively. A total of eight species of intestinal parasites were identified in this study, Trichuris trichiura being the most common parasite identified followed by Ascaris lumbricoides. Study participants who had a habit of open defecation were two times more likely to be infected with soil-transmitted helminths (adjusted odds ratio = 1.9, 95% confidence interval: 1.0–3.4). The overall prevalence of stunting and thinness were 21.0% (n = 85) and 6.9% (n = 28), respectively. The odds of stunting was significantly high (adjusted odds ratio = 4.0, 95% confidence interval: 1.7–9.7) among children who had fathers working as daily labourers and children with personal dietary diversity score of ⩽3 (adjusted odds ratio = 3.5, 95% confidence interval: 1.5–8.0). T. trichiura infection (adjusted odds ratio = 9.4, 95% confidence interval: 2.0–44.8) was identified as an independent predictor of stunting among school-age children.
Conclusion:
Both the prevalence of soil-transmitted helminths and stunting are high among school-age children in Jimma Town. Although there was no statistically significant association between the STHs, in general, T. trichiura was reported as predictor of stunting. The results imply the need for strengthening strategies for reduction of parasitic infection to curb the pervasively high prevalence of stunting.
Introduction
Soil-transmitted helminths (STHs) are among the neglected tropical diseases (NTDs) affecting more than 2 billion people worldwide. The reported global Disability-Adjusted Life Years (DALYs) of the STHs is estimated to be as high as 2 million in 2017.1,2 Helminthic parasites categorized as the STHs include Ascaris lumbricoides (A. lumbricoides), Trichuris trichiura (T. trichiura) and the hookworms (Ancylostoma duodenale (A. duodenale) and Necator americanus (N. americanus)). As faecal contamination of soil is mandatory for the life cycle of STHs, the burden of their infection is higher in developing countries where sanitary facilities are scarce and hygienic practices are poor.1,3 The majority of the global prevalence of STH occurs in sub-Saharan African countries, Americas and Asia. Ethiopia is among the countries having high burden of the STHs with estimated 81 million people living in endemic areas.4–6 There is disparity in the transmission of the disease among populations related to differences in demographics, socio-economic and environmental factors. School-aged children (SAC) are population subgroups with high risk of STH infections owing to their playing habits and frequent contact with soil.6,7 Studies conducted in Jimma Town showed a high prevalence of STH infections among SAC.8,9
Soil-transmitted helminthic infections may disrupt the health, nutritional status and cognitive development of the children. Especially, poly-parasitism and high intensity of helminths could lead to morbidity and malnutrition which could end in mortality. Malnutrition of the human hosts may be exacerbated by helminths due to their physical barrier and/or competition for food.6,10,11 Anaemia is more related to intestinal blood loss and insufficient micronutrient absorption. 12 The overall effect may alter cognitive development of the children including their memory, reaction time, learning and innate intelligence, through the exact mechanism which is not known yet. 13 Generally, epidemiological studies among SAC showed a direct effect of STH infections on school performance and school absenteeism.14,15
On the contrary, childhood malnutrition remains to be the salient public health problems making major contribution to the global disease burden. A global report in 2018 indicated that globally, about 150 million children were stunted, 50 million wasted and 38 million overweight. 16 Sub-Saharan Africa is the home of stunting with the proportion of SAC affected being as high as 57% in Burundi and 22% in Ethiopia. The main causes for such a high-level malnutrition are identified to be food insecurity, inadequate child care and poor health systems.17–19 Similar to STHs, malnutrition also contributes to low school enrolment, school absenteeism and poor school performance in developing countries. In Jimma, Wolde and Belachew 20 reported a prevalence stunting of 17% among SAC was predisposing factor for low school performance. 14
In developing countries, both malnutrition and STH infections often co-exist. The synergetic effect of both conditions considered as top global health problems is exacerbating the general poor health status of SAC influencing their wellbeing across generations.21–24 The cause and effect relationship of malnutrition and STH infections is reciprocal in which malnutrition may be a predisposing factor for STH infections and several communicable and non-communicable diseases as it affects physical fitness of the host including their immune status. On the contrary, STH infections may result in malnutrition by consuming nutrients from the host and/or adversely affecting nutrient absorption.18,22,25–27 Stunting and STH infections are major problems of SAC of which developing nations share the great number.4,6 Studies in different parts of Ethiopia often reported significant relationship of these problems.28,29 However, some studies documented contradicting findings on the association of malnutrition and STH infections among SAC.30–32 The variation may depend on socio-demographics and disparities in level of disease burden in the different geographic localities of the country. However, there was no study on association between malnutrition and STH infections among SAC is in the study area. Therefore, the aim of this study was to determine the prevalence of STH infections and malnutrition and their association among primary school children in Jimma Town.
Methods
Study setting
School-based cross-sectional study was conducted among SAC in four selected public elementary schools in Jimma Town from April to May 2014. Jimma Town is located 352 km southwest of Addis Ababa. In 2013–2014 academic year, a total of 19,449 SAC (9047 males and 10,402 females) were enrolled in 14 public elementary schools of the Town. These schools were grouped by Jimma Town Office of Education into four groups based on their geographical location from the centre of the Town. Four elementary schools were selected randomly from each group and included in this study (Figure 1).

(a) Map of Jimma zone and (b) map of Jimma city indicating specific study sites (four schools) within the Jimma Town, Capital of Jimma Zone.
Sample size and sampling techniques
The sample size required for this study was calculated using single population proportion formula, with the following assumptions: previous prevalence report of STH infections among elementary schools in the study area of 53.5%, 8 95% confidence level, 5% margin of error and 10% non-response rate. The final sample size calculated was 420. Study participants were selected using systematic sampling technique from the four schools using roster as sampling frame based on the total number of students enrolled in the academic year 2013–2014. The total sample size was proportionally allocated to the number of children in each selected school; accordingly, 69, 89, 147 and 118 study participants were selected from Jiren-No. 2, Ginjo, Hermata and Mendera elementary schools, respectively (Figure 1).
Data collection
Socio-demographic data
Semi-structured questionnaire was used to collect data on demographic characteristics of the children, risk factors of STH infections, malnutrition and dietary diversity. These data were obtained from parents/guardians of the children. The interview was performed by two trained health extension workers who were conversant with the local languages. The questionnaire was first prepared in English and then translated to the local languages (Afan Oromo and Amharic) and pilot-tested in 5% of the sample sizes in non-selected school before the actual data collection.
Stool sample collection and processing
Each study participant was provided with clean, labelled stool cup and instructed to bring about a thumb-sized stool sample of their own. The stool samples were collected at each school, and direct wet mount examination was performed immediately at the site. The remaining samples were then transported to Jimma University STH laboratory for further analysis. McMaster technique was used for the detection and quantifying eggs of the STHs as follows. After homogenizing each stool sample using wooden spatula, 2 g of stool was suspended in 30 mL of saturated salt solution. The faecal suspension was poured three times through a wire mesh to remove large debris. Then, two chambers of a McMaster slide were filled with this suspension. Both chambers were examined under a light microscope using a 100× magnification; and faecal egg count (FEC) and eggs per gramme of stool (EPG) for each helminth species were obtained by multiplying the total number of eggs by 50. 33 Intensity of the STH infections was classified as light, moderate and heavy based on the number of the EPG of stool according to World Health Organization (WHO) classification criteria. 34
Anthropometric measurements
Trained clinical nurses measured the height and weight of the children and assessed for the presence of oedema. Weight measurements were taken using portable, calibrated digital weight scale (Detecto, Webb City, MO, USA) with light clothing and shoe removed, to the nearest 0.1 kg. The height of each study participant was measured in the recumbent position to the nearest 0.1 cm using height-measuring upright wooden base and movable headpiece board supplied by United Nations International Children’s Emergency Fund (UNICEF). 35 The weight and height of the study participants were measured twice by independent nurses to reduce enter-individual error and the mean measurement value was recorded. WHO AnthroPlus 2009 (v 1.0.4) was used to calculate Z-score of height for age (HAZ) and body mass index for age Z-score (BAZ). Children with Z-score < −2SD were classified as stunted (HAZ < −2SD) and wasted (BAZ < −2SD).
Dietary diversity score
Dietary consumption 24 h prior to the survey was assessed using questionnaire developed based on Food and Agriculture Organization (FAO) guidelines. 21 Consumed foods were classified into 10 food groups. The food groups included: (1) starchy staples, (2) dark green leafy vegetables, (3) other vitamin-A-rich fruits and vegetables, (4) other fruits and vegetables, (5) meat and fish, (6) eggs, (7) legumes, nuts and seeds, (8) milk and milk products, (9) oils and fats and (10) other foods and sweets.36,37
Consumption from each food group was reported, and one point was given if the food is consumed at least once the last 24 h and zero point if not consumed within that period. Therefore, the dietary diversity score (DDS) ranged from 0 to 10, and children with DDS of ⩽3 were classified as poor diversity whereas children with DDS 4–6 and >6 were classified as middle and high diversity. 38
Data processing and analysis
The data were entered into excel spread sheet, cleaned and exported to the STATA-MP (College Station, TX, USA) software for analysis. Socio-demographic characteristics of the study participants were summarized using descriptive statistics. Bivariate and multivariable logistic regressions were used to determine independent predictors of STH infections and malnutrition. Explanatory variables with P value <0.25 in bivariate analysis were candidates for multivariable analysis. Odds ratios and the corresponding 95% confidence intervals (CIs) were used to show the strength of the association. P value less than 0.05 was considered as statistically significant.
Results
Socio-demographic characteristics of the study participants
A total of 404 school children were included in this study, giving a response rate of 96.2%. More than half of the study participants (53.7%, n = 217) were female. The mean (±SD) age of the children was 9.8 (±2.16) years, ranging from 6 to 14 years. Most of the children (65.8%, n = 266) lived in households with family size of ⩾5 (Table 1).
Socio-demographic characteristics of the school children, Jimma Town, Southwest Ethiopia.

Prevalence of STH infections and associated factors
The overall prevalence of STHs was 55.0% (n = 222) of which T. trichiura was the most common parasite, 34.9% (n = 141) identified among the SAC (Figure 2). A. lumbricoides and the hookworms were detected in 28.5% (n = 115) and 11.4% (n = 46) of the children, respectively. Eight species of intestinal parasites were identified and 68.6% (n = 277) of the study subjects were infected with these intestinal parasites.

Frequency of intestinal parasites identified among school children in Jimma Town, Southwest Ethiopia.
The prevalence of the STHs is relatively higher among age group of 11–14 years old, 92 (56.4%), as compared to 6–10 years old, 130 (53.9%), although not statistically significant. The number of infection is also higher among SAC with dirt under their finger nails, 54 (63.5%) than children without observed dirt (trimmed nails), 168 (52.7%). Children who had habit of open-field defecation had significantly higher risk of acquiring STH infections (adjusted odds ratio (AOR) = 1.9, 95% CI: 1.0–3.4; Table 2) as compared to children using toilet usually. Similarly, children who reported to walk barefoot had higher risk of being infected with the hookworms (crude odds ratio (COR) = 2.2, 95% CI: 1.1–4.3) than children who wore shoes.
Factors associated with STH infection among school children in Jimma Town, Southwest Ethiopia.

COR: crude odds ratio; AOR: adjusted odds ratio; CI: confidence interval; STHs: soil-transmitted helminths.
Parameters adjusted for family size, fingernail cutting, paternal education, maternal education and sex of the child.
Intensity of STH infections
The intensity of each species of STHs infection is presented in Figure 3. Majority of the children had light infections of each of the STHs. In this study, there were no heavy infections caused by the hookworms.

Intensity of STH infections among school children in Jimma Town, Southwest Ethiopia.
Nutritional status and associated factors
The mean DDS of the school children was 5 (±1), ranging from 2 to 8. Of all the children, 31.7% (n = 128) consumed <3 food groups indicating low dietary diversity. Except one of the study subject, all of the study participants consumed starchy staples within 24 h prior to the survey. Other highly consumed food groups include fruits and vegetables (99.5%); oil and fat (98.5%); sweets and other foods (61.4%) and legumes, nuts and seeds (60.1%). Relatively less commonly consumed food items were Vitamin-A-rich fruits and vegetables (42.8%), meat and fish (20.8%), milk and milk products (10.4%), eggs (6.9%) and dark green vegetables (4.0%).
The overall prevalence of stunting and wasting were 21.0% (n = 85) and 6.9% (n = 28), respectively. The prevalence of stunting among study participants who had DDS ⩽ 3 and >3 was 46.2% (n = 12) and 19.3% (n = 73), respectively.
Stunting was significantly associated with DDS (AOR = 3.7, 95% CI: 1.6–8.7) where children with DDS of ⩽3 were four times more stunted than children with DDS of >3. Fathers’ occupation also had significant association with stunting where daily labourers (AOR = 3.9, 95% CI: 1.9–9.6) and employed (AOR = 2.9, 95% CI: 1.2–6.8) fathers had stunted children as compared to self-employed fathers (Table 3).
Factors associated with stunting and wasting among school children in Jimma Town, Southwest Ethiopia.
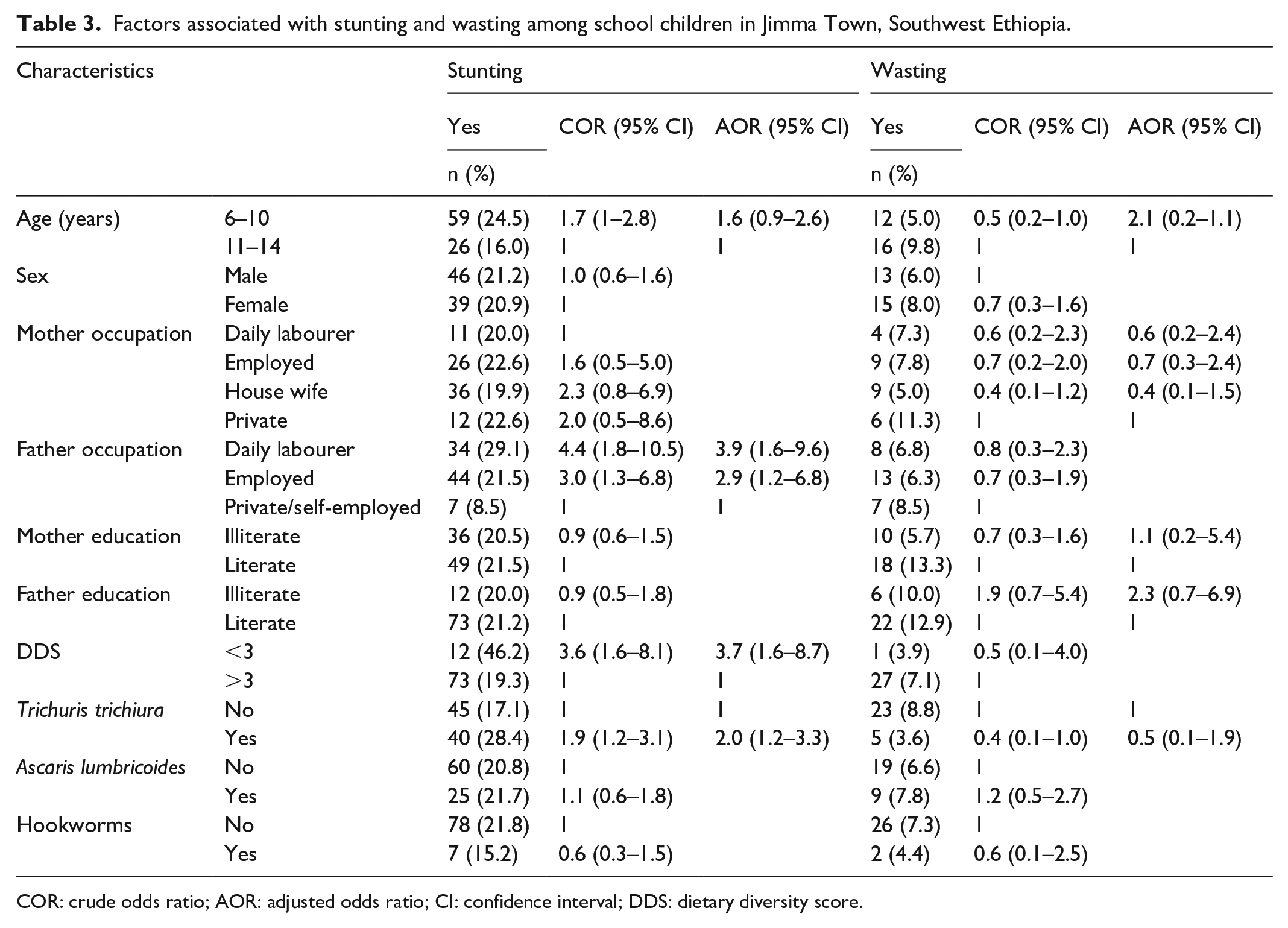
COR: crude odds ratio; AOR: adjusted odds ratio; CI: confidence interval; DDS: dietary diversity score.
The Pearson’s correlation between the prevalence of stunting and DDS shows moderate negative correlation (R = −0.56), which means there is high prevalence of stunting for low value of DDS (Figure 4).
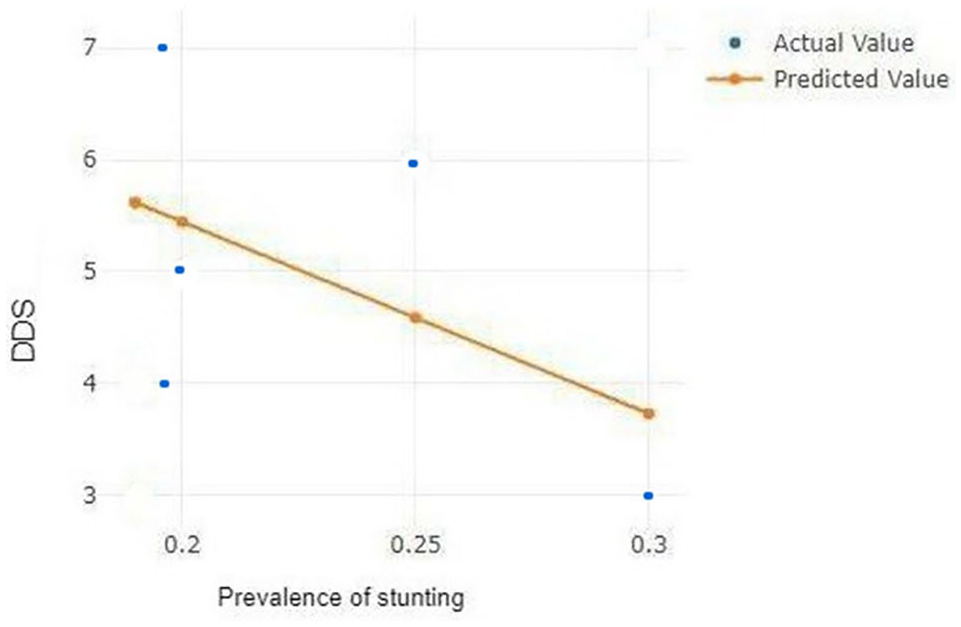
The Pearson’s correlation between the prevalence of stunting and DDS among school children in Jimma.
STH infections and nutritional status
Multivariable analysis of each of the STH infections and nutritional status showed that T. trichiura infection was the main predictors of stunting among the school children (AOR = 2.0, 95% CI: 1.2–3.3). None of the STH infections were associated with wasting (Table 3).
Discussion
Intestinal parasites, in general, and the STHs, in particular, are common public health concern in developing countries, SAC being at higher risk. 6 Aimed at determining prevalence of the STHs and assessing its association with nutritional status among primary SAC, this study revealed that more than half of the children had at least one species of the STHs. Earlier studies also reported high prevalence of the STHs in Jimma Town.8,9 Open-field defecation habit was significant predictor of STH infections where the prevalence of STHs was significantly higher among SAC who reported open-field defecation. Similarly, studies also reported open-field defecation as a risk factor for the acquisition of STH infections.9,39 Thus, the STHs pose impact on SAC health by triggering vital micronutrient deficiency and/or causing pathophysiological disorders in the study area.12,40 There was also significant association between T. trichiura infections and stunting among the study participants. Due to chronic inflammation that these parasites lead to, the gut suffers from environmental enteropathy leading to leaky gut and poor absorption of nutrients and vaccines.41,42 Nutritional interventions need to consider the importance of environmental interventions including water, sanitation and hygiene. However, contradicting study results on the association of STH infections and under-nutrition were reported among SAC in Ethiopia.31,32 This may be due to the geographical distribution of individual STH species and different level of prevalence or other major factors that may cause under-nutrition in different parts of the country. 18
Under-nutrition, on the contrary, is among major health problems of SAC in developing countries. 18 Stunting, one indicator of under-nutrition, is a significant public health problem among SAC in Ethiopia, which may affect their physical and/or mental development.19,30,43 The prevalence of stunting in this study was higher than a previously conducted study among SAC in Jimma Town 44 but almost similar with the national figure. 19 Father’s occupation was significantly associated with stunting in the present study finding. Although contradicting reports exist on the association of parents’ occupation with stunting,30,43,45 our finding might support the fact that, the major share of family income is generated by men in developing countries including the current study area, 46 which likely affect indirectly nutritional status of the children. Moreover, consumption of variety of foods has great role on physical and mental development in less than 5 years, pre-school and school children. 47 Our finding also illustrated that SAC with low DDS were more likely stunted as compared to moderate and high. The significance of dietary diversity in under-nutrition has also been reported previously. 45 Although other STH species (A. lumbricoides and hookworms) did not show significant association with stunting, T. trichiura had significant association with stunting. While the impact of STHs on nutritional status of children has long been established, 48 T. trichiura in particular may cause gastrointestinal bleeding leading to life-threatening anaemia. 49
Generally, this study indicates statistically significant association of T. trichiura infections and stunting which calls for consideration of integrated control mechanism. The acquisition of STHs also associated with open-field defecation and thus, enhancing sanitary disposal of human excreta should be practised at school and/or household level. Under-nutrition particularly, stunting is associated with DDS of the SAC and family household income sources. These two risk factors also have direct relationship in which improvement of family income may improve the household dietary diversity.
The limitations of this study are as follows: (1) micronutrient intake and red blood cell levels of the children were not determined and (2) there could be seasonal variations in the level of under-nutrition that this study did not capture due to its cross-sectional nature. Thus, it is our recommendation that the future similar studies shall consider the above limitations into account.
Conclusion
Both the prevalence of STH infections and stunting are high among SAC in Jimma Town. Stunting was significantly associated with T. trichiura infections, low DDS and father’s occupation. Open-field defecation habit of the SAC is the only predictor of STH infections in the study. The results imply the need for strengthening integrated strategies for reduction of parasitic infection to curb the pervasively high prevalence of stunting.
Supplemental Material
English_Version_of_the_Questionnaires_Supplementary_file_S1 – Supplemental material for Soil-transmitted helminth infections and nutritional status of school children in government elementary schools in Jimma Town, Southwestern Ethiopia
Supplemental material, English_Version_of_the_Questionnaires_Supplementary_file_S1 for Soil-transmitted helminth infections and nutritional status of school children in government elementary schools in Jimma Town, Southwestern Ethiopia by Zeleke Mekonnen, Derartu Hassen, Serkadis Debalke, Abebaw Tiruneh, Yaregal Asres, Legese Chelkeba, Endalew Zemene and Tefera Belachew in SAGE Open Medicine
Footnotes
Acknowledgements
The authors thank the study participants, data collectors and each school staff of Jimma Town Health Department and Education Office. They would also like to thank Jimma University for support in materials and postgraduate fund.
Author contributions
Z.M., D.H., S.D. and E.Z. were involved in design of the study and coordinated data collection. Y.A., L.C. and T.F. supervised the field and laboratory work. D.H., A.T. and T.B. analyzed the data. Z.M. and A.T. drafted the manuscript. All authors revised subsequent drafts, read and approved the final revised manuscript.
Availability of data and materials
All datasets supporting this study are included in the article. We have also uploaded the English version of the questionnaire(s) used in this study as a separate supplementary file.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval and consent to participate
Ethical clearance was obtained from Ethical Review Board of Jimma University College of Health Sciences. Permission was obtained from Jimma Town Office of Education and each school administration. The aim of the study was explained to the study participants and their parents/guardians. Written informed consent was obtained from parents/guardians of the children. Confidentiality of the data was maintained. Children who were infected with intestinal parasite(s) were treated according to the national guidelines, and those with undernutrition were referred to Jimma University Specialized Hospital for further investigation. Ethical approval for this study was obtained from the Ethical Review Board of Jimma University College of Health Sciences (approval number: RPGC/03/436/2014).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Jimma University Postgraduate Research Office provided small financial support for this study, but funders had no role or influence in the scientific output.
Informed consent
Written informed consent was obtained from parents/guardians of the children and assent/verbal informed consent was obtained from all subjects before the study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
