Abstract
Tissues usually super compensate during the period that follow physical exercise. Although this is widely accepted for muscle and glycogen, the compensatory effect is not usually applied to fat tissues. Notwithstanding, evidence for this has been present since the 1970s when it was first suggested that the increased lipogenic activity in response to training might be an adaptation that enables to restore an energy reserve that can be used in times of need. In this context, the present review aimed to summarize information about the effect of detraining on fat metabolism and the physiological responses associated with fat regain. A systematic search on PubMed and Scielo was performed using “training cessation,” “detraining,” “exercise detraining,” and “exercise cessation” combined with “fat tissue,” “adipose tissue,” “adipose metabolism,” and “fat metabolism,” as descriptors. From 377 results, 25 were included in this review, 12 humans and 13 rodents, resulting in a sample of 6772 humans and 613 animals. The analysis provided evidence for fat super compensation, as well as differences in humans and rodents, among different protocols and possible mechanisms for fat gain after exercise cessation. In summary, exercise cessation appears to increase the ability of the adipose tissue to store energy. However, caution should be taken, especially regarding conclusions based on investigations on humans, considering the multiple factors that could affect fat metabolism.
Keywords
Introduction
It is recognized that different biological stressors can induce several positive and negative responses. 1 These responses are derived from a physiological mechanism called the General Adaptation Syndrome (GAS), defined as the sum of all nonspecific systemic reactions that occur in response to stress, 2 contributing to adaptation. 3 In general, the stressful stimuli induce an alarm period and, if adequate conditions are provided, a compensatory effect occurs and a higher level of resilience is generated. 4 Physical exercise is among the possible stressors, as it induces different responses that trigger multiple adjustments. 5
These compensatory effects have been described in response to exercise in specific tissues such as bones 6 and muscles. 7 The same pattern appears to occur in different energetic substrates such as glycogen, 8 phosphocreatine, and muscle adenosine triphosphate (ATP). 9 However, this topic has been poorly discussed regarding adipose tissue. It is known that moderate-intensity continuous exercise (MICE) results in increased fat oxidation by adipose tissue.10,11 This stimulus acutely stresses fat depots, causing triglyceride breakdown and increasing free fatty acid availability into circulation to subsequent oxidation. 11 Although this might lead to a reduction of adiposity and favorable changes in the lipid profile, 12 it is possible that adipose tissue undergoes a compensatory effect.13–15
The compensatory effect on fat-related outcomes has been investigated in endurance athletes who showed elevated intramuscular lipid storages 16 which is called the “endurance paradox.” On the other hand, these effects should also be expected on adipose tissue, especially after exercise cessation and might explain fat regain. This process might be related to the increases in tissue-specific insulin sensitivity 17 or glycerol-3-phosphate esterification rate. 18 Those concepts have been previously presented as hypotheses; 19 however, understanding those responses is highly relevant, as it might help in reviewing the theoretical concepts for exercise prescription.
Moreover, when analyzing at a longer period, exercise cessation also leads to a detraining effect, with the reversal of many of its benefits.20–22 For example, an increase in the respiratory exchange ratio is expected after short- (<4 weeks) and long-term (>4 weeks) detraining periods, which means a shift in substrate utilization from lipids to carbohydrates. 20 This detraining effect might be also evident in adipose tissue, leading to an increased fat accumulation due to changes in fat metabolism.
In this context, the main objective of this review is to summarize information about the effects of detraining on adipose tissue and physiological responses related to fat regain. The article also discusses differences between humans and rodents, different protocols, and possible mechanisms for fat gain after exercise cessation.
Methods
This systematic review searched original articles, conducted with rodents or humans, preferably randomized controlled trials (RCT), and clinical trials (CT), quasi-experimental designs, and prospective studies when humans were considered. The interventions had to include physical exercise followed by a detraining period with reevaluation in this final period.
Primary outcomes were fat cell mass, diameter, and number. Secondary outcomes were hormonal and enzymatic alterations related to adipose tissue metabolism, total body mass, lipid and glycemic profile, insulin sensibility, and transcriptional factors related to adipose super compensation.
The search was conducted independently by two of the authors (VSC and LDC) and compared against each other. If a discordance was found, a third author was consulted (FBDV). Pubmed and Scielo databases were used by applying the following descriptors: “training cessation,” “detraining,” “exercise detraining,” and “exercise cessation” combined with “fat tissue,” “adipose tissue,” “adipose metabolism,” and “fat metabolism.”
Regarding eligibility criteria, to be included, studies should (i) be conducted with humans or rodents; (ii) have both training and detraining periods; (iii) have described training and detraining characteristics, considering at least weekly training volume or its reduction (i.e., km/wk, session/wk. . .); (iv) have objectively measured any of the primary or secondary outcomes. Studies without one of the periods (training and detraining) or without direct comparison between periods were excluded.
The initial search resulted in 377 articles selected for title and abstract reading, as described in Figure 1. After adequacy analysis for the minimum criteria of pre-selection, the articles were evaluated according to Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA Statement). After that, 27 full articles were read and 25 of them were included in the present systematic review. The Consensus on Exercise Reporting Template 23 was applied to assess the quality of description of the exercise protocols, as described in the supplementary file.
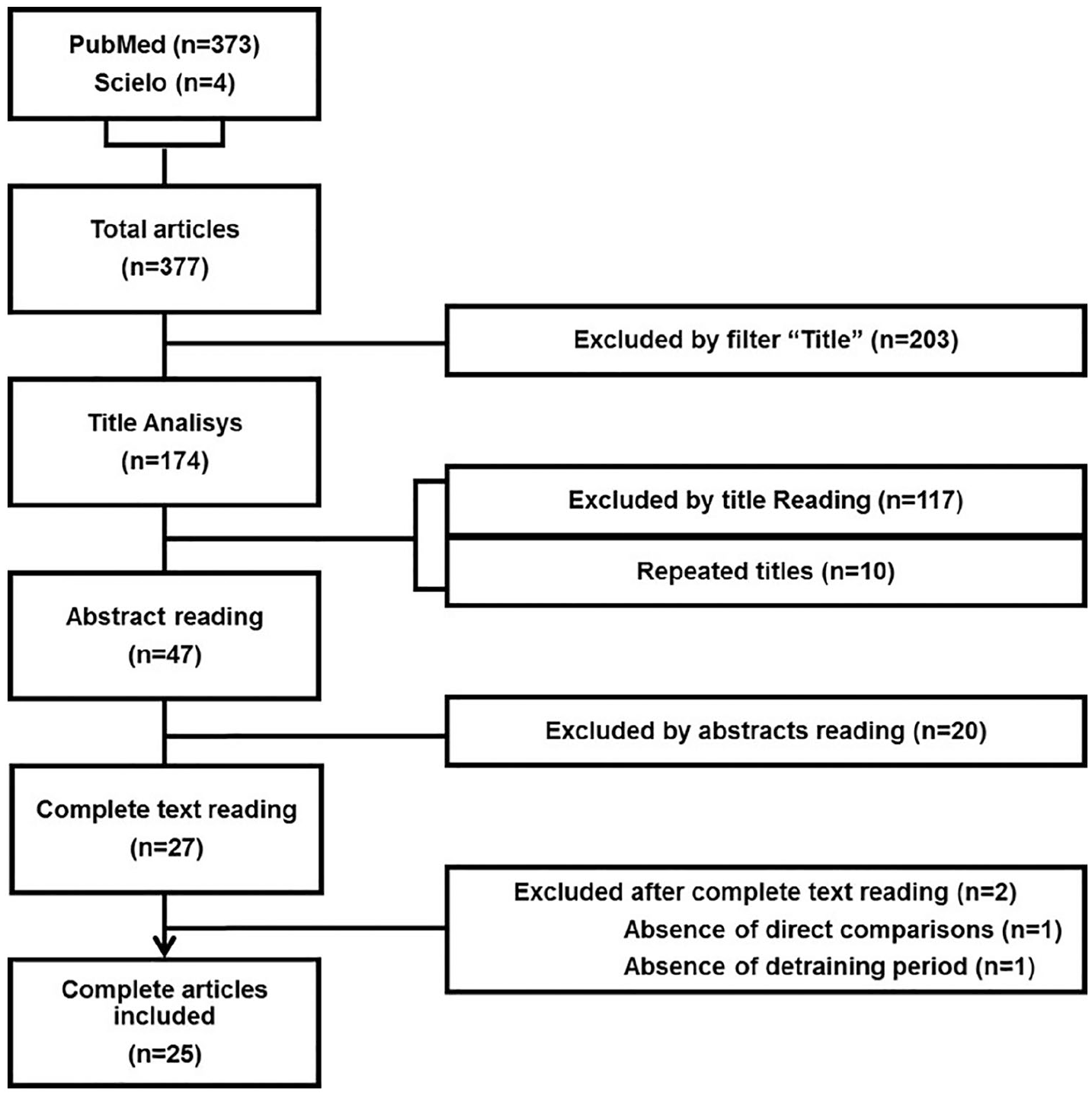
Flow chart illustrating the different phases of the search and study selection.
Results
From the 25 articles inserted in the present review, 12 involved humans and 13 rodents. Together, these articles have a sample of 6772 people, of whom 6406 were from one prospective study 24 and 613 rodents. The mean scores for risk of bias were 10.6 and 11.9 for studies with humans and rodents, respectively, from the 19-item CERT scale. 23 In studies involving humans, the most frequent problems were related to item 5 (detailed description of how adherence to exercise is measured and reported), 6 (detailed description of motivation strategies), 11 (description of the type and number of adverse events that occur during exercise), 16a (description of how adherence or fidelity is assessed/measured), and 16b (description of the extent to which the intervention was delivered as planned). Regarding the studies in rodents, the most frequent issues were found for item 2 (detailed description of the qualifications, expertise, and/or training) and 11 (description of the type and number of adverse events that occur during exercise).
The consequences on fat-related outcomes after cessation of different types of training in humans are summarized in Table 1. Seven from 12 articles measured body/fat mass as the main outcome,24–30 while five analyzed metabolic aspects as main outcomes (i.e., insulin sensitivity, lipogenesis, etc.).31–35 Seven articles had a cohort design, analyzing previous physical activity or exercise, with no intervention,24,25,27,29,30,33,34 while the additional studies investigated responses to endurance and resistance training, mostly using running.26,28,31,32,35 The training period lasted from 1 week 27 to 13 months, 25 while detraining periods lasted 9 days 35 to 7 years. 24
Characteristics of studies with humans included in the systematic review (n = 12).

Final speed after progression; HR: Heart rate; DT: detraining; LPL: lipoprotein lipase; BM: body mass; MSG: monosodium glutamate; SAL: saline solution; §: indirect conclusions based on the study results due the absence of specific discussion on this point; OLETF: otsuka long-evans tokushima fatty; HFD: high fat diet.
The studies involving rodents are summarized in Table 2. Of the 13 studies, five were conducted using voluntary wheel running,16,26,27 while the others used treadmill running. Exercise interventions lasted from 3 16 , 28 to 16 weeks of training. 36 Detraining periods lasted from 53 hours16,28 to 8 weeks. 36 Regarding main outcomes, 5 from the 13 studies investigated changes in body composition,36–40 while 8 analyzed metabolic/mechanistic responses.15,17,41–46 For both humans and rodents, the large heterogeneity among protocols did not allow further comparisons.
Characteristics of studies with rodents included in the systematic review (n = 13).

SN: sedentary normotensive; SH: sedentary hypertensive; TN: trained normotensive; TH: trained hypertensive; DN: detraining normotensives; DH: detraining hypertensives; *Final speed after progression; BM: body mass; DT: detraining; LPL: lipoprotein lipase; MSG: monosodium glutamate; SAL: saline solution; §: indirect conclusions based on the study results due the absence of specific discussion on this point; OLETF: otsuka long-evans tokushima fatty; HFD: high fat diet.
Discussion
The main objective of this review was to discuss the effects of detraining on adipose tissue and the physiological responses related to weight regain. In summary, the compensatory effect is present in most investigations as shown by changes in fat mass or body weight, as well as in fat-related outcomes such as lipid synthesis, insulin sensitivity, transcriptional factors, and others. In rodents, it seems evident that exercise causes compensatory effects. However, some caution is needed regarding the conclusions based on human studies because multifactorial aspects related to human behavior could influence the results, especially when investigations do not control for confounding factors, like diet and physical activity.
Studies involving rodents
It has been previously shown that adipose tissue shows a compensatory effect after stressful stimuli.44,45 Part of the explanation is derived from the so-called “lipostatic hypothesis,” that is, by removing part of the adipose tissue of the body, the organism will strive to recover the removed tissue, increasing its amount. 47
Evidence of the compensatory effect on adipose tissue after exercise cessation is not recent; in 70’s, an “
Braga et al. 36 analyzed body fat responses to detraining in rats performing continuous or intermittent exercise. The animals were divided into subgroups that received 4 milligrams (mg) of monosodium glutamate per gram (g) of body weight or a saline solution every 2 days for 14 days. The continuous exercise involved 45 minutes of swimming with individual overload equivalent to 5% of the animal’s body weight; interval training used effort to pause ratio of 1:1 for 15 seconds with an additional load equivalent to 15% of animal’s body mass, in a total duration of 45 minutes. Both groups trained 5 days a week for 12 weeks. The results revealed that detraining along with early supplementation of monosodium glutamate was associated with an increase in the carcass fat content and epididymal adipose tissue weight in both groups.
More recent studies provided further evidence for the responses of adipose tissue to detraining and its mechanisms. Evidence suggests that chronic endurance exercises may lead to a reduction in resting lipolysis, both in visceral and subcutaneous adipocytes. 45 In this regard, Laye et al. 37 investigated fat metabolism after 5 h and 173 h of the wheel running cessation and found a rapid increase in adipose tissue content and a deficiency in fat oxidation associated with reductions in energy expenditure and PGC-1α expression. The results suggest the occurrence of rapid metabolic changes in animals leads to increases in lipogenic processes and fat gain. Interestingly, there was an increase in the ability to oxidize fat in the skeletal muscle and liver; however, according to the authors, this was not able to prevent the increase in total fat. 37 A similar study by the same group investigated the effects of physical activity interruption in hyperphagic/obese rats and reported an increased risk of hepatic steatosis by increasing hepatic fatty acid synthesis and malonyl-CoA formation due to detraining, with a significant increase in the expression of enzymes involved in fatty acid synthesis. 41
In works by Goodpaster et al. 16 and Lo et al., 28 rats had free access to the running wheel for 21 days and then were analyzed 5, 10, 29, or 53 hours after the exercise cessation. According to the results, 53 hours after exercise cessation, there was an increase in total and relative body mass, an increase in the diameter and the total number of fat cells and a higher concentration of blood triglycerides. 44 In another study, there was an increase in the activity of mtGPAT1, a key regulator in triacylglycerol synthesis, in the detrained animals at 10, 29, and 53 hours when compared to sedentary animals. 15 Moreover, there was an increase in the plasma insulin of mice after 53 hours of exercise interruption. 15 The authors propose two mechanisms to explain the increase of lipid deposition and the reduction of fatty acid oxidation: i) intracellular accumulation of malonyl-CoA, a substrate for the synthesis of long-chain saturated fatty acids via fatty acid synthase; and ii) malonyl-CoA formation, which could reduce fatty acid oxidation by inhibiting carnitine palmitoyltransferase 1 (CPT-1), which is responsible for the transformation of acyl-CoA into acylcarnitine and its entry into the mitochondria. 15 The increases in malonyl-CoA and acetyl CoA carboxylase in rodents submitted to endurance training was confirmed by previous studies. 49 Interestingly, higher velocities were associated with a lower activity of these enzymes. 49
Sertié et al.
17
investigated metabolic and adipocytes changes and after exercise cessation. The study period was 12 weeks, and the rats were divided into the following: (i) control group; (ii) trained group, which performed exercise for 12 weeks; and (iii) detrained group, which exercise for 8 weeks and detrained for 4 weeks. The detrained group showed increased expression of genes associated with adiponectin synthesis (which is related to increases of newly differentiated adipocytes), increased PPARγ gene expression (favorable for adipogenesis), increased
Sértie et al. 46 submitted rats to training, detraining, and control conditions. Training consisted of five weekly sessions of 60 minutes at approximately 50–60% of the maximum running capacity for 12 weeks. The detraining group trained for 8 weeks and then had 4 weeks of detraining. According to the results, exercise induced several positive morphological and biochemical adaptations, but these were reversed during detraining. Moreover, total adipose content was increased in the detraining period, as well as its lipogenic capacity in comparison to controls.
Similarly, Yasari et al. 40 conducted a study involving 8 weeks of exercise, composed by treadmill training followed by 4 weeks of detraining. Some animals also received a high-fat diet (42% fat). Interestingly, the abdominal fat content tended to be higher in detrained animals than in the other groups, suggesting the occurrence of a super compensatory effect due to the cessation of exercise. In addition, the increases in fitness and resting metabolism, obtained after training, underwent a rapid regression with detraining, reaching lower values than control. 40
In an earlier study by Craig et al., 43 rats were divided into sedentary and control groups and trained and detrained groups. Training involved 10 weeks of swimming for 5/days a week for 6 h/day followed by detraining for 7, 14, and 21 days. The results showed an increase in deoxyglucose uptake, indicating an increase in insulin uptake, especially in the first 7 days of detraining. Based on these findings, the authors suggested that MICE was associated with an increased ability to respond to insulin and a consequent increase in adipose tissue. 43
It has been shown that MICE-type exercises increase lipoprotein lipase activity,20,36 lipolysis rate, 50 and insulin sensitivity,37,42 which might promote greater fat and glucose uptake. Detraining does not seem to immediately reverse these responses, as shown by previous studies where 2 or 3 weeks after exercise cessation, lipoprotein lipase activity,20,36 and insulin sensitivity did not return to baseline levels. In this regard, detraining for 1 and 2 weeks resulted in higher insulin sensitivity, GLUT-4 gene expression, and GLUT-4 contents in the heart, gastrocnemius muscle, and epididymal white fat tissue. 38 This suggests that an increased insulin sensitivity might be a key factor in the lipogenic process.37,39
In order to better explain these processes, Sertié et al. 46 compared a control group to a group of detrained rats who exercised for 8 weeks and detrained during the next 4 weeks. The results indicate higher glucose uptake and oxidation in the adipocytes of the detrained group when stimulated by insulin. The association of these results and fat super compensation might be related to the greater lipogenic capacity that would elevate glucose oxidation to provide energy for triglycerides synthesis and storage. While the improvements in the capacity of the adipocyte to uptake and oxidize glucose might be important to metabolic control during the training period, this might lead to accumulating fat after the cessation of exercise. 51
Therefore, the studies in rodents show that that adipose tissues have a compensatory response after exercise cessation, as shown by fat regain and metabolic changes related to increases in adipogenesis and reductions in lipolysis. Interestingly, the exercise protocols generally involved MICE. However, it is not possible to make inferences regarding different protocols because of a lack of evidence.
Studies involving humans
In general, it has been shown that, in humans, when adipose tissue is stressed by different mechanisms, such as liposuction, 13 cryolipolysis, 52 or caloric restriction, 51 it tends to super compensate 19 in the same way other tissues and substrates do.6,8,9 The question is whether these lipogenic and adipogenic changes occur in response to exercise cessation in humans. In this regard, it has been shown that 1 day after MICE (65%VO2max for 90 min), there is an increase in diacylglycerol acyltransferase (which catalyzes the reaction in which the diacylglycerol is attached to a long-chain acyl-CoA to form the triglyceride), mitochondrial glycerol-3-phosphate acyltransferase (responsible for connecting fatty acids to glycerol), and sterol-CoA desaturase, which might induce the production of monounsaturated fatty acids in the liver. 53
After 2 weeks of detraining, marathon runners experimented an increase in the activity of the lipoprotein lipase enzyme in the adipose tissue, with a decrease of this enzyme in the muscle tissue, collaborating to an increased adiposity. 34 There is evidence that blood levels of adiponectin are inversely related to body fat content, 32 and although there was an increase in its levels after 12 weeks of training, the levels decreased after detraining, suggesting an increase in total fat mass, which might be related to the worsening of inflammatory markers during detraining. 32
When young swimmers interrupted training after a competitive season, there is a significant increase in fat mass and waist circumference along with a decrease in aerobic fitness and resting metabolic rate. 29 Moreover, long-term detraining contributed to a substantial increase in body fat in swimmers, apparently by a mechanism of repair and restoration of lipid balance. 25 Data given by Liao et al. 27 suggest that those adverse results could be not exclusive for MICE protocols, since after detraining from specific and vigorous training, taekwondo athletes showed an increase in insulin resistance and body fat. However, it is not possible to make specific conclusions from these data, since the pre-competitive period has additional confounding factors that could interfere with these results, as rapid weight loss (frequent in combat sports) and subsequent rapid weight regain.
When recreationally active young adults performed MICE for 13 weeks and detrained for 9 days, 35 there was an increase in lipoprotein lipase activity and lipemic responses in comparison to the sedentary controls, which indicates higher fat uptake by muscle and adipose tissues. Over the long term, exercise cessation in highly endurance-trained subjects induced compensatory fat gain (~ 6.5 kg), as well as a decrease in high-density lipoprotein (HDL) cholesterol and an increase in body mass index, leptin, and low-density lipoprotein (LDL) cholesterol, even with reduced caloric intake. 54 A super compensatory effect was also observed because of the recurrence of rapid weight loss (lasting 1 to 3 weeks). 55 In part, successive cycles of weight loss and regain seems to stimulate compensatory mechanisms in the adipose tissue and reduce the resting metabolic rate relative to body mass, probably making the organism more efficient in accumulating adipose mass. 56
A previous study with obese subjects investigated the effects of a weight loss program involving caloric restriction and physical exercise and reported a large decrease in body weight, which was accompanied by a reduction in the resting metabolic rate. 57 When the participants of the television program “The Biggest Loser,” which involved caloric restriction combined with physical exercise, were reassessed after 6 years, Fothergill et al. 58 reported a high amount of weight regain, even though the participants maintained a reduced caloric intake and high levels of physical activity. According to the results, this might have been due to a metabolic adaptation, as can be shown by the reduction in the resting metabolic rate.
Considering insulin sensitivity, Bajpeyi et al. 31 showed that overweight and obese individuals maintained an increased insulin sensitivity after detraining, which was dependent on an interaction between training intensity and volume. Low volume/moderate intensity and high volume/vigorous intensity showed longer effects (15 days), while the effects of low volume/vigorous intensity were lost earlier. While increased insulin sensitivity could indicate important improvements in health-related outcomes, the nature of the test (intravenous glucose tolerance test) did not consider tissue-specific behavior, which keeps open the question whether insulin sensitivity could increase glucose uptake (and lipogenic factors) in fat tissue or in skeletal muscle or in both. However, compensatory effects that we are suggesting might be different (or it is not exclusive to) from the insulin-related mechanism, since Rimbert et al. 30 reported that insulin sensitivity was not directly associated with a fat mass gain after detraining in healthy men.
In summary, the cessation of physical exercise, especially MICE, especially those performed in an intensity that promotes high levels of fat oxidation, 10 tends to induce super compensatory effects on adipose tissue by different mechanisms, including an increase in specific tissue insulin sensitivity and increased activity of lipogenic enzymes.
These evidences of a negative impact on fat metabolism and body composition add to the observed negative effects of detraining previously reported in physical function, cardiovascular system, muscle morphology, and metabolism.20–22,59 Considering that excessive fat accumulation is related to many important health problems, it is important to be alert about the risks of exercise detraining; exercise should be performed continuously in order for its benefits to be sustained.
Interestingly, this super compensatory effect might be minimized by the addition of resistance training or high-intensity interval training. 19 ,59 A previous study involving humans analyzed the metabolic effects of a training session composed of 2 min at 90% VO2max by 2 min of recovery at 50% VO2max performed to failure (~90 min). The protocol depleted glycogen stores with no modification in skeletal muscle triacylglycerol. However, 3 hours after the exercise, the skeletal muscle triacylglycerol started to drop until the evening of the following day, while the glycogen was progressively replenished, which suggests that metabolism was shifted to a more lipolytic state to allow glycogen to restore. 60 This “metabolic shift” was also confirmed in later studies that reported higher lipolysis after high-intensity activities 61 and might be related to metabolic consequences of performing higher intensity exercises, such as the increase in lactate levels. In addition, recent evidence shows that lactate is associated with the browning of white adipose tissue, increasing its metabolic activity, and thermogenesis, opening new perspectives for the control of adipose tissue due to high-intensity exercises. 62
Therefore, it is possible that high-intensity interval training exercises do not stress adipose tissue during efforts, avoiding its compensation and increasing adipose substrate and tissue mobilization after training. 63 Interestingly, previous studies suggested that athletes involved in high-intensity activities have similar levels of body fatness than endurance athletes, disregarding the lower energy expenditure and lower training volume of their routines. 64 Coincidently, these athletes are involved with activities that have a low reliance on lipolysis, such as 100, 200, 400, and 800 m. Later, evidence for low body fatness in people involved with higher intensity activities (> 9METs) was confirmed in non-athletes, even in the presence of lower energy expenditure.63,64
There is also similar evidence for resistance training, where short duration and high-intensity modalities promoted higher increases in blood lactate and maintained lipolysis elevated for up to 22 hours after its cessation, while higher volume and lower intensity protocols resulted in less pronounced metabolic responses. 65 Interestingly, this kind of stimulus resulted in higher reductions in body fat than endurance training or lower-intensity resistance training, even in the absence of dietary interventions.66,67 Based on these findings, high-intensity exercises as resistance training 66 and high-intensity interval training 67 should be considered in weight-loss programs.
Limitations
The present study has some important limitations. The more consistent evidences, especially about mechanisms, are from studies in animals. Moreover, while studies in animals provide important information, 68 the application of these results in humans is difficult, 69 especially regarding the magnitude of the changes and the time they take to occur. 70 Moreover, the conclusions based on human studies are limited because multifactorial aspects related to human behavior might influence the results, especially when investigations do not control for confounding factors, like diet and physical activity.
Conclusion
Exercise cessation, especially after MICE, appears to increase the ability of the adipose tissue to store energy. This can be a response to training stress on this tissue and might be a necessary, and expected, response to preserve fat storages. However, caution should be taken, especially with the conclusions based on investigations involving humans, considering the multiple factors that could affect fat metabolism. Data regarding the mechanism of this phenomenon are inconclusive and controversial; while some data suggest that increased insulin sensitivity after exercise cessation might be the main factor for fat accumulation, others failed to show this relationship. Further studies should investigate the effects of exercise cessation on fat metabolism after different types of exercises, including high-intensity interval training and high-intensity resistance training, in order to propose alternatives for more efficient and sustainable with loss.
Supplemental Material
PRISMA-checklist – Supplemental material for Effects of exercise cessation on adipose tissue physiological markers related to fat regain: A systematic review
Supplemental material, PRISMA-checklist for Effects of exercise cessation on adipose tissue physiological markers related to fat regain: A systematic review by Fabrício Boscolo Del Vecchio, Victor Silveira Coswig, Leo Dutra Cabistany, Rafael Bueno Orcy and Paulo Gentil in SAGE Open Medicine
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval was not sought for the present study because this is a review article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: PG receives a research grant from CNPq (304435/2018-0)
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
